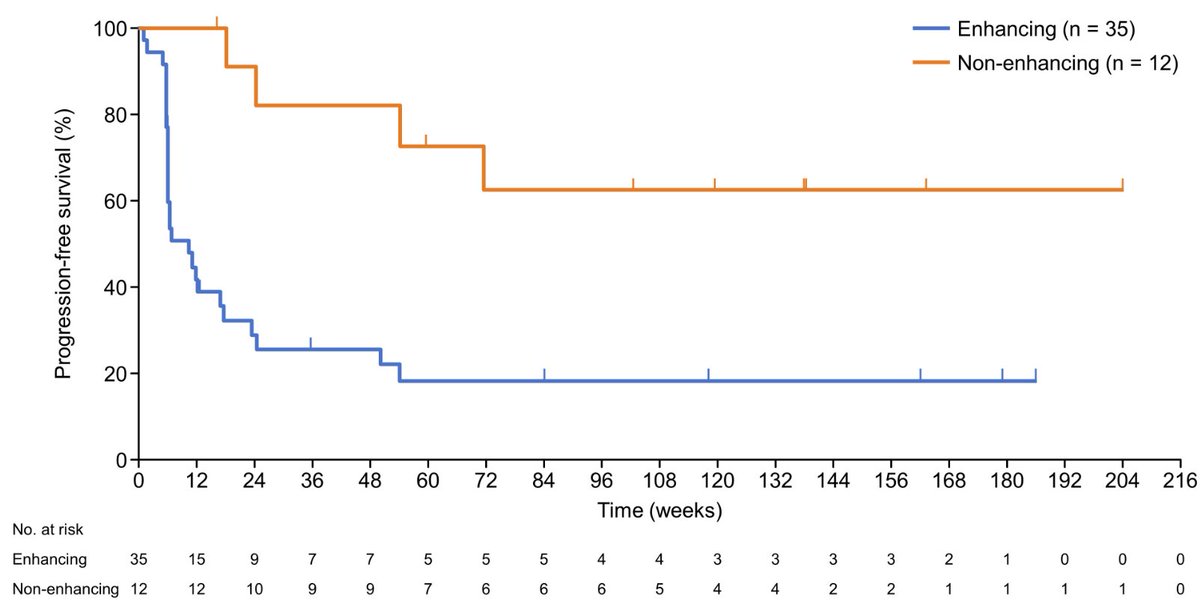
@augurbio Imo the messaging has been while skin fxn can be used, its not strong enough to stand on its own, ie clear efficacy needs to be there. 1yr 10 patients who cant even deteriorate on upright stability, the only metric where there is no placebo effect isn't all that compelling.
English