Etienne Gauthier retweetet

Making C-glycosides SWEET and simple! Today in @ChemRxiv we disclose (chemrxiv.org/doi/full/10.26…), in collaboration with @GroupAggarwal, an incredibly easy way to achieve radical functionalization of sugars. In this video (youtu.be/Fqdbgmx7zEI), a two-step synthesis of the billion dollar drug Dapagliflozin is achieved using household vinegar and dextrose powder from the local supplement store.
High Level Summary:
The work addresses a longstanding challenge in carbohydrate chemistry: the efficient, scalable, and stereocontrolled synthesis of C-aryl glycosides directly from unprotected native sugars. C-Aryl glycosides form the core pharmacophore of the SGLT2 inhibitors (dapagliflozin, canagliflozin, empagliflozin, and related agents), which are frontline therapies for type 2 diabetes and represent one of the highest-grossing classes of small-molecule drugs.
Conventional synthetic routes to these molecules generally require extensive protecting-group manipulations, multi-step activation of glycosyl donors, or organometallic additions under demanding conditions. Recent advances in radical and transition-metal-catalyzed cross-couplings have improved access, yet most approaches still depend on protected precursors, specialized reagents, or protocols that are difficult to scale.
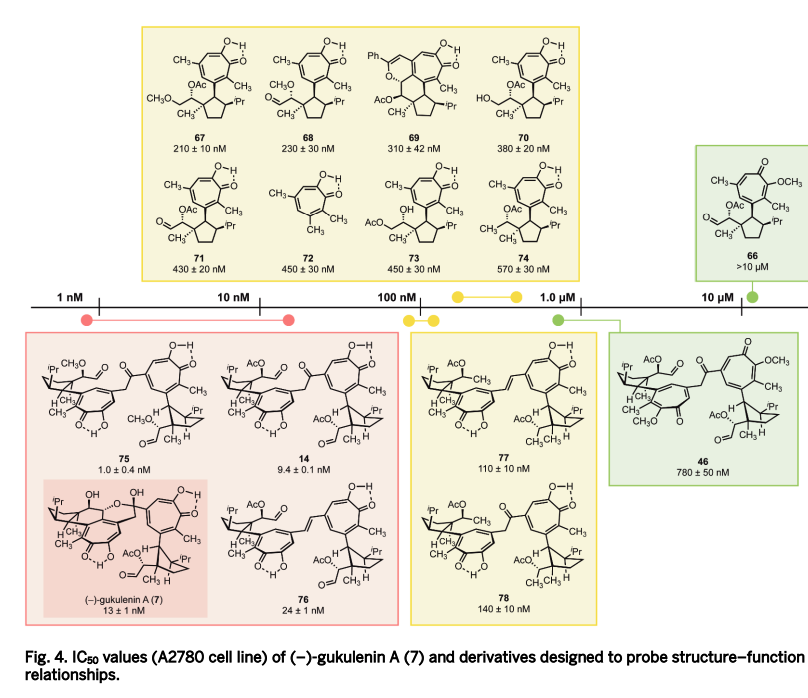
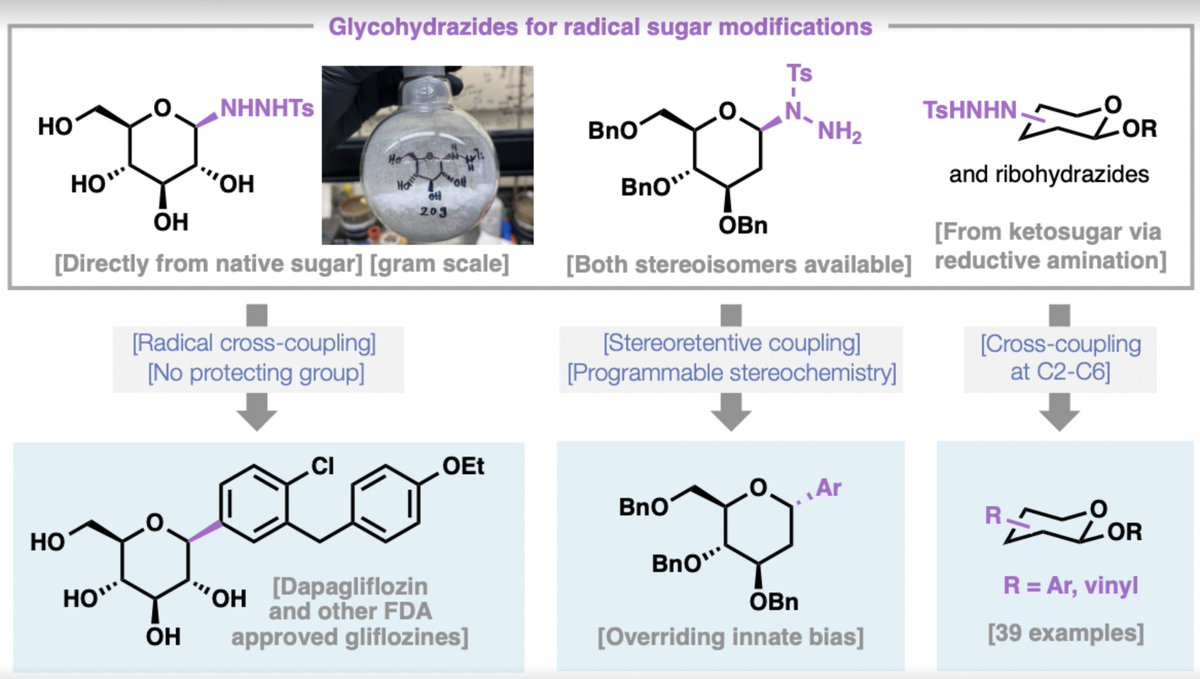
We report a practical alternative based on glycosyl sulfonyl hydrazides—stable, crystalline radical precursors that are prepared in a single step from unprotected sugars by treatment with tosylhydrazine in acetic acid, followed by simple crystallization. These hydrazides undergo redox-neutral nickel-catalyzed radical cross-coupling with aryl iodides or bromides under mild conditions (70 °C, DMSO, tetramethylguanidine as base). The reaction requires no external oxidant or reductant, no photocatalyst, and no organotin species. In glucose-derived systems the coupling typically delivers high β-selectivity (>19:1 in many cases), an outcome that appears to depend on hydrogen-bonding interactions between tetramethylguanidine and the free hydroxyl groups.
The main findings are as follows:
All five FDA-approved SGLT2 inhibitors, as well as several clinical candidates, can be prepared in a single coupling step from the corresponding glycohydrazide.
Decagram-scale synthesis of dapagliflozin was demonstrated starting from commercial dextrose; the product was isolated by aqueous workup and recrystallization (no column chromatography required at this scale).
Di- and trisaccharides (lactose, cellobiose, maltose, maltotriose) couple directly to give aryl-linked oligosaccharides.
Several natural products and medicinally relevant structures (salmochelin-SX, neopetrosin C, the tryptophan-mannose conjugate, and a ribose-derived IMPDH inhibitor) that previously required 9–20 steps or costly reagents are now accessible in 1–4 steps with good stereocontrol.
The platform extends to non-anomeric C–C bond formation at positions C2–C6 on glucose and ribose scaffolds, providing the first systematic exploration of radical diversification across these positions.
Stereoretentive radical cross-coupling, using configurationally pure hydrazides, enables programmable delivery of either α- or β-anomers, overriding inherent substrate biases and providing access to stereoisomers not previously obtainable by radical methods.
The chemistry builds on our earlier development of sulfonyl hydrazide-based redox-neutral cross-coupling and stereoretentive radical arylation, here adapted and optimized for carbohydrate substrates. The method is operationally straightforward, uses inexpensive reagents and starting materials, and eliminates protecting-group strategies.

YouTube
English