ResearchPulse
4.7K posts






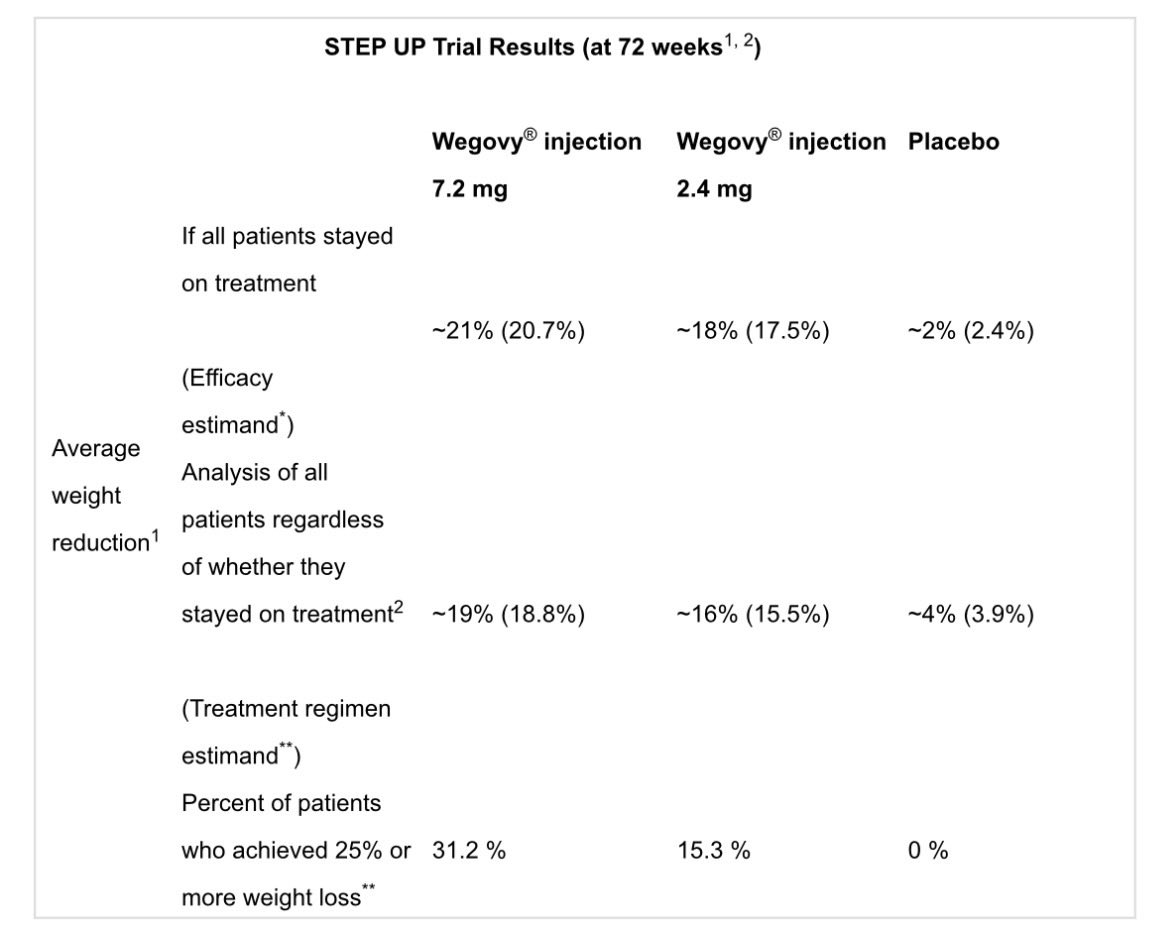
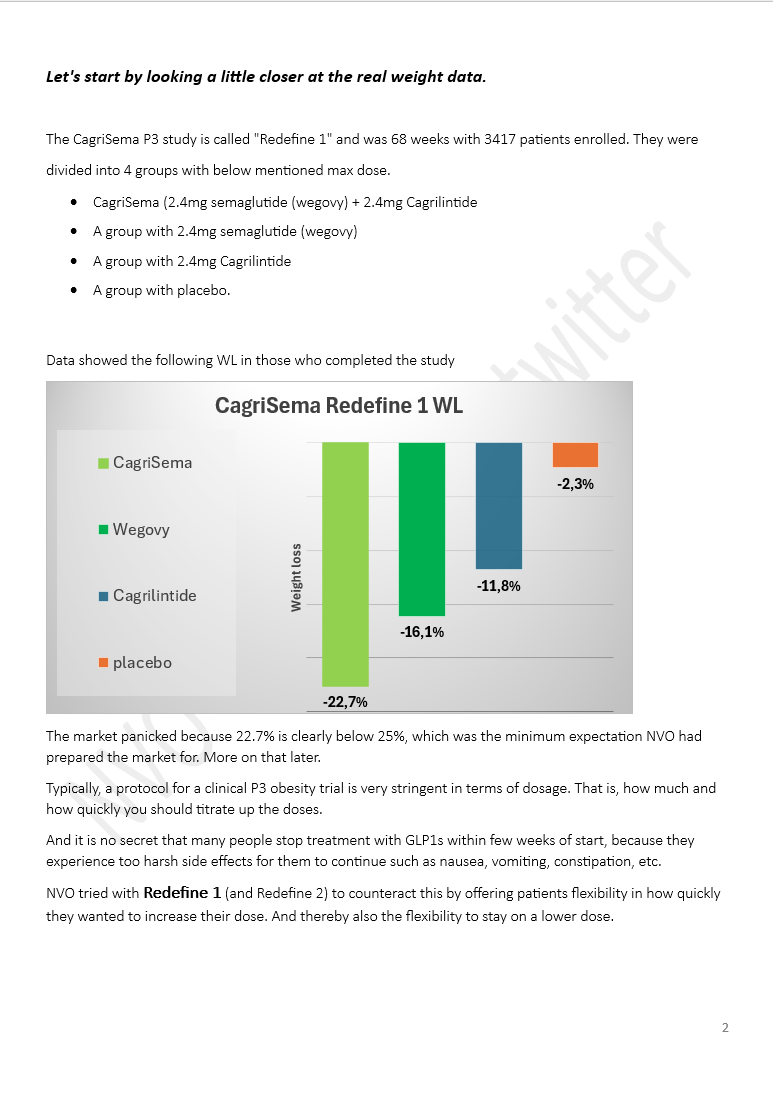
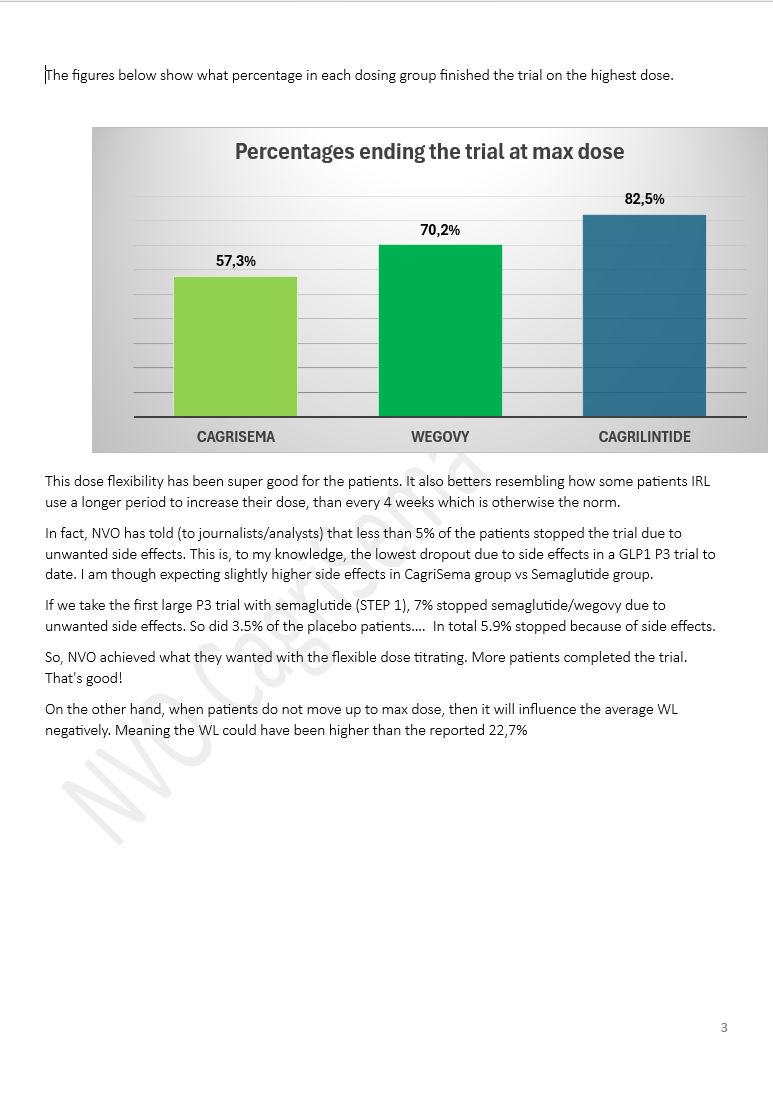
The Week of $NVO: Wegovy HD Approval, India Price War, Oral Competition and Rising Global Stakes Another packed week for Novo Nordisk. Here are the key developments. Wegovy HD raises the bar • FDA approves higher-dose Wegovy (7.2 mg) • ~20.7% weight loss in trials, close to $LLY’s Zepbound • Launch expected April 2026 Oral GLP-1 race intensifies • Structure Therapeutics reports ~16% weight loss with oral GLP-1 in phase 2 • Citi flags it as a potential challenger to Novo’s pill • $LLY ’s orforglipron shows stronger A1C and weight loss in diabetes India becomes a major disruptor • Semaglutide patents expire • 40+ generics entering, 50+ brands expected • Prices collapsing >50% Investor sentiment remains divided • Bernstein initiates “underperform”, PT DKK 175 (lowest on the Street) • HSBC cuts PT to DKK 280 (Hold) • CCB initiates Outperform Protean buys back Novo after selloff • Cites strong oral Wegovy launch - “The pill is magical.” • Highlights SNAC tech edge (patented into 2030s) • ~8% FCF yield at ~DKK 250 Competition keeps building • Lilly expands access with Employer Connect (~$449/month) • Retatrutide shows strong Phase 3 data • Roche says the market will segment, not winner-takes-all Regulatory and structural shifts • GLP-1 trials challenged as patients drop out (placebo issue) • Growing pressure to compare new drugs vs existing treatments instead of placebo • Sweden reimbursement rejected, Novo appeals Geopolitics and global expansion • Iran conflict could impact operations near Tehran • China signals support for Novo’s presence • Japan: Novo expanding self-pay access and national partnerships New data expands the story • Semaglutide linked to lower risk of depression and anxiety • Suggests GLP-1s may impact more than weight Distribution is evolving fast • $HIMS now offering real Wegovy and Ozempic from ~$149/month • Shift away from compounded GLP-1s Share buybacks continue • 6.58M shares repurchased since Feb • Value: DKK 1.78B #stocks #Investing