
Lenin K. Verdhi
56 posts


Lenin K. Verdhi
@LVerdhi
Postdoc @LCSA,@EPFL, Switzerland. Ph.D. @AMSzpilman @Arieluniversity, Israel. Research in Asymmetric Catalysis.



SAXITOXIN simplified: Appearing today in @ChemRxiv is a short, highly scalable, convergent approach to this entire natural product family using a tactical combination of radical retrosynthesis, biocatalysis (in collaboration with Merck), and C–H functionalization logic: chemrxiv.org/engage/chemrxi…


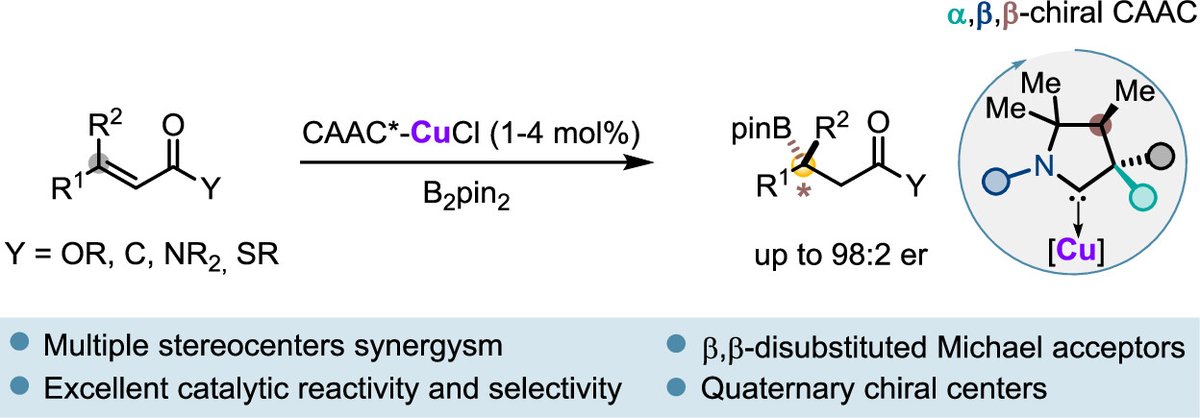

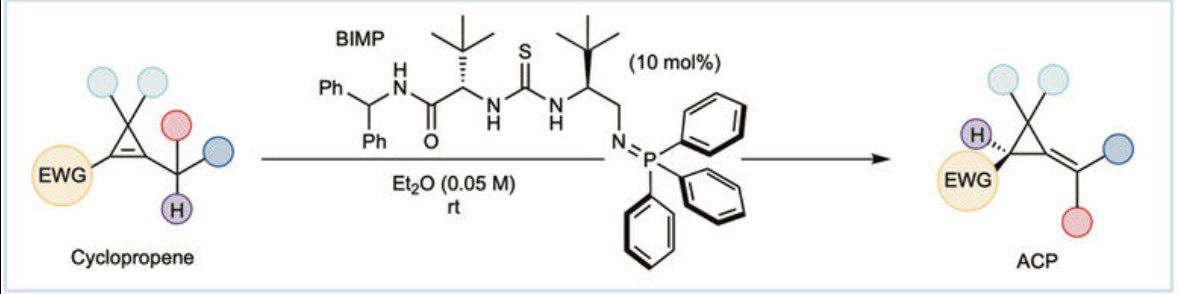




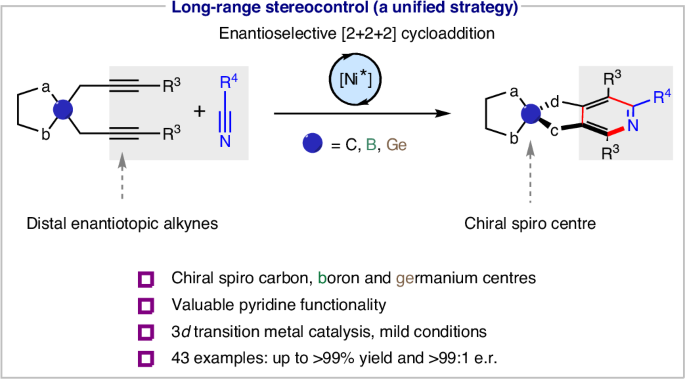




Just out @J_A_C_S Check out our latest work on enantioselective cobalt(III)-catalyzed [4 + 1] annulation of benzamides with cyclopropenes to access chiral isoindolinones. @LCSA @EPFL_CHEM_Tweet @NCCR_Catalysis | pubs.acs.org/doi/10.1021/ja…


Finally, you can do Umpolung α-Functionalization of Esters, Imides, AND Ketones via Iodine (III) enolonium species: Out now in Cell Reports Physical Science (Open Access) cell.com/cell-reports-p… Congrats @Rajakravi2012 @el75787382 Raz @AsitGhosh_Org @Subrata09894140 & Natalia






