Heidi Ko
450 posts


Heidi Ko
@CancerDocKo
Director of Medical Affairs @Labcorp Oncology. Breast Medical Oncologist. Former Hem/Onc Fellow @MontefioreNYC. Former resident @UTHimres. Alum @UNC. #BCSM

pCR after neoadjuvant chemotherapy has long been considered a strong prognostic marker. But adding ultra-sensitive ctDNA changes the picture. In the PREDICT-DNA trial (NeXT Personal @PersonalisInc ), ctDNA-negative patients among non-pCR cases showed outcomes comparable to pCR. @JCO_ASCO Small sample size—but a highly impactful finding. ascopubs.org/doi/10.1200/JC…



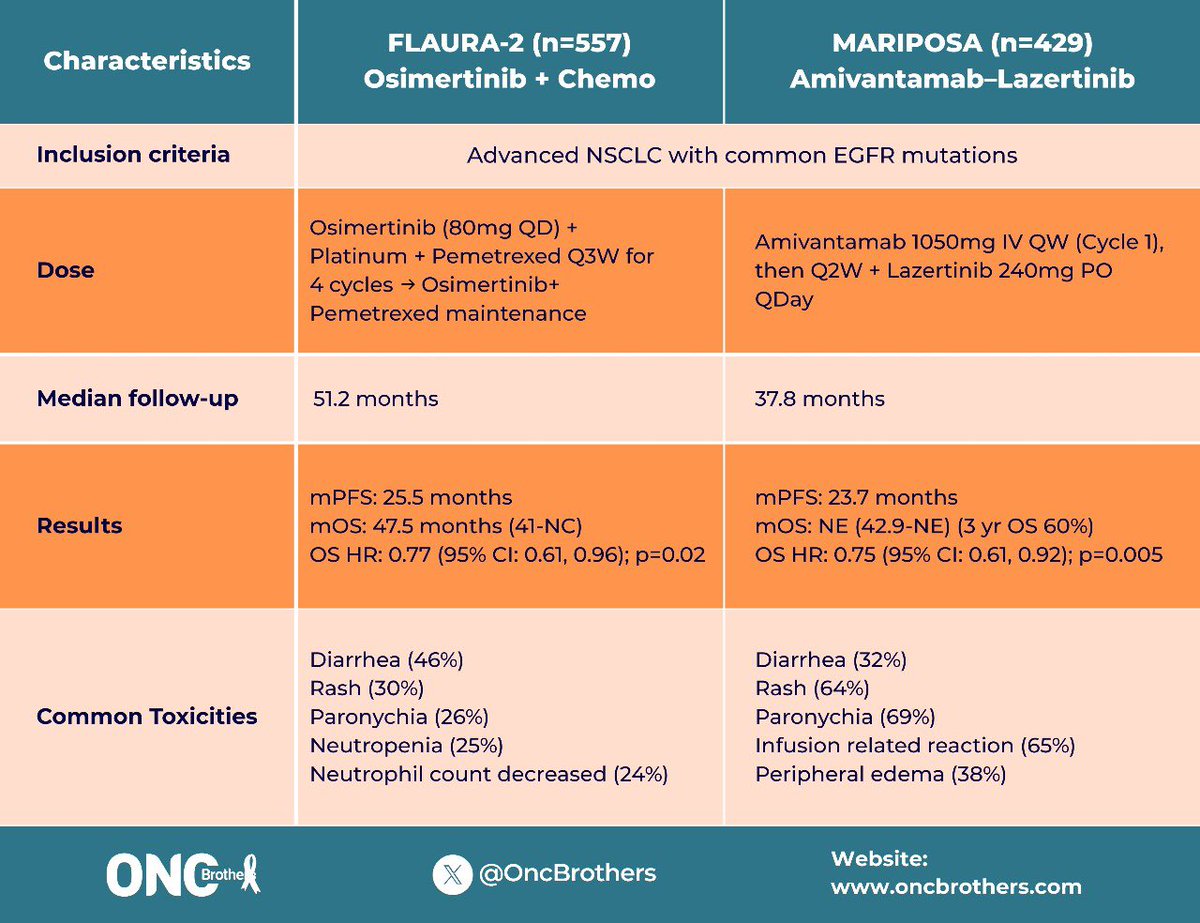
The new FLAURA2 OS data presented at #WCLC25 is exciting - nearly four years of median OS with osimertinib + chemotherapy is remarkable progress for our community! Alongside the MARIPOSA data, it's essential to remember -- this isn't about competition. There is no one-size-fits-all "best" treatment for #EGFR #NSCLC. It's about patients and families. Clinicians need the right tools to match the right treatment with the right patient. That is why I implore companies to invest in research that helps us understand who benefits most from each approach, through biomarkers, disease characteristics, AND A REAL understanding of #QOL from #PROs (Safety/AE grades are endpoints for trials, not tolerability) - THAT is personalized medicine and how progress becomes meaningful. As the discussant @danieltanmd said, we need GPS maps to help guide patient treatment pathways. Options are hope, but only if we know how to navigate them. #LCSM #LungCancer @EGFRResisters #OptionsAreHope














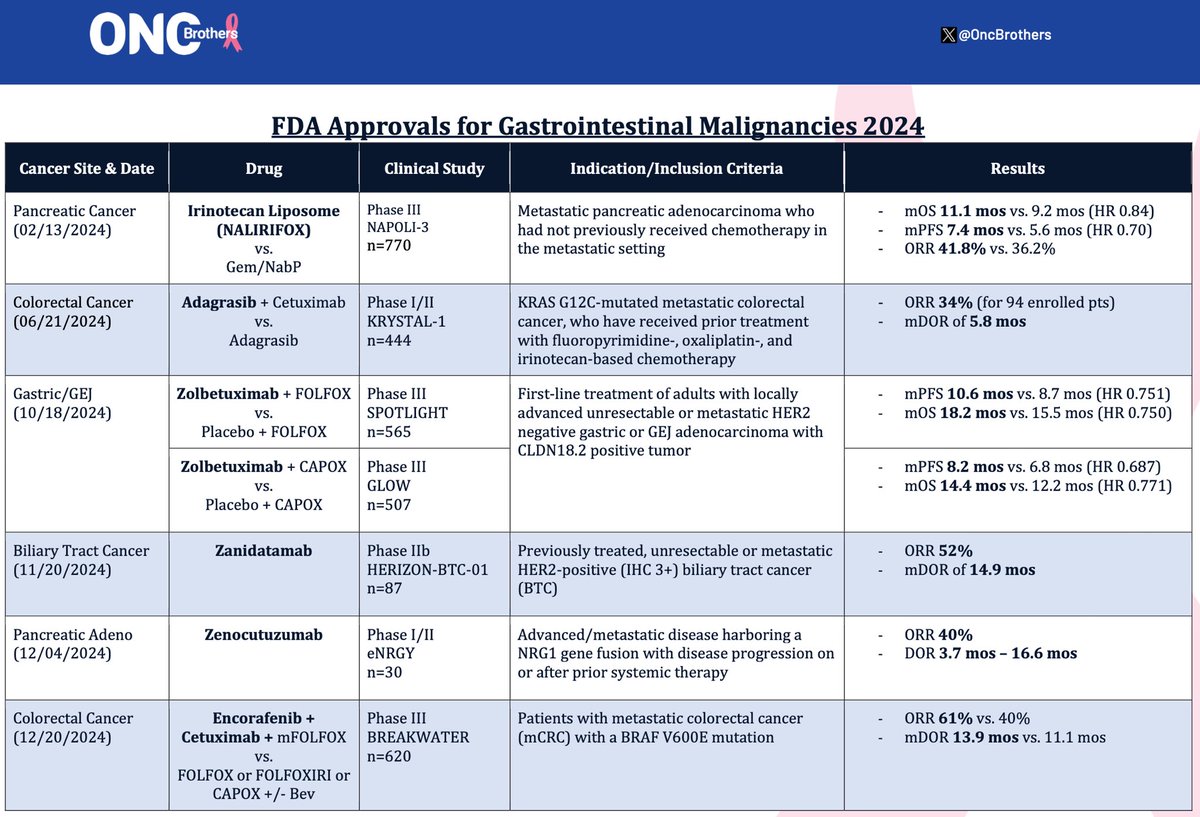
Summary of 5 new drugs/indications in GI malignancies that were @FDAOncology @US_FDA approved in 2023: - #Tucatinib - Mountaineer - #TAS102 + #Bevacizumab - Sunlight - #Pembro + Cis/Gem - KN966 - #Fruquintinib - FRESCO2 - #Pembro + Chemo - KN859 #OncTwitter #gism #crcsm





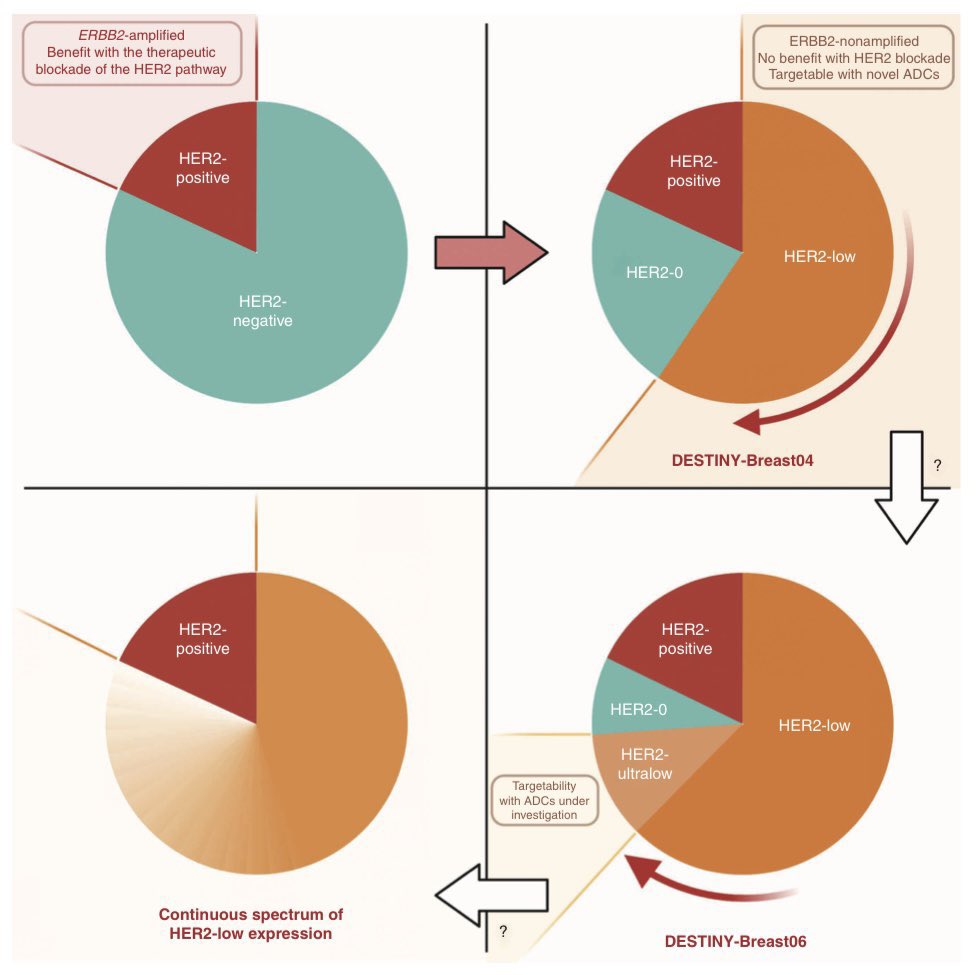
In 2020 we proposed to add a #HER2Low slice to the HER2 pie chart, an addition that has now entered practice. But further changes await us in the coming years. Excited to share our latest Cancer Discovery piece, outlining the present & future of HER2-low! aacrjournals.org/cancerdiscover…