
Emmanuele
69.1K posts


Emmanuele
@Biotech2050
No investment advice. Do your own DD Market is all about taking the shares from the weak holders& providing opportunities to those that have the stamina to hold



$TGTX almost daily now you see Ocrevus patients switching to Briumvi sighting “crap gap” often “I was on O for 6 years. Crap gap kept being earlier and earlier. Switched to Briumvi 2 years ago. It has been nothing short of a normal life with no crap gap. @1capplegate will only accelerate starts/switches




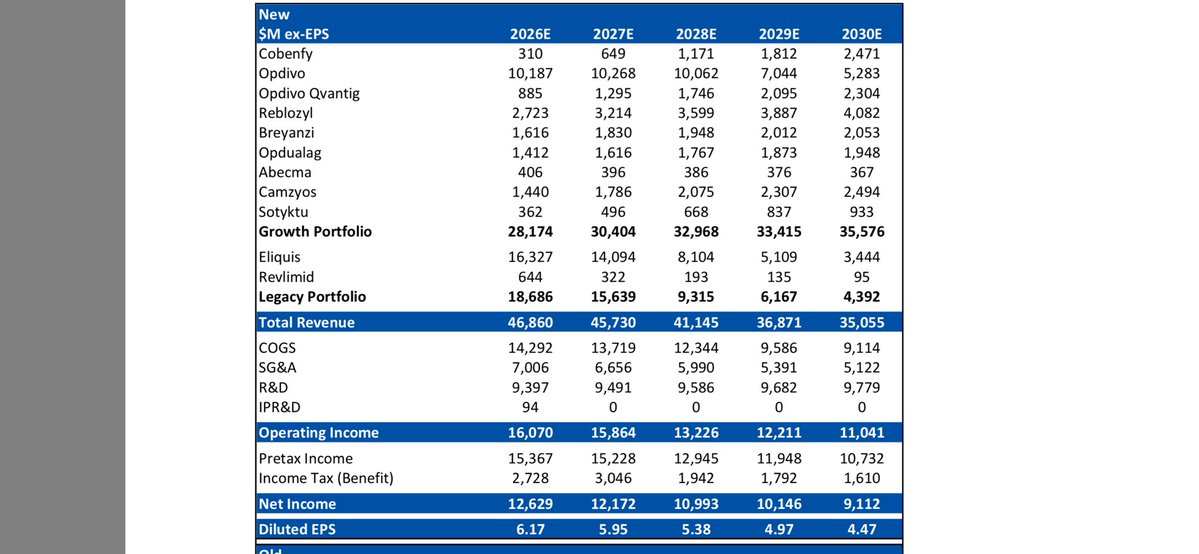
$BMY why it needs multiple deals - Revenue will decline in 2026 - 2030 8 billion ?? $XBI #Biotechs

@JacobPlieth Special request from an ex Celgene holder. Can you pls get feedback on $BMY celmods iberdomide (CC-220) and CC-92480. Maybe also have a chat with @SagarLonialMD at #ASH23 A video of his on the “Benefits of CELMoDs over IMiDs in multiple myeloma” #mmsm youtube.com/watch?v=i1pfSm…

Dr Sagar Lonial @SagarLonialMD discussing CC-92480 CC-220 #mmsm $BMY @grpetersen1 youtu.be/VeFjDVVjpqw



$tgtx another big detail just noticed. In cohort 1 - 62% experienced the “crap gap” with Ocrevus In cohort 2 - 57% experience the “crap gap” With Ocrevus This is super high & all wud b potential switches imho. That’s a huge positive

$TGTX corresponding to recent study published in the journal MSology, a whopping 61% of Ocrevus patients in the study experienced the dreaded "crap gap." That's a pretty significant percentage!


$TGTX JP pic showing $TGTX -#Briumvi Duration/retention coming into full view

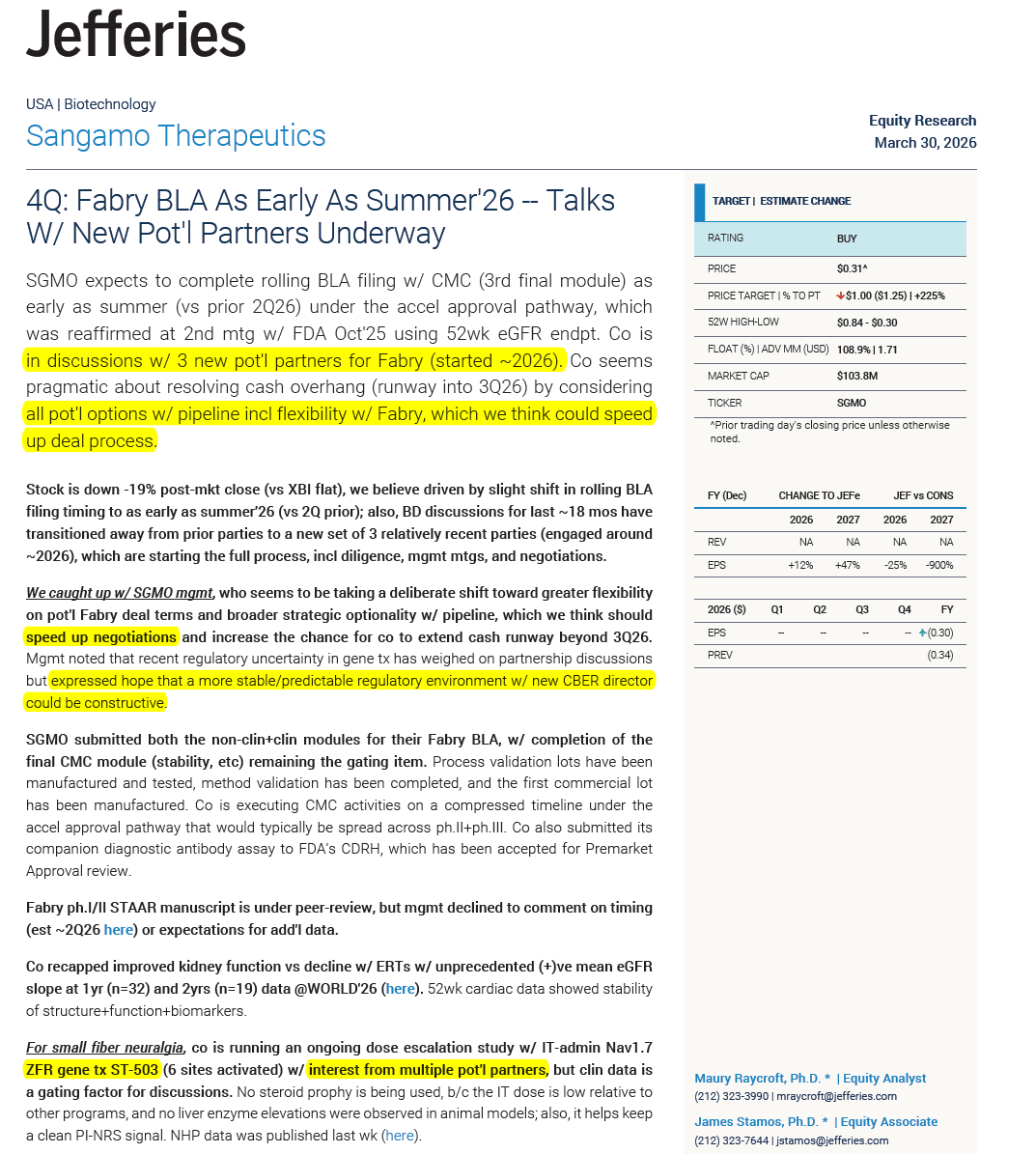


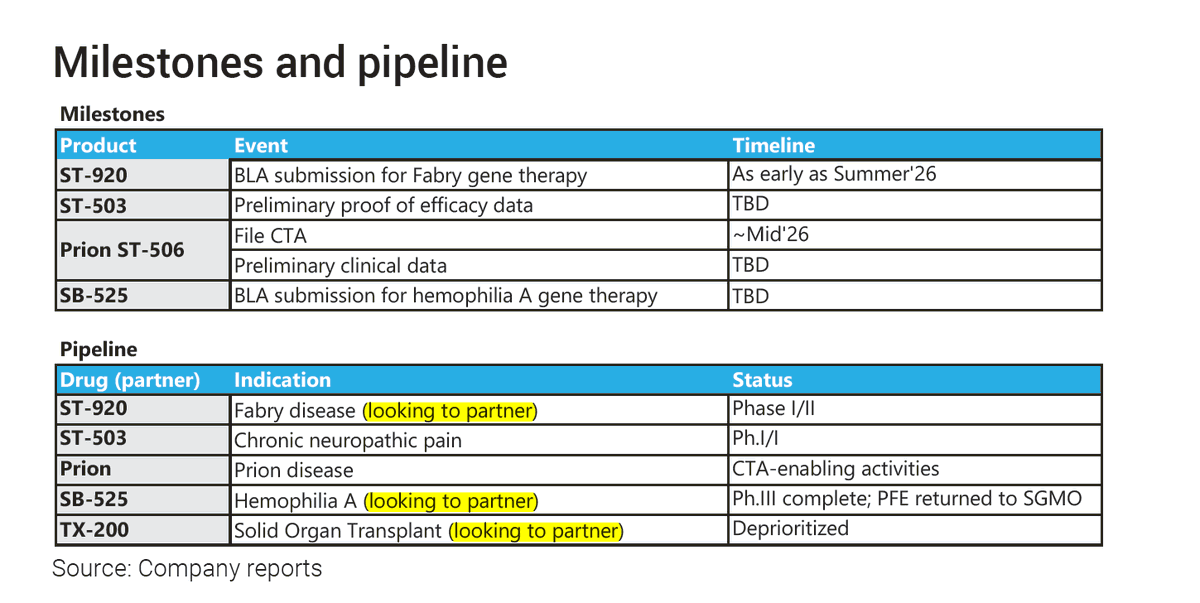

$SGMO I expect nothing but the usual word salad. Promising conversations regarding licensing deals. One of those (MINT) already in very advanced discussions. They might also highlight that partners are really excited about their progress in pain and highly anticipate the prion trial start — expecting new deals and potentially the first milestones within 2026. The biggest update is going to be that they’ve started the pain trial and are already advanced in dosing (25+ patients dosed, I hope), with first data potentially before the end of Q2 — and that they feel very positive about initial observed tolerability and safety. I expect to hear that the Fabry BLA submission is expected to be completed within the next 4–7 weeks and that they are very positive about it. I don’t expect clarity on the commercial path forward (whether asset sale, solo commercialization, or a partner). I also expect cash to arrive and a timely solution to boost SP and avoid delisting, but I can’t imagine them to announce something like that during ER call anymore… So I’d take any positive cash news as a positive surprise…


$SGMO We are excited to announce the publication of a manuscript in #ScienceTranslationalMedicine detailing the preclinical safety and pharmacology of ST-503, our investigational epigenetic regulator for the treatment of chronic neuropathic pain. investor.sangamo.com/static-files/0…









