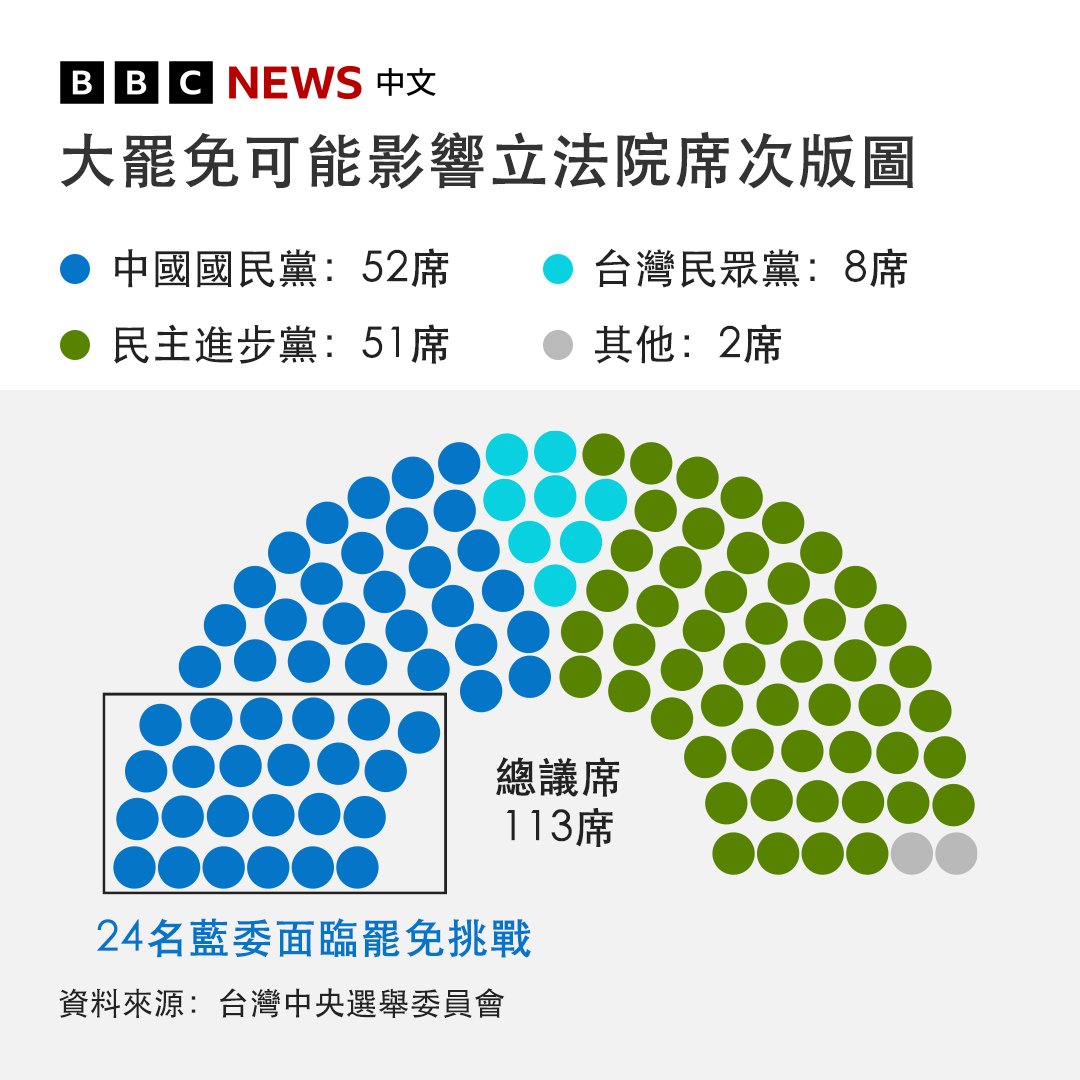
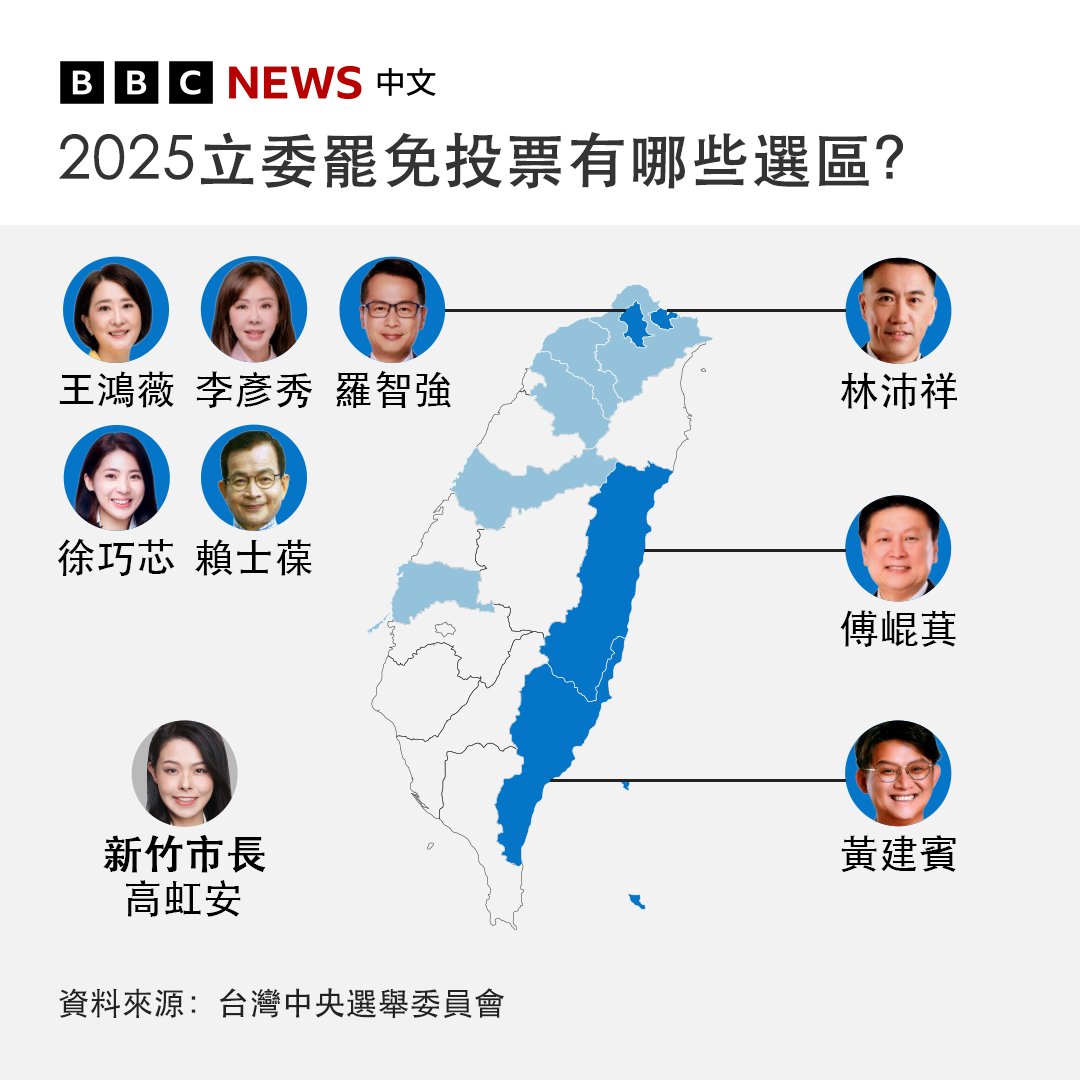
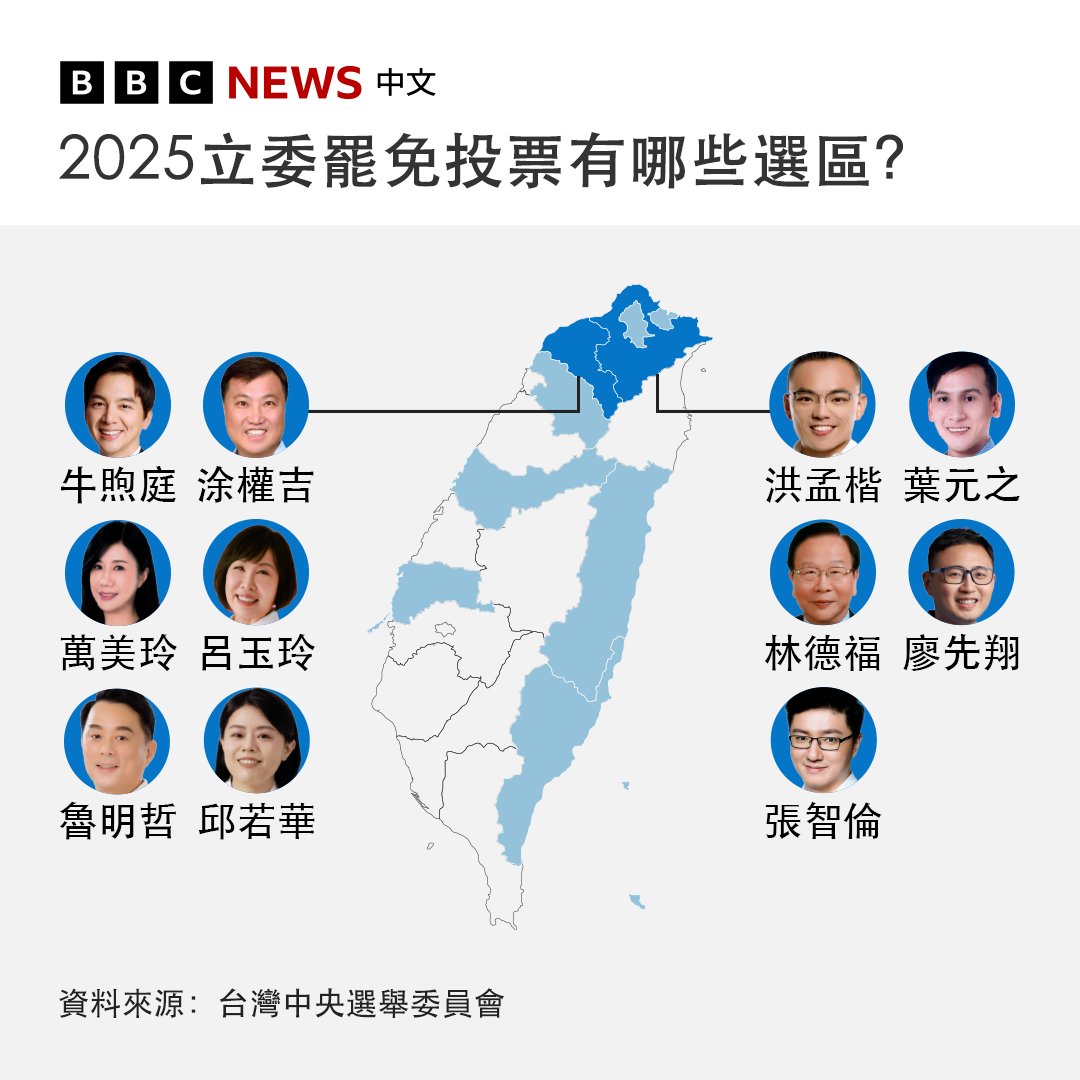
青天耀白日 retweetledi

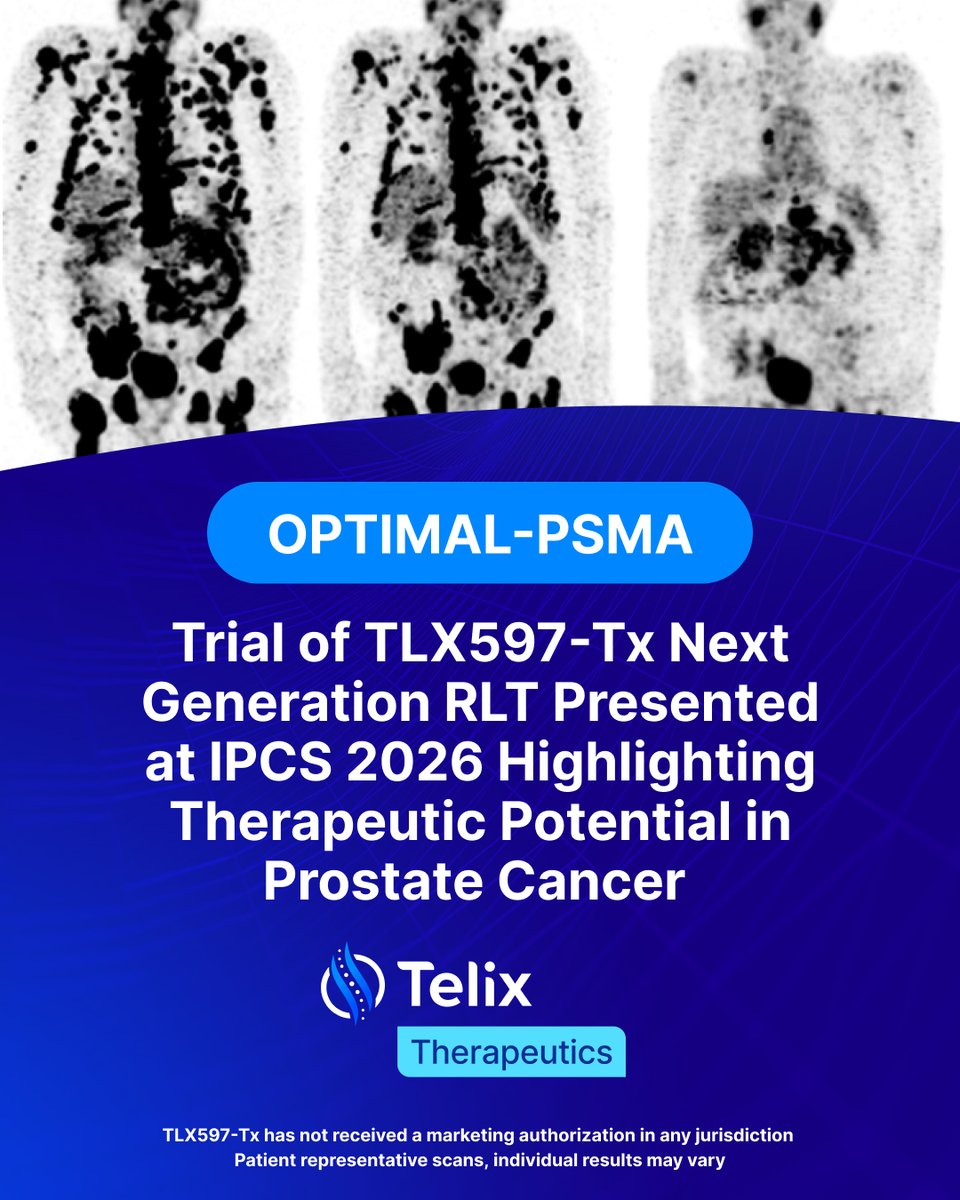
Telix announces encouraging dosimetry results from the Phase 2 OPTIMAL-PSMA trial of TLX597-Tx, presented at IPCS 2026 by Prof. Louise Emmett from St Vincent’s Hospital in Sydney, Australia.
TLX597-Tx is a novel PSMA-targeting small molecule radioligand therapy candidate, designed to improve quality-of-life and efficacy in earlier-stage prostate cancer.
Initial dosimetry, from the OPTIMAL-PSMA investigator-initiated trial (IIT) in metastatic castration-resistant prostate cancer (mCRPC), showed minimal salivary gland and kidney uptake, supporting dose intensification.
Telix believes these data support further evaluation of TLX597-Tx in earlier-stage disease and is initiating OPTIMAL-E, a Phase 2 study in metastatic hormone-sensitive prostate cancer (mHSPC).
TLX597-Tx is being developed alongside TLX591-Tx as part of a prostate therapeutic portfolio designed to address distinct disease stages and patient conditions — TLX591-Tx is in Phase 3 development in combination with standard of care for mCRPC, and TLX597-Tx intended for earlier-stage mHSPC.
Join today’s educational webcast to hear more about this multi-product approach to PSMA-targeted radionuclide therapy.
Register for the webcast here: s1.c-conf.com/diamondpass/10…
And read more about the results here: telixpharma.com/news-views/opt…
TLX597-Tx has not received a marketing authorization in any jurisdiction.
English