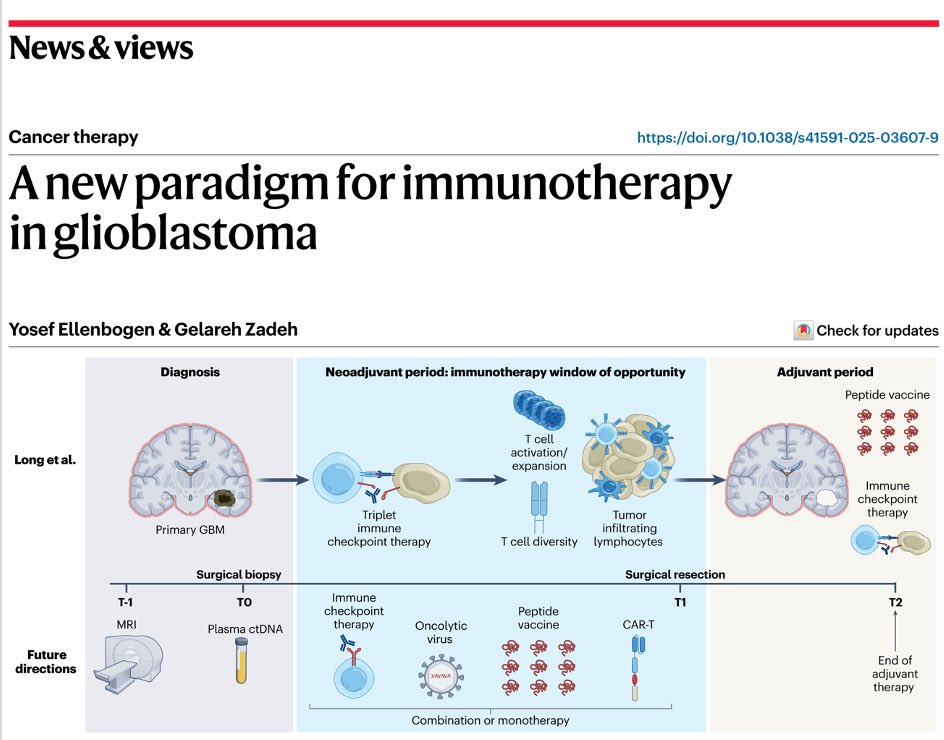
Published yesterday (Feb 27, 2025), a groundbreaking case study by Georgiana Long et al. may redefine GBM immunotherapy. The team reports the first known instance of upfront neoadjuvant administration of immune checkpoint inhibitors (a-PD-1 + a-CTLA-4 + a-LAG3) before surgery in GBM, demonstrating robust anti-tumor immune activation and extending survival well beyond the typical prognosis for MGMT-unmethylated GBM. After a single neoadjuvant triplet dose and maximal safe resection, the 56-year-old patient remains recurrence-free for 17 months. These findings have led to a planned first-line neoadjuvant immunotherapy trial (NCT06816927) in newly diagnosed GBM. If confirmed in larger cohorts, this approach could radically change clinical practice, offering renewed hope for patients battling one of the deadliest cancers.