Tristan Cordier
2.3K posts


Tristan Cordier
@SharkStats
Views are my own fleeting reactions. Statistics, Healthcare, Tennis, Soccer, Cooking, Family, Friends. We're all just atoms, bouncing around.


Elon Musk and Jeff Bezos shouldn't be paying the same amount in Social Security tax as someone making $175,000 a year. Billionaires are not paying their fair share.


Matteo Berrettini defeats world #10 Daniil Medvedev 6-0, 6-0 in 50 minutes to reach the last 16 in Monte-Carlo for a 3rd time. What the...


If you’ve been wondering why I can’t let go of the ousting of Vinay Prasad from the FDA, here it is, plain and unvarnished. He was a once in a lifetime hire. Prasad was one of the strongest appointments the agency has ever made, and the FDA should have fought like hell to keep him. That doesn’t mean I agreed with every call. For example, I support advisory committees. But it’s worth noting that advisory committees were already in decline prior to his appointment. Besides, disagreement with one aspect does not negate the whole. The failure to retain Prasad was a consequential mistake; it signals exactly how the agency, the media, and the investor class handle serious challenges to the status quo. And the backdrop makes it worse. Prasad walked into the CBER director’s office after a truly rough stretch in FDA history: overly broad use of COVID vaccines, with related resignations of two top vaccine officials, Paxlovid cheerleading, Elevidys and Aduhelm debacles, and misguided misinformation-policing, among others. This stretch had eroded public trust. He inherited a scarred institution, and the expectation was that he would challenge the status quo and repair the perception of the FDA as acting merely as a rubber stamp. What’s been maddening to watch is the yawning gap between what everyone claims to want, like higher evidence standards, fewer weak approvals, more appropriate use of accelerated approval, and how violently the system recoils the moment someone tries to deliver it. Modest, data driven moves were treated like heresy. The loudest critics rarely engaged the substance; they only amplified an outrage coming squarely from the perspective of pharma and investor interests. A clearer accounting of what actually changed and what didn’t during the time Vinay Prasad was at the FDA is still worth laying out. Accomplishments included the COVID vaccine framework, the Elevidys label narrowing, and proposing to strengthen CBER research and vaccine standards more generally. The Moderna flu vaccine refuse to file decision was an example of using an unconventional approach. It was resolved quickly in a manner that will result in better evidence generation, but with much media fanfare. And worth noting: not all of his accomplishments were around tightening standards. He co-authored significant approaches to introduce regulatory flexibility and support true innovation: formal guidance documents for the new plausible mechanism pathway and Bayesian methodology. Both of these are being praised, but no one is bothered that a key architect on these projects is leaving the FDA? Not to mention the single trial paper. Instead, the public narrative chose to fixate on three leadership overrides, endlessly framed as “chaos” or a break from sacred norms. Conveniently omitted: every single override aligned decisions with the new COVID vaccine framework that Prasad had developed to navigate uncertainty without rubber stamping. Approval volume stayed roughly in line with historical norms. Rare disease decisions weren’t some override spree; they were part of long running debates over accelerated approval pathways that predate Prasad by years. Reasonable people can quarrel over individual calls. What’s indefensible is pretending the record was defined by recklessness rather than a short, intense effort to tighten standards in an agency long accused of being too cozy with industry. ...






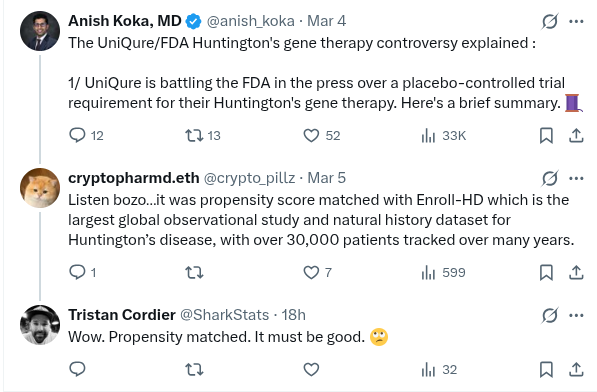





FDA official calls UniQure's gene therapy a 'failed' treatment for Huntington's disease cnbc.com/2026/03/05/fda…






FDA formalizes its shift to requiring only one pivotal trial (via NEJM) and analysts are already pointing at companies this could help -- another pro-pharma push for Makary, and another 180 for Prasad who wrote extensively in his academic life against trials focusing on anything less than overall survival endpoints.news/fda-formalizes…


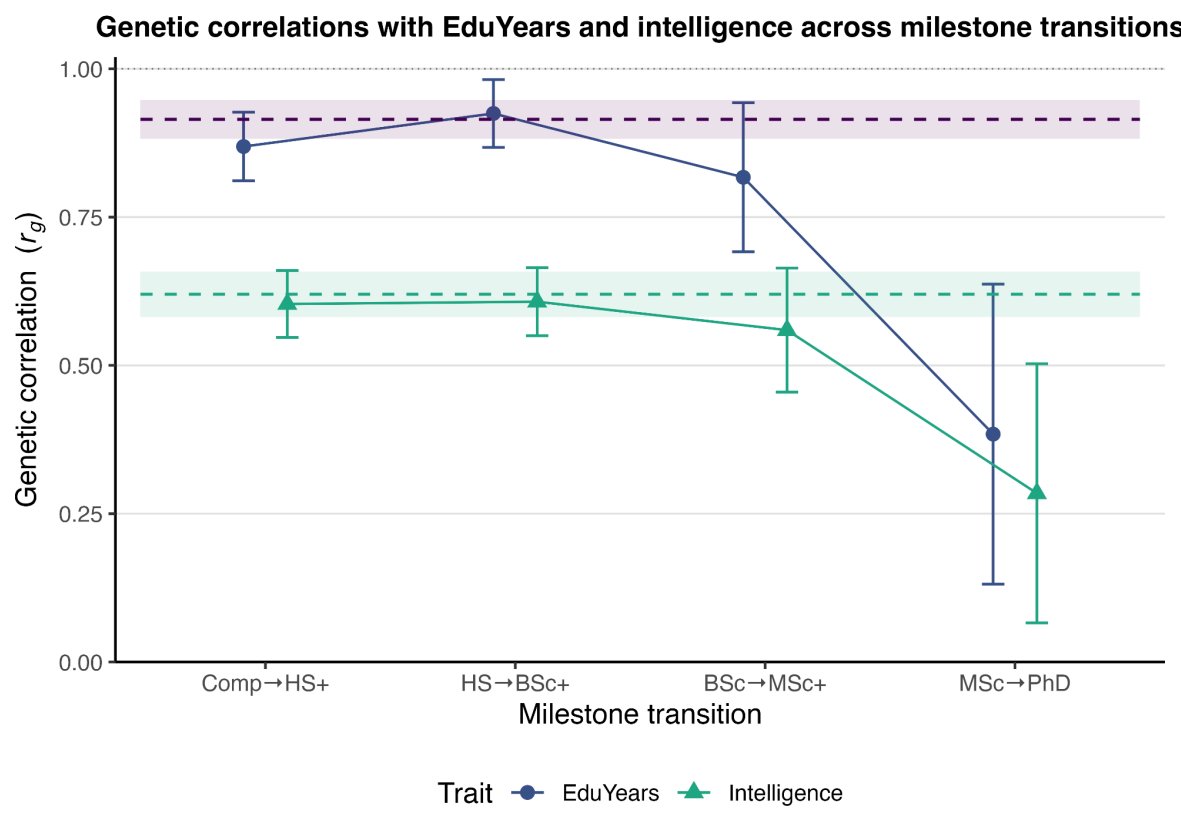
Educational attainment is a garbage phenotype: what it measures varies radically over time, types of education, and so on. As it turns out, once you get to the PhD level, its meaning radically drifts. Earning a PhD is less about intelligence or earlier EA than other EA levels: