Rasmus H
1.8K posts


Rasmus H
@RazBioTech
Founder @ https://t.co/rvCWR16DUM | 🧬 #biotech retail investor | 👀 $OCUL maxi | 🌎 Global citizen | 👋 always happy to connect










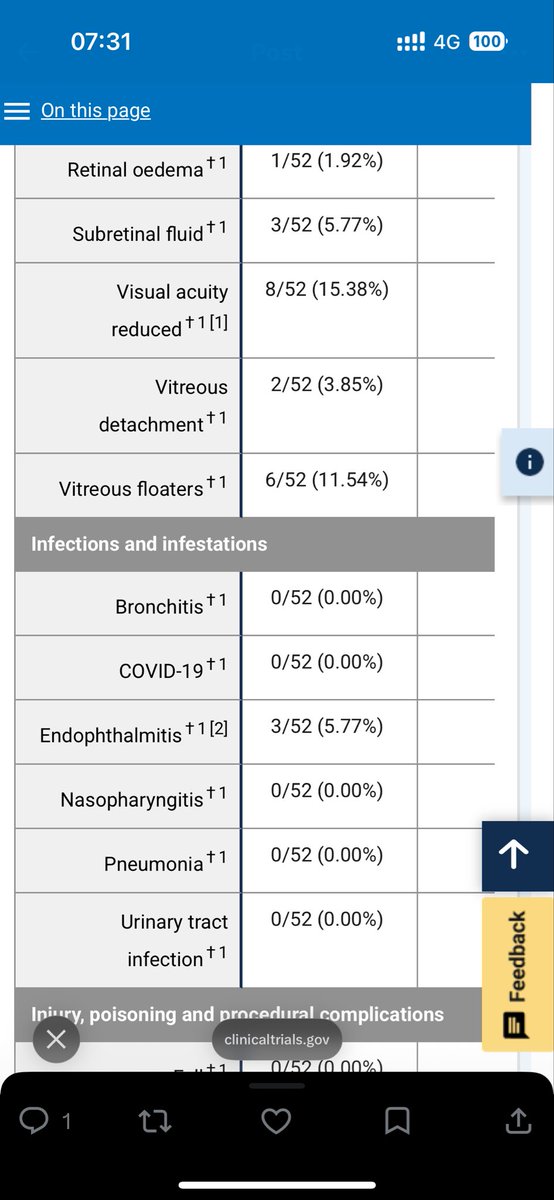
@wallstlobo Would love data on how control group in SOL-1 performed with SOL-R rescue criteria applied. Would love “tap-out-data”, i.e is the BCVA data muted in Ax arm because vision maxed out? Would love other -relevant- subgroup analysis. $OCUL













@JeremyFalmouth @Sjfms1Steven @Biotech_FC @sububoston @AscendingBio @InvestorLatin One big unknown is redosing, and especially if HD will perform as well after redose 1 as after the first dose? Here ax might have a slight advantage as SOL-1 suggests it’ll be 30%+ drier at wk 24. HD enters 2nd phase wetter & without 6 months of recent loading doses to help it.