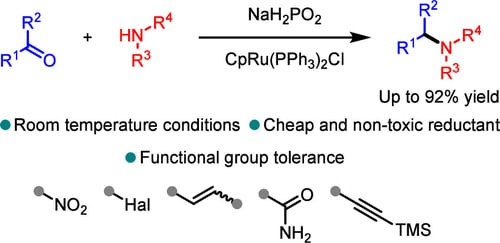
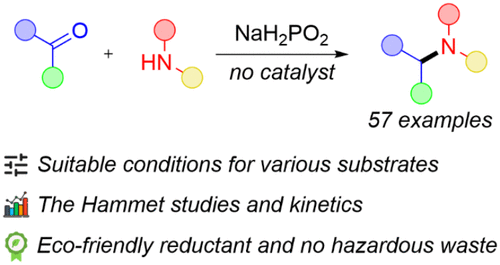
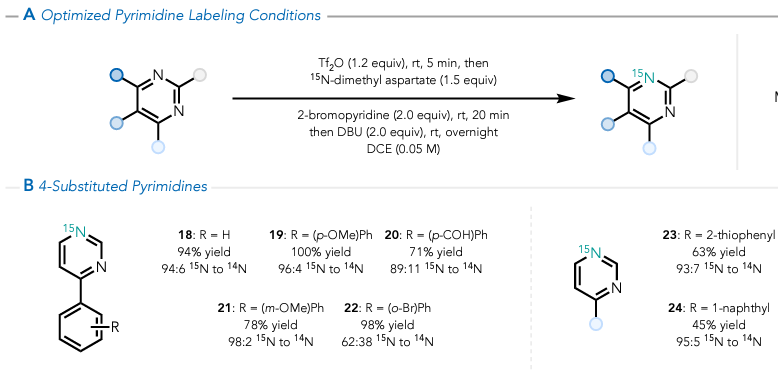
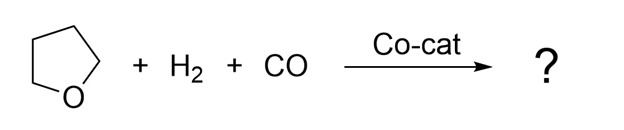Another approach to discovering new chemical reactions

OdanChem@OdanChem
Chemists are no longer pouring promising reactions down the drain just because the product is hard to isolate and understand the structure. They now use OdanChem to analyze the complex mixture directly from the flask and discover novel reactions. doi.org/10.26434/chemr…
English