Bertrand Delsuc
91.7K posts

Bertrand Delsuc
@BertrandBio
Views=mine only, RT-like=/=endorse, no advice. 16y in Tech (chip design) & 10y following bios. Preplanned pause.




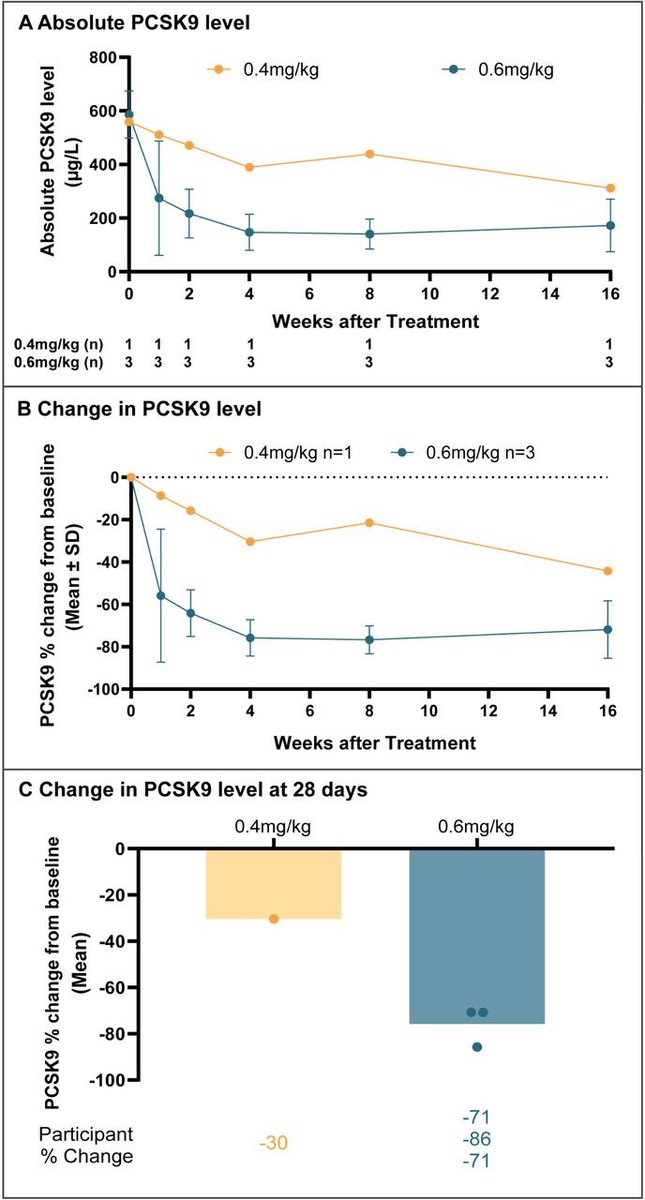










No comments needed 🤩🥳😍 congrats super @stolaney1 from @DanaFarber and all involved in the #DB09 trial !!!! #ASCO25 @OncoAlert @ASCO @DFCI_BreastOnc






In oncology, we don’t usually love overlapping Kaplan-Meier curves. Except that sometimes we really do. Thanks to NSABP-B51, countless pts will be spared the toxicity of nodal irradiation, and many will live free from a permanent lymph edema that would have impacted their lives.

Listen to Deep ASCO Review; BioNTech & Regeneron Deals: BI Drug Call; Bloomberg Intelligence #np on #SoundCloud on.soundcloud.com/MnqU57Bv7gQkjB… $REGN $HNSPF $JNJ $BNTX $BMY $SMMT $TIL $PFE $TRSBF $AKESF $AZN $IVBXF $RHHBY $JAZZ $AMGN $ZYME $RVMD $DSNKY $GILD $MRK $RCUS $LEGN $MRUS $BCAX








