
World's fastest man at the #EnhancedGames belongs to Fred Kerley (9.97s). Captures the $250,000 grand prize.
Jing Liang 🇺🇦
36.1K posts


@AppleHelix
Entrepreneur. Drug hunter. Anti-ideologues, Anti-medical nihilists, Optimist My NGO: https://t.co/yDWRnOYAyY https://t.co/SOf8c5OZm6

World's fastest man at the #EnhancedGames belongs to Fred Kerley (9.97s). Captures the $250,000 grand prize.


PROTRACT (#ASCO26, Abstr 5017) — ctDNA-guided treatment selection in mCRPC after abiraterone. 🔹 The idea Use ctDNA fraction (ctDNA%) to pick the next therapy. Rationale: high ctDNA% (≥2%) predicts a poor response to a second ARPI but preserved sensitivity to chemo. So the biomarker arm sent ≥2% → docetaxel and <2% → enzalutamide, vs clinician's choice. 🔹 The trial Small randomized phase II, stopped early for slow accrual — only 42 pts (17 biomarker-directed vs 25 clinician's choice). 🔹 Results Both PFS (5.6 vs 2.5 mo, HR 0.4) and OS (46.3 vs 15.3 mo, HR 0.4) favored the ctDNA-guided arm. PSA50 numerically better but not significant. 🔹 My take: Nice study! Thought-provoking, but very small numbers. Most pts on clinician's choice got enzalutamide (second ARPI), which tends to be the weaker option after abiraterone (à la CARD trial). So it's unclear whether the benefit is really from better biomarker selection or just from the guided arm using docetaxel more often (11/17 vs 4/25). What I'd really want to see is how the pts whose biomarker favored ENZA actually did on ENZA — IMO that's the real test of the biomarker. Looking forward to seeing that. 🔗 asco.org/abstracts-pres… #pcsm #ctDNA #ASCO26



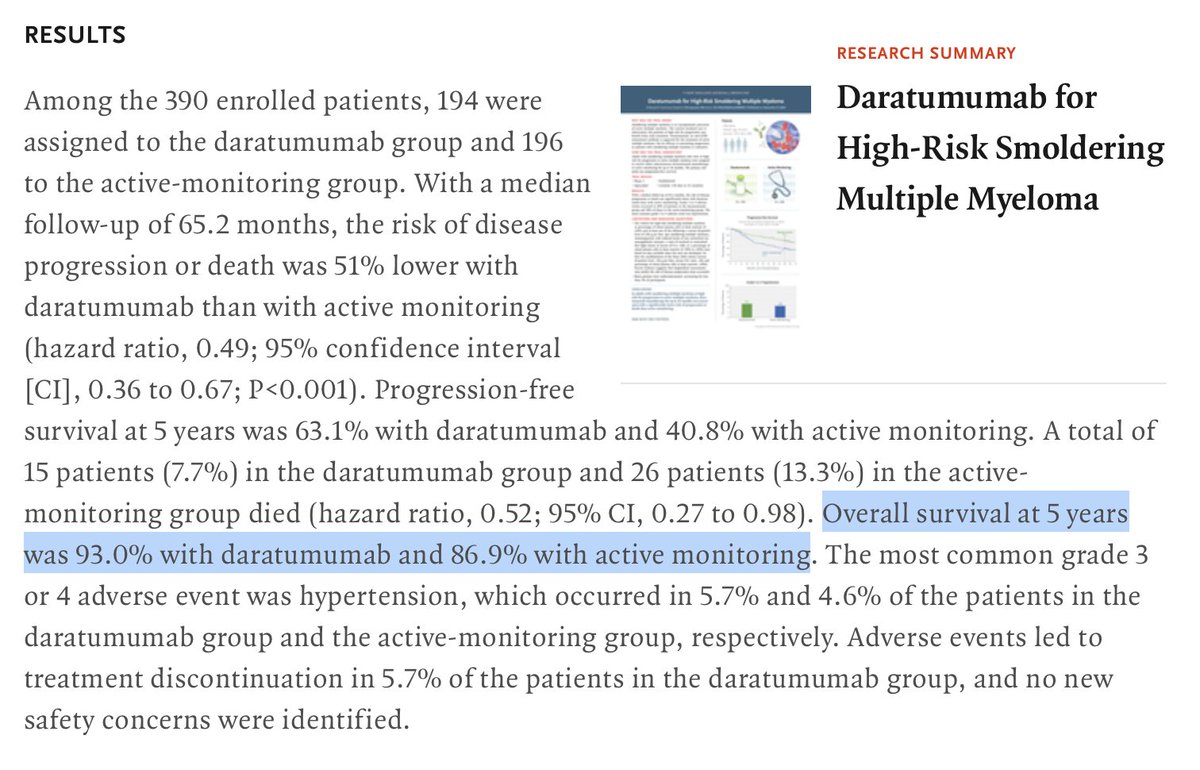
Daratumumab prevents progression of asymptomatic to asymptomatic 5≥60% plasma cells in dara vs 16 in control-no symptoms! 2 anemias in dara vs 14 in control (below arbitrary threshold 2 pts ⬇️ lower limit) 10 cases bone dz in dara vs 18 control 0 fractures/0 renal failure






When consenting your patient for daratumumab for smoldering myeloma please remember to tell them dara increases risk for severe infections that will hospitalize you Grade III/IV infection (severe infection that requires hospitalization) was 16.1% in dara arm vs 4.6% in control


I find the ODAC vote on camizestrant a bit depressing. This is what futuristic cancer treatment looks like. You develop a drug that targets a key resistance mechanism for breast cancer. You develop a non-invasive ctDNA blood test to surveil the emergence of this resistance mutation. You only give this drug when this mutation was detected, but cancer is still being controlled. "Nip it in the bud" so to speak. You demonstrate much better PFS than SOC (HR 0.44-0.46). The drug obviously works! But you have FDA and some ODAC members using the red-herring which are basically: - How can you ensure there is a OS benefit after patients moving onto subsequent lines treatments - How do you know if you should give this drug early (at detection of resistance mutations) or when it is full blown progression To answer both of these questions, you need a much larger trials. If this is the bar for cancer drug approvals, there will be fewer MOAs investigated. The total amount of R&D dollars investors and pharma companies willing to deploy is relatively fixed. On top of that, there are only so many patients you can recruit. If you demand large trials for a particular MOA, this means fewer patients available for other trials. The regulatory bar that drives the most human progress is not necessarily the bar that gives you the most certainty. fiercebiotech.com/biotech/astraz…

🔥 NEWS Camizestrant with a CDK4/6i recommended for approval in the EU by CHMP for advanced ER+ BC at detection of ESR1 mutation, before clinical progression After all the post-ODAC discussions, for me this is a wonderful news, especially for patients astrazeneca.com/media-centre/p…



Built a free biologics COGS estimator covering mAbs, recombinant proteins, ADCs, and C>, with GMP/Tox/RG options. For founders, investors, and BD folks who need a defensible cost figure for pitching/evaluating. Feedback welcome; still iterating! biologics.tools







Here you go Ron. Hope this helps