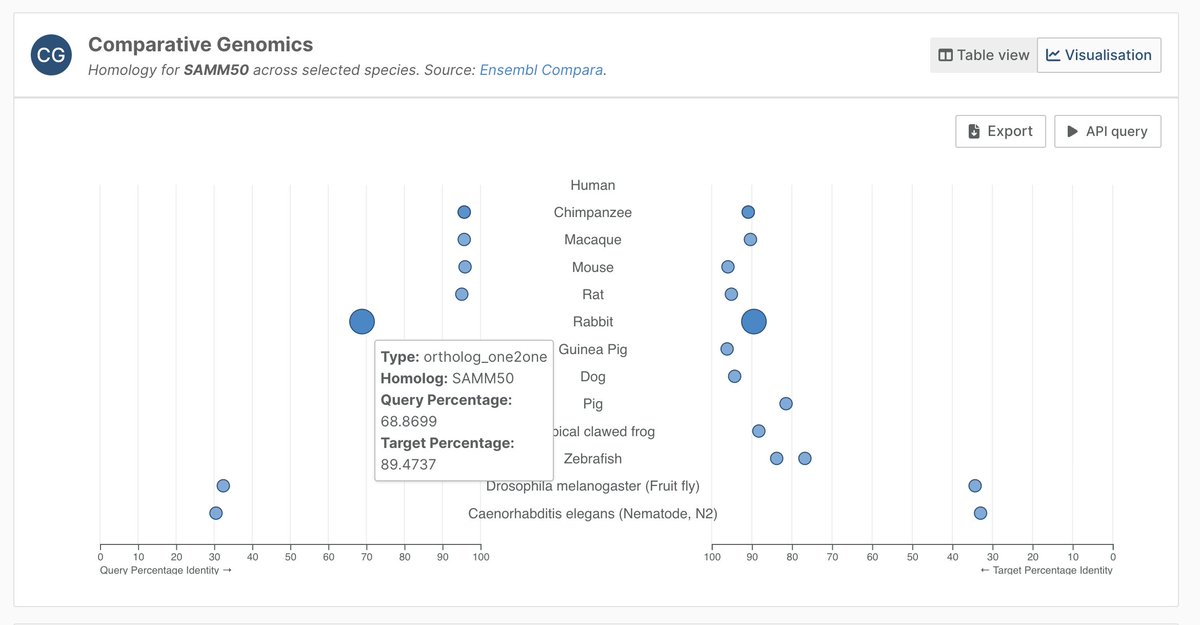
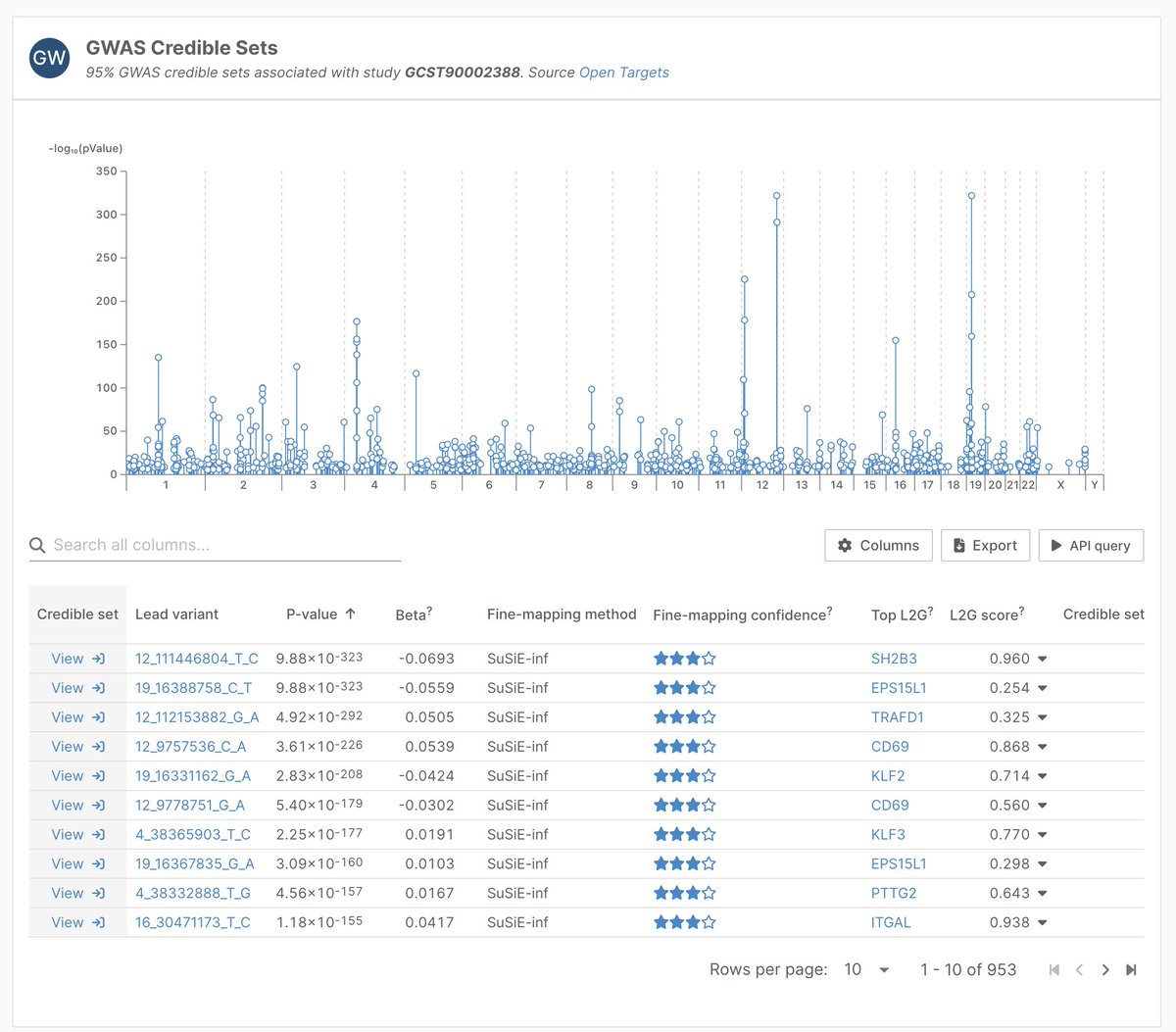
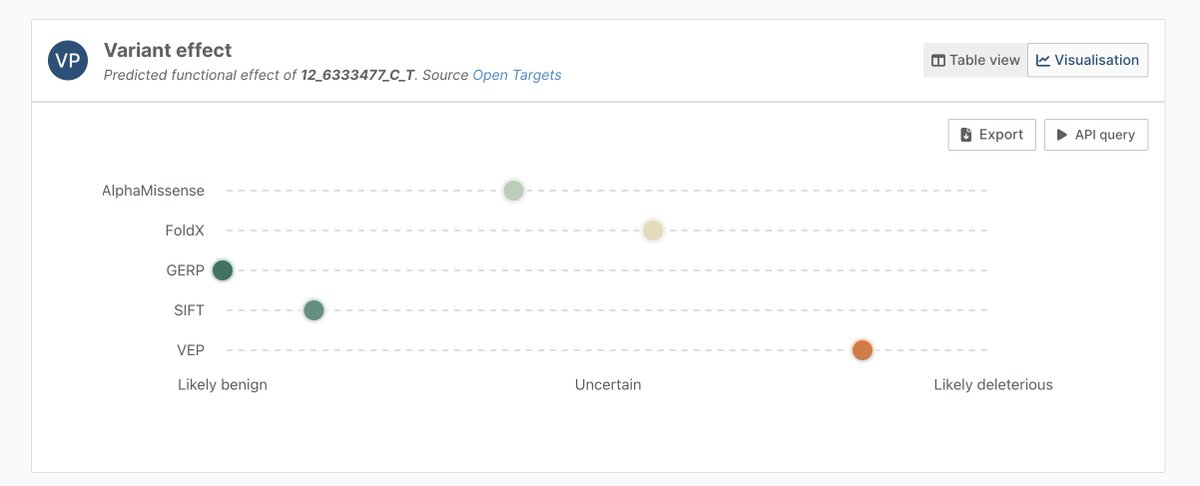
Open Targets retweetledi

The @OpenTargets Platform spring 🌼 release is out — and this one marks the beginning of something we've been building towards for a while.

English
Open Targets
2.7K posts


@OpenTargets
Public-private partnership using human genetics and genomics data for systematic drug target identification and prioritisation We're on Bluesky @opentargets.org