Walid El-Sharoud أُعيد تغريده

Almost done with a prototype Rust port of my Scriptoscope python app for transcriptome browsing, all in-terminal. Inspired by @Psy_Fer_'s ask to read from the holy books of Rust.

English
Walid El-Sharoud
2.2K posts

@Walidws
Cell Biologist, University Professor, Editor of Science Progress









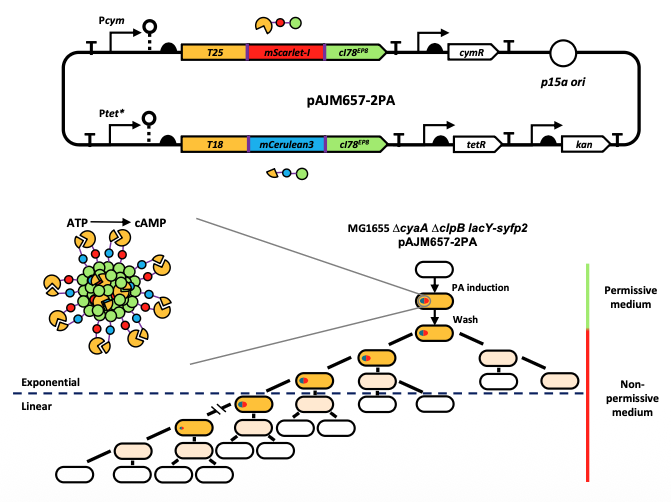
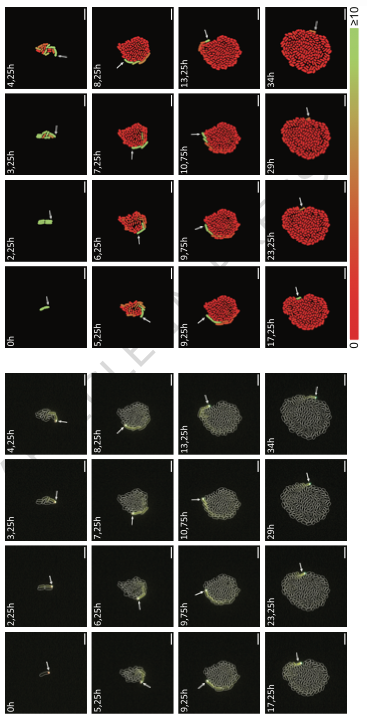


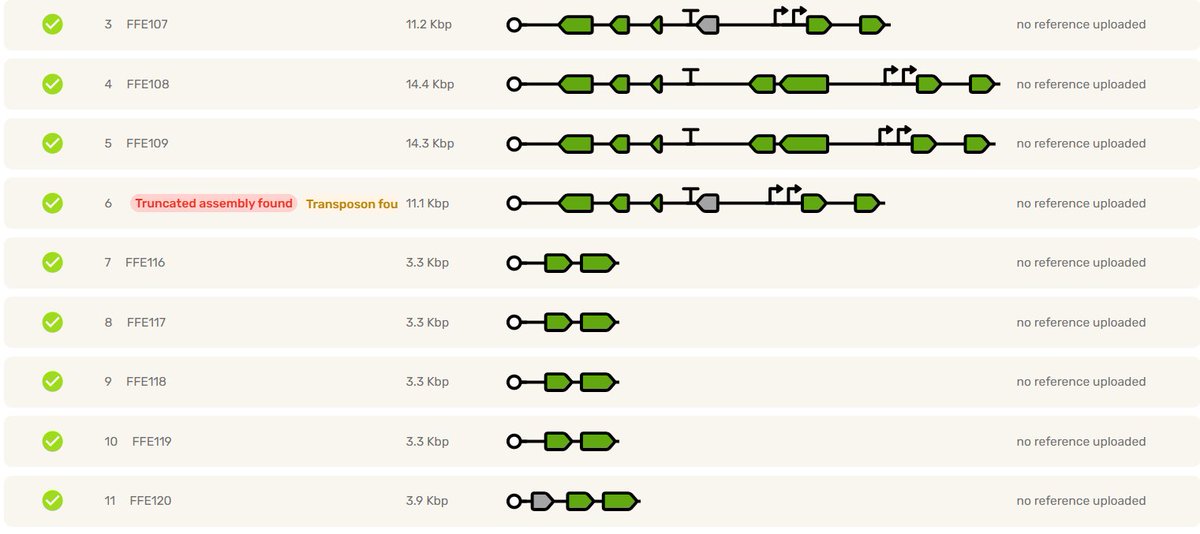





Writing a prompt = ordering a burger. You would 100% be able to prompt a better burger if you actually knew what went into one and had some experience of the flavor profiles of various real-world burgers. You can't be creative without experience.