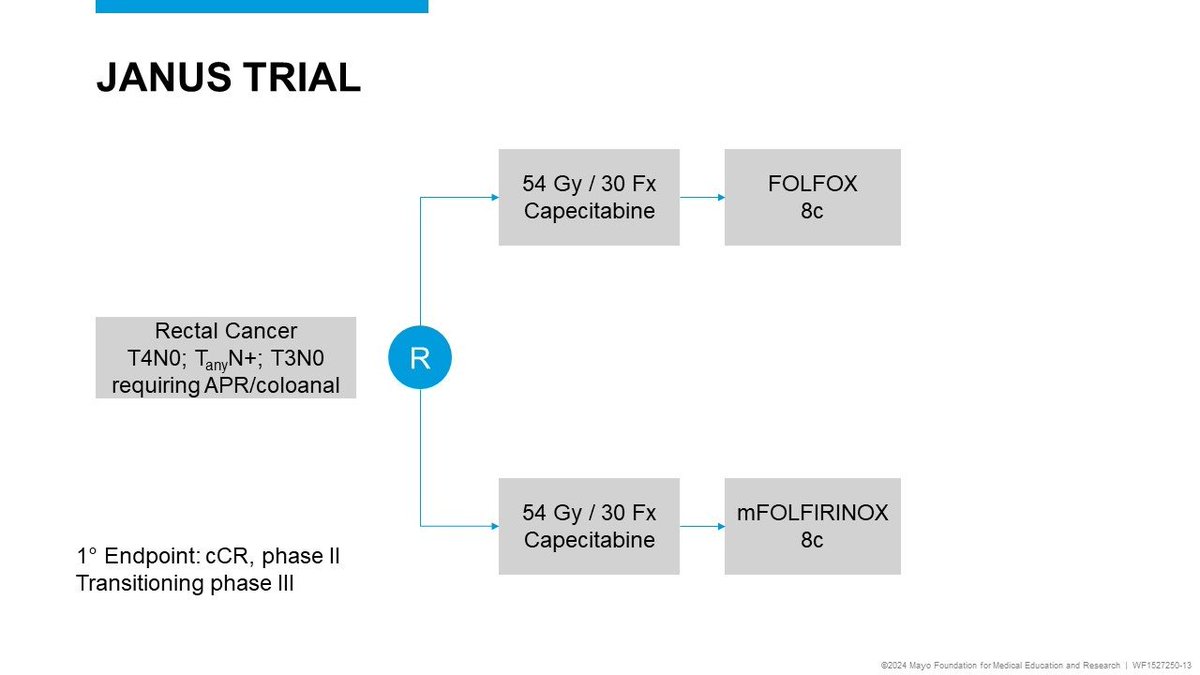
Daniel Catenacci
1.9K posts


Daniel Catenacci
@DocCatenacci
Medical Oncologist. "Targeted therapies for targeted populations”







#CM577 there is no benefit (especially CPS0) in CPS low/neg tumors. 44% of pts. And this is not even by histology which would make adenocarcinoma look even worse. Why is this buried in the supplement? & Why would you give this drug for a year to people having no benefit?





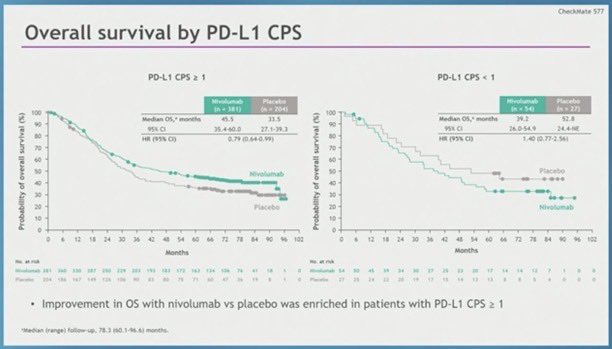


@pashtoonkasi @NEJM @OncoAlert @sitcancer @DocCatenacci @GIcancerDoc @agrothey @EileenMOReilly @AnwaarSaeed3 @marklewismd @ILSONDavid @HH_Oncodr @KlempnerSam 💡Some OncGI fellows have pointed: ➡️Supplementary Appendix: 📌 CPS PDL1 =/> 5 (371 pt): 29.4 ❌ 10.2 m 📌 CPS PDL1 <5 ( 295 pt) : 16.3 ❌ 11.1 m



@LizzySmyth1 HR 0.78 NS per the presentation but not in the paper (rather HR 0.99 <12m HR 0.67) >12m) Where have we seen this tuning fork curve before? ~1/3 pts no diff. CM577 I think it’s a good idea to see the results by PDL1 1, 5, 10: will likely show same thing as all previous studies




Presented at #ASCO25: In resectable gastric and gastroesophageal junction cancer, adding durvalumab to perioperative chemotherapy improved event-free survival and pathological complete response, with no major increase in high-grade adverse events. Full MATTERHORN trial results: nej.md/437IXTF



Better late than never, I guess.

📢 FDA ODAC gets it ✅️ for patients Immune checkpoint inhibitors are a PD-L1 targeted 🎯 therapy in GEA. No target, no efficacy. Let's focus on: ➡️ alternative targets (CLD 18.2, FGFR2b, MTAP...) ➡️ overcoming immune evasion (bispecifics, T-cell engagers, CAR-T..)






Time to move away from radiotherapy in operable GEA #ESMO24 TOPGEAR ➡️ no survival benefit when RT added to periop chemo TOPGEAR + ESOPEC : both great trials with a definitive answer = no more RT ✅Great drugs in development - let's focus on systemic Rx for cure @myESMO