Peter Adams🇪🇺🇺🇳
1.6K posts


Peter Adams🇪🇺🇺🇳
@AdamsBioAging
Director NCI-designated Cancer Genome and Epigenetics Program at SBP, Co-Editor-in-Chief Aging Biology Journal, Road/Gravel/Mtn Cycling, Wolf Pack Cycling.










If you are in the San Diego area on March 27, 2026 you are cordially invited to attend our inaugural 2026 1st Annual Scintillon Innovator Forum. The main focus of this meeting is aging research. I will be presenting along with other fantastic speakers including Dr. Peter Adams @AdamsBioAging from Sanford Burnham Prebys and Dr. Hideyuki Okano, Scientific Advisory Board Member of @Scintillon, President of @ISSCR and Professor at @Keio_Univ. Registration is free, please read more about our meeting and RSVP here: scintillon.org/2026-1st-scint…






Join us today for Dr. Peter Adams' Johnson & Sokatch lecture, “Aging: It's Role in Cancer and Targets for Interventions." @AdamsBioAging

Do saunas really boost your health? bbc.com/news/articles/…

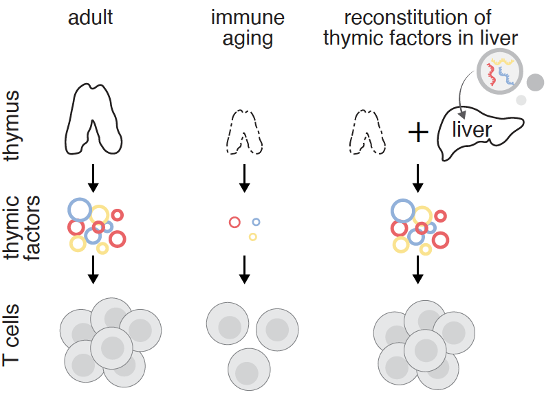
Mitochondrial RNA cytosolic leakage drives the SASP nature.com/articles/s4146… 👉"Furthermore, BAX and BAK play a key role in mtRNA leakage during senescence, and their deletion diminishes SASP expression in vitro and in a mouse model of Metabolic Dysfunction-Associated Steatohepatitis (MASH). These findings highlight mtRNA’s role in SASP regulation and its potential as a therapeutic target for mitigating age-related inflammation." 🔥 Fig. 1: Cytosolic mtRNA leakage is a feature of senescent cells. 👨⚕️👇