

Pierre Blanchard, MD
4K posts


@PBlanchardMD
Full Prof Radiation Oncology @gustaveroussy @UnivParisSaclay. EiC https://t.co/EihrJjVxi0. Director @SciencesCancer. Adjunct Prof @MDAndersonNews.





Colleagues at #GU26 ,A reminder that #APCCC26 🇨🇭is coming in 2026 This is a Conference is OPEN TO EVERYONE, not just the Panel members and EARLY REGISTRATION is up until February 28, 2026 👉️ REGISTER HERE: buff.ly/XpCWPdR Only 🔟 days left until the Early Bird deadline (28 February 2026) Enjoy 20% off when registering for both APCCC & IPCS 2026 Awarm invite to the Advanced Prostate Cancer Consensus Conference #APCCC26 in Lugano🇨🇭 Event Details: 📍 Palazzo Congressi, Lugano, Switzerland 🎟️ Register here 🎓 Credits requests: SGU – SSU, SSMO, SRO, SGMN In an era of rapid innovation and expanding treatment options for advanced #ProstateCancer , APCCC provides a vital forum for clinicians and researchers to address complex clinical questions where high-level evidence is limited. Over two days, the world’s leading experts will engage in high-level presentations, debates, and discussions to clarify and conceptualize the current landscape of care, with a focus on areas of controversy or uncertainty. The insights and consensus developed during the meeting are later published in European Urology, helping shape global standards. Creating new opportunities for younger investigators & the next generation of leaders in advanced prostate cancer! @Silke_Gillessen & @AOmlin @fabioturco92 @Prof_IanD @drjefstathiou @Prof_Nick_James @ChrisSweens1 @BertrandTOMBAL @ZilliThomas @neerajaiims @bjartell @PBlanchardMD @BourlonMaite @Albert0Briganti @cdanicas @Ecastromarcos @mdesantis234 @nachoduranm @drlouiseemmett @stefanofanti4 @fontev1 @Nicola_Fossati @marty_gleave @ProfKHerrmann @DrMHofman @BarbaraJereczek @ravikanesvaran @FacsRaja @LoebStacy @quimmateo @DrRanaMcKay @amerseburger @CaPsurvivorship @declangmurphy @DrPaulNguyen @WilliamOhMD @piet_ost @DrSpratticus @cnsternberg @DrYukselUrun Ping @nataliagandur @OncoAlert 🚨



ARTO Trial: Long-term overall survival analysis from a randomized phase II trial testing the benefit of SBRT addition to abiraterone acetate in oligometastatic #CRPC patients. Presentation by @GiulioFrancoli1 @AOUCareggi. #GU26 written coverage by @RKSayyid @UAUrology > bit.ly/4b2BKa8 @ASCO

#ASCOGU26 ARTO trial (Ph2, n=157; f/u: 53 mo): Abiraterone +/- SBRT in 1L oligometastatic CRPC Benefit with SBRT: 🔷 bPFS: HR 0.49 (p<0.001) 🔷 rPFS: HR 0.48 (p<0.001) 🔷 OS: HR 0.55 🔷 PCSS: HR 0.37 (p=0.006) 📌63% relative reduction in prostate cancer–specific death with metastasis-directed SBRT. @urotoday


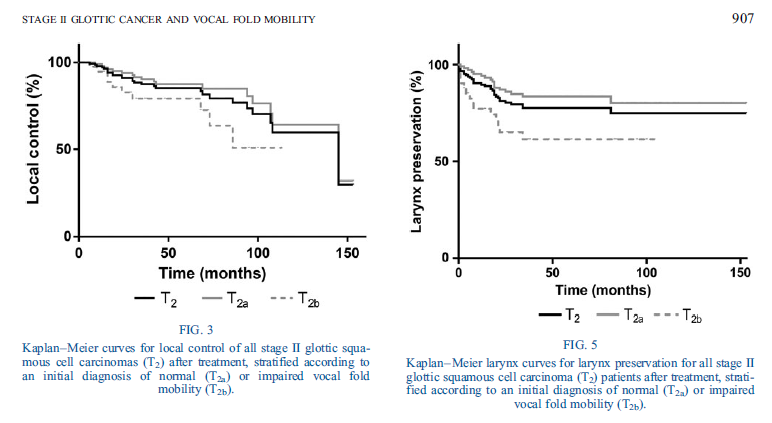

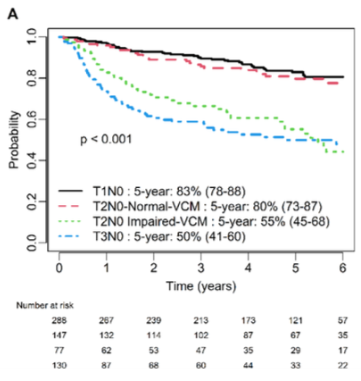
🔥 Hot off the press: "Prognostic Value of Impaired Vocal Cord Mobility in T2N0 Glottic Cancer Treated With IMRT" ➡️In T2N0 glottic cancer, impaired vocal cord mobility is associated with worse LRF (aHR 3.7) and DFS (aHR 2.7) doi.org/10.1002/lary.7… @PMResearch_UHN @UofTDRO




📣Updated ESMO #ClinicalPracticeGuideline on advanced and metastatic #ProstateCancer: ✅ Substantial advances in castration-sensitive & -resistant prostate cancer ✅ Updated algorithms & recommendations reflecting latest clinical data #ESMOGuidelines 🔗 ow.ly/32vY50Y9T99

Excited to share the final publication in The Lancet of our phase 3 GORTEC 2018-01 (NIVOPOST-OP) trial results. We hope these results will support changes in clinical practice and future guideline updates. @PBlanchardMD @DrMLChua @GorphePhilippe thelancet.com/journals/lance…



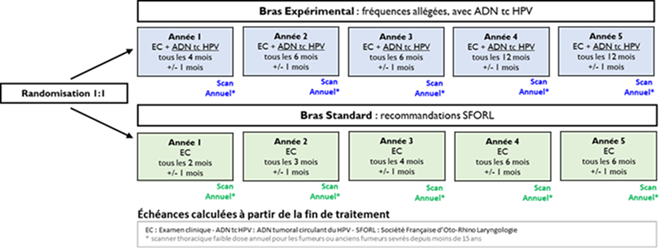
The linked paper is a nice document and summary product, but let's emphasize a few highlights: 1. Statement 16, strong consensus: ctHPVDNA should supplement conventional surveillance tools, rather than reducing or replacing them. Natural corollary: if it is not reducing or replacing surveillance, what is the point? 2. Statement 9, strong consensus: Detectable ctHPVDNA without clinical or radiographic evidence of disease often increases patient distress. 3. Statement 5, strong consensus: Large-scale, prospective randomized controlled trials are necessary to validate ctHPVDNA's role in HPV+ oropharyngeal cancer management. I would change that statement from validate to determine. Validate assumes it has a role today, but as the document states, it doesn't reduce or replace any currently established surveillance tools. There are so many exciting ways that ctHPVDNA may influence care (screening/diagnosis, de-escalation, non-imaging-based surveillance) but these strategies need to be established in prospective randomized trials.



@DavidSherMD @PBlanchardMD Link to the final publication : jamanetwork.com/journals/jaman…


📢 Welcome Aileen Duffton, PhD as the new Editor-in-Chief of tipsRO! ✨Aileen is Lead R&D Radiographer at BWoSCC (Glasgow) and Senior Honorary Lecturer at the University of Glasgow. 🙏We thank Michelle Leech for her outstanding leadership as former EiC! #Radiotherapy #RTT