
joe iniowa
4.1K posts


joe iniowa
@Microcapreturns
Long time investor, retired from tech space





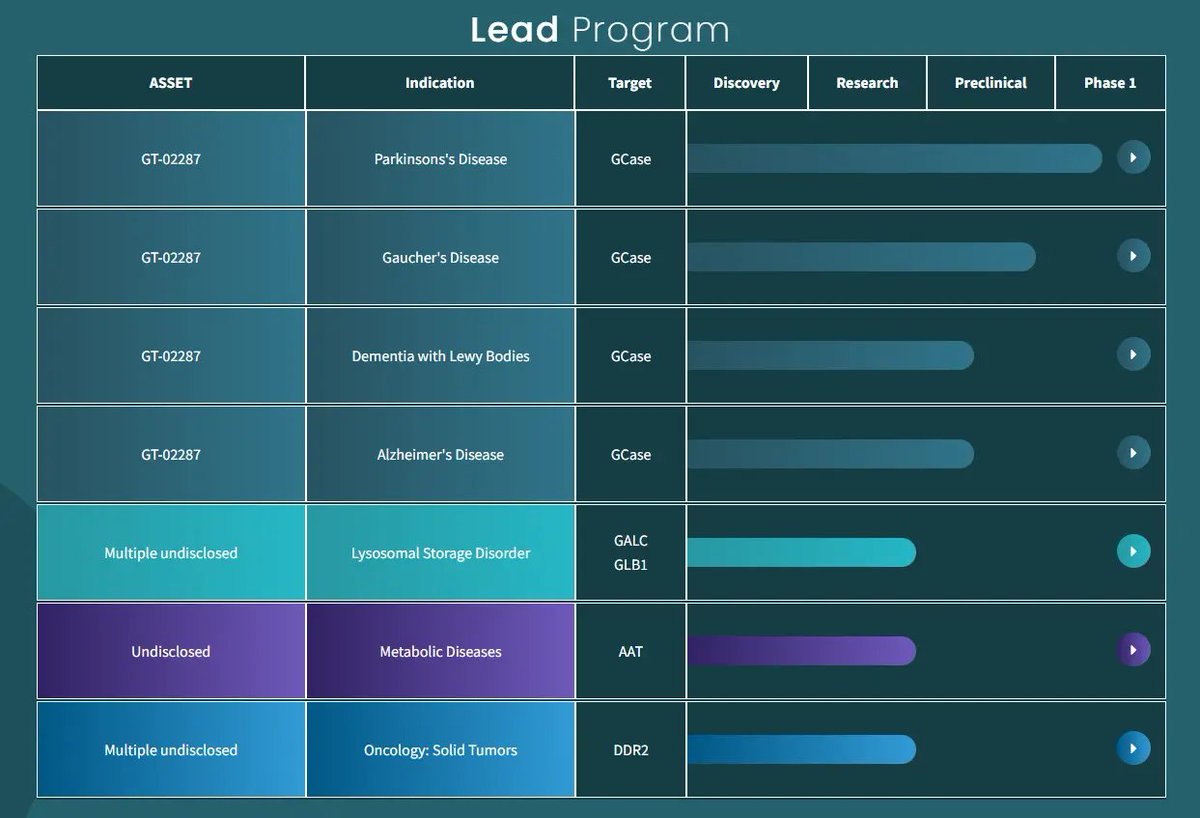
Gain Therapeutics Presents Additional Clinical and Biomarker Data from Phase 1b Clinical Study of GT-02287 and Preclinical Data from Novel Chemical Series at AD/PD 2026 $GANX crweworld.com/article/news-p…


$GANX a twitter friend likes this company

$GANX Unaffected By The Macro Craziness, Healthy Consolidation Here At 3 Month Highs






Now Gain Therapeutics has 2 biomarkers confirming reversal of PD symptoms! $ganx @parkinsons @ParkinsonsUK @ParkinsonsEU @CureParkinsonsT @PunchParkinsons @ParkinsonsPod @APDAParkinsons @ParkinsonsMeds @ParkinsonsNSW

$GANX Update on Gain Therapeutics First, a short summary on what Gain Therapeutics is trying to do in aiming for “disease-modification”… most Parkinson’s treatments help manage symptoms like tremor or stiffness, but they do not slow the disease itself. A disease-modifying therapy is different. It aims to slow, stop, or even partially reverse the underlying disease process, not just mask symptoms. Today, there are no approved disease-modifying treatments for Parkinson’s disease. Gain Therapeutics ($GANX) believes its drug candidate GT-02287 is showing early signs of disease modification — which, if confirmed in larger studies, would represent a major breakthrough not only for Parkinson’s, but potentially for related diseases such as Lewy body dementia, Gaucher disease, and possibly Alzheimer’s disease. February Corporate Update: Gain released an updated corporate deck recently (link in comments), and there are two Phase 1b findings that greatly change the risk profile going into Phase 2: GluSph reduction (upstream biological signal) About one-third (maybe more) of patients entered the Phase 1b with elevated CSF glucosylsphingosine (GluSph), a toxic lipid linked to dysfunction in Parkinson’s (and Gaucher’s disease). In this subgroup, 100% of patients saw GluSph reduced toward normal, with an average ~81% reduction after 90 days on GT-02287. Statistically significant functional improvement That same GluSph-elevated subgroup also showed a statistically significant improvement in combined MDS-UPDRS Parts II + III, with a mean improvement of 6.17 points (p < 0.05). This was actual improvement, not just “less worsening,” and it occurred over ~90 days — which is unusual for programs aiming at disease modification in PD. The “statistical significance” means that within that GluSph-elevated subgroup, the probability that the observed UPDRS clinical improvements occurred by chance alone is less than 5% (p < 0.05). In other words, If GT-02287 had no real effect in that GluSph-elevated group, the probability of observing an improvement this large (or larger) purely by chance is less than 5%. This reduction in GluSph and link to clinical improvements has never been seen before in Parkinson’s patients. If 30% is representative of the number of Parkinson’s patients who have elevated levels of GluSph, this is a giant number. Importantly, it also does not mean (1) that patients who do not have elevated levels will not develop elevated levels in the future, and (2) that GT-02287 would not be beneficial to individuals who do not have elevated GluSph levels. This (very large) sub-population simply looks like low-hanging fruit. For phase 2, this matters because: • It links mechanism to biomarkers to function in humans, not just animals • It identifies a defined responder population, which allows for clear phase 2 planning and setting it up for success • It reduces reliance on noisy, purely clinical endpoints by anchoring outcomes to GluSph • It lowers the chance of a “clean safety but messy efficacy” Phase 2 readout This is what big pharma teams look for when assessing whether early PD signals are real or just statistical noise. The company is explicitly framing this as “translation” from animal models to clinical success in humans. Upstream correction of cellular dysfunction with a strong correlation to functional benefits. CEO Gene Mack succinctly summarized what they are seeing with GT-02287 in a BioSpace interview a few weeks back: “Rather than simply trying to boost enzyme activity in the lysosome, GT-02287 stabilizes GCase folding and trafficking throughout the cell, restoring function across multiple compartments, including those critical to mitochondrial health. That matters because Parkinson’s can be seen as a disease of cellular stress, impaired waste clearance, and energy failure.” 16 out of 19 of the patients who completed the initial 90 days elected to continue into the extension study, despite the further testing and lumber puncture. If they weren’t experiencing benefits, it is unlikely they’d choose to continue. Data from the extension will show whether improvements continue over time. They should already have the 180 day blood and UPDRS data, and judging from the CEO’s statement above, the data continues to support disease-modification. Despite the recent surge, the share price is considerably lower than it was in mid-December pre-data release. Most people will look at this and assume that something must have been wrong. But the market gets misprices companies all the time, especially in small biotechs. To me, this is a great opportunity to buy what might turn out to be the first and most important disease-modifying drug in neurodegenerative disease history. @LouBasenese @PhilipEtienne @RealAvidTrader @BiotechStockRsr @odibro @yaireinhorn @thebiotechforum @BiopharmIQ @BPharmCatalyst @SupNovaTrading @StocksPursuit @Microcapreturns @dixielee1969 @fundmyfund @makedatbread88 @SheffStation @bwsm12702 @TopStockAlerts1

$GANX Rare disease oncology angle with focused pipeline execution. Lean structure keeps milestones meaningful to valuation. Any regulatory or partnership update could quickly reset perception. Are traders positioning ahead of the next catalyst?

Drugging GCase: The Challenges With Targeting A Multi-Compartment Enzyme | drughunters.com/3OeJgqD GCase (encoded by GBA1) is a pivotal genetic risk factor in Parkinson’s and Gaucher’s diseases, where folding/trafficking defects disrupt lysosomal lipid metabolism and drive aggregation. In this review we will highlight: -What made drugging this target so challenging? -How active-site pharmacological chaperones showed promise in rescuing misfolded enzymes but faltered due to lysosomal inhibition -The next generation of CNS-penetrant allosteric activators, including pariceract (BIA 28-6156), GT-02287, and VQ-101 that are currently in clinical trials to test if non-inhibitory modulation can deliver on the promise of the GCase mechanism Read it on Drug Hunter: drughunters.com/3OeJgqD






