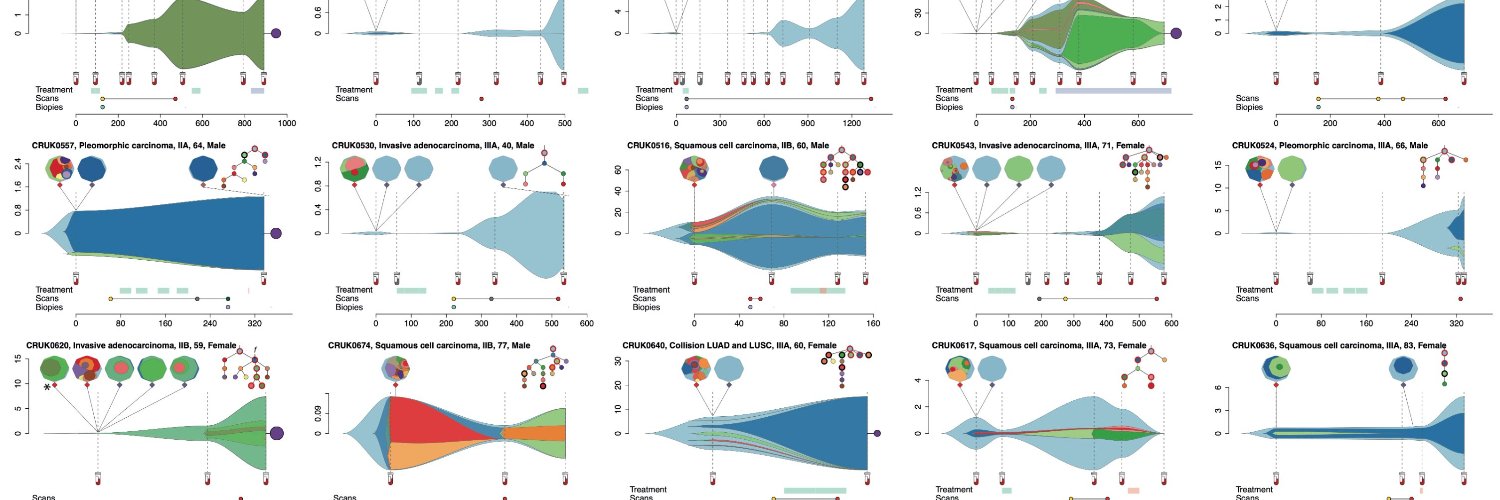
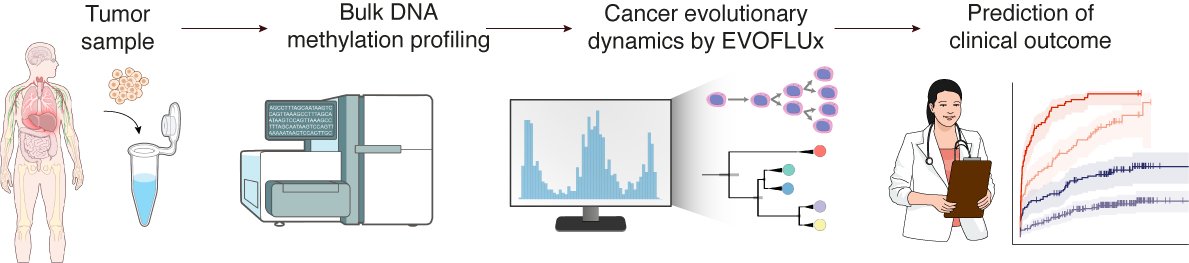
Naka-pin na Tweet

📢 We're hiring! Two posts available in SEM lab @CRUKCamCentre to drive a @wellcometrust funded £2.6M tech dev programme in evolutionary tracking and diagnostics with world-class clinical studies and based in a newly revamped @EarlyCancerCam institute. Deadline Nov 10th! (1/3)


English