FollowTheFilings
2.1K posts

FollowTheFilings
@fdzmurillo
Investor and researcher. I dig into SEC filings, patent courts, and buyback programs to find what corporations don't want you to see. When actions contradict wo
LONDON Katılım Haziran 2009
1.6K Takip Edilen105 Takipçiler

The Week of $NVO: Wegovy HD Approval, India Price War, Oral Competition and Rising Global Stakes
Another packed week for Novo Nordisk. Here are the key developments.
Wegovy HD raises the bar
• FDA approves higher-dose Wegovy (7.2 mg)
• ~20.7% weight loss in trials, close to $LLY’s Zepbound
• Launch expected April 2026
Oral GLP-1 race intensifies
• Structure Therapeutics reports ~16% weight loss with oral GLP-1 in phase 2
• Citi flags it as a potential challenger to Novo’s pill
• $LLY ’s orforglipron shows stronger A1C and weight loss in diabetes
India becomes a major disruptor
• Semaglutide patents expire
• 40+ generics entering, 50+ brands expected
• Prices collapsing >50%
Investor sentiment remains divided
• Bernstein initiates “underperform”, PT DKK 175 (lowest on the Street)
• HSBC cuts PT to DKK 280 (Hold)
• CCB initiates Outperform
Protean buys back Novo after selloff
• Cites strong oral Wegovy launch - “The pill is magical.”
• Highlights SNAC tech edge (patented into 2030s)
• ~8% FCF yield at ~DKK 250
Competition keeps building
• Lilly expands access with Employer Connect (~$449/month)
• Retatrutide shows strong Phase 3 data
• Roche says the market will segment, not winner-takes-all
Regulatory and structural shifts
• GLP-1 trials challenged as patients drop out (placebo issue)
• Growing pressure to compare new drugs vs existing treatments instead of placebo
• Sweden reimbursement rejected, Novo appeals
Geopolitics and global expansion
• Iran conflict could impact operations near Tehran
• China signals support for Novo’s presence
• Japan: Novo expanding self-pay access and national partnerships
New data expands the story
• Semaglutide linked to lower risk of depression and anxiety
• Suggests GLP-1s may impact more than weight
Distribution is evolving fast
• $HIMS now offering real Wegovy and Ozempic from ~$149/month
• Shift away from compounded GLP-1s
Share buybacks continue
• 6.58M shares repurchased since Feb
• Value: DKK 1.78B
#stocks #Investing
English

The heterogeneity across cancer types is the key finding. Semaglutide specifically showing the strongest signal across multiple indications (obesity, CV, depression, and now oncology data) suggests molecule-specific effects beyond the GLP-1 class.
What makes oral delivery particularly relevant here: if long-term daily exposure drives the protective effect, then a convenient oral formulation with high adherence may outperform intermittent injectable use in real-world oncology prevention.
The intersection of metabolic phenotype, drug exposure duration, and delivery route deserves dedicated study.
$NVO #GLP1
English

Add kidney protection to the $NVO list.
New systematic review (Diabetes Therapy, March 2026): GLP-1 agonists and tirzepatide show renal benefits across CKD stages in patients with diabetes and obesity.
The indication count for semaglutide:
• Obesity ✔️ Approved
• Diabetes ✔️ Approved
• Cardiovascular ✔️ Approved
• MASH/liver ✔️ Approved
• Depression — 44% risk reduction
• Anxiety — 38% risk reduction
• Anti-aging — under investigation
• Cancer risk — reduced across 5 types
• Kidney disease — renal benefits confirmed
9 indications. One molecule. One daily pill. No oral rival.
Every new indication multiplies the value of SNAC — the only patented way to deliver this orally. Protected into late 2030s.
850 million people worldwide have chronic kidney disease.
The market prices $NVO at 14x for a "weight loss company losing patents."
The science says it's a multi-organ therapeutic platform with an oral monopoly.
$NVO $LLY #GLP1 #SNAC #CKD
English

I am pleased to share our recent publication in Diabetes Therapy on Renal Outcomes of GLP‑1 Receptor Agonists and Tirzepatide Across CKD Stages and Metabolic Phenotypes (Type 2 Diabetes and/or Overweight/
Obesity): A Scoping Review. Link: link.springer.com/content/pdf/10… @SLANH_ @visanpolo @SLANHJoven @asocolnefmedios @endocatracha

English

Great recap. Two things missing that change the picture.
1. India is NOT disrupting oral.
40+ generics entering? Yes — for injectables. The compound patent expired. That's priced in.
But oral semaglutide needs SNAC at 0.6-2.1 mmol. Delhi High Court (March 20) forced Torrent Pharma to stay at 0.53 mmol — below effective range. $NVO fought three simultaneous cases in India. Lost semaglutide. Won SNAC.
They didn't fight for the molecule. They fought for the delivery system. That tells you where the value is.
India disrupts injections. SNAC protects the pill. Into the late 2030s.
2. The buyback pattern changed.
You note 6.58M shares repurchased. But the critical context: SEC 6-K filings from 2023-2024 show Novo Holdings sold B shares proportionally during every buyback to "maintain ownership around 28%."
In 2025, they stopped selling. In 2026, the company buys 250K/day at historic lows. No evidence Novo Holdings is selling.
Every cancelled share raises the Foundation's 28.1% for free. Under Danish law, 90% = squeeze-out and delisting.
The $220B Foundation controls 77% of votes. The pill is a lifetime subscription drug — stop taking it, weight returns. SNAC patent into late 2030s. No oral rival.
Would you share this with public markets? Or take it private?
$NVO $LLY #GLP1 #SNAC
English

Great question. Follow the SEC filings.
In 2023 and 2024, Novo Holdings sold B shares to $NVO proportionally during every buyback. They filed 6-K forms stating they intended to "maintain ownership around 28%."
In 2025, Novo Holdings stopped selling. Their own annual results confirm: income was "negatively affected by the lack of proceeds from a share buyback."
In 2026, the company buys 250K shares/day at DKK 250. No evidence Novo Holdings is selling this time.
If they don't sell while shares are cancelled, their 28.1% rises for free.
Who benefits? The foundation that controls 77% of votes and sits on $220 billion.
$NVO
English

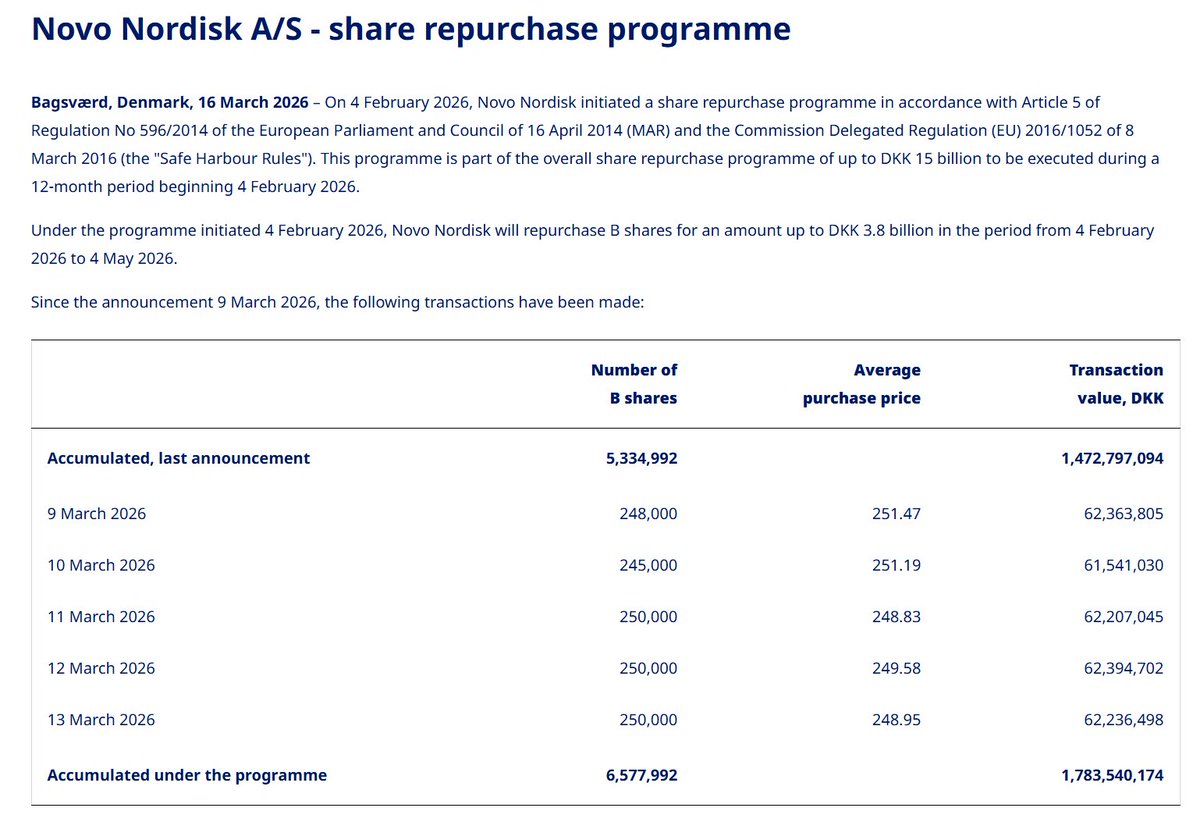
The receipts. $NVO share repurchase programme, straight from the filing.
250,000 shares bought. Every single day. At ~DKK 250.
6.6 million shares. DKK 1.78 billion spent. In 6 weeks.
This is tranche 1 of a DKK 15 billion ($2.1B) programme.
At these prices, every DKK 15 billion buys roughly 60 million shares — over 1.3% of the company. Cancelled shares raise the Foundation's 28.1% ownership automatically.
In 2023 and 2024, Novo Holdings sold B shares proportionally during every buyback to stay at 28%. SEC filings confirm this.
In 2025, they stopped selling.
In 2026, the company buys 250K shares per day at historic lows. And silence from Novo Holdings.
The guidance said -13%. The buyback says otherwise.
Follow the filings.
$NVO #GLP1 #SNAC
English

Good research. Let me add the key nuance.
The base SNAC compound patents have expired. But that's irrelevant. Knowing SNAC exists and being able to use it effectively in an oral GLP-1 are two very different things.
What matters is the co-formulation patents — the specific SNAC concentration range (0.6-2.1 mmol) optimized for semaglutide absorption. Those are active into the late 2030s.
Delhi High Court just proved why this matters: Torrent Pharma can make oral semaglutide with SNAC, but only at 0.53 mmol — below the effective range. Lower SNAC = lower absorption = inferior product. They'd need their own clinical trials to prove efficacy at that dose.
Re: @Doctor_Salomon's question about $VKTX — Viking's oral VK2735 is a GLP-1/GIP dual agonist. Their oral delivery mechanism isn't fully disclosed, but their Phase 2 data showed a 12.2% dropout rate and only 13 weeks of data. Compare that to $NVO's Wegovy pill: 16.6% weight loss at 64 weeks, 600K Rx in 10 weeks, FDA approved.
The base molecule can go generic. The optimized delivery cannot. That's the moat.
$NVO #SNAC #GLP1
English

I asked grok about the current state of Novo’s SNAC related patents in the US. It’s interesting to note that the initial SNAC patents have expired, with several more expiring this year.
That said, patents related to specific SNAC formulations and enhancers, as well as certain Semaglutide + SNAC patents are still active and live into the late 2030s.
grok.com/share/c2hhcmQt…
Investseekers@investseekers
Update on the $NVO vs Torrent Pharma case in India: The Delhi High Court records an interim arrangement: • Torrent can continue selling oral semaglutide (Semalix, Sembolic) • But must stay outside Novo’s patented SNAC range (0.6–2.1 mmol) • Torrent’s product uses ~0.53 mmol, below the patented threshold Torrent will also keep sales records and provide samples. Link to article: livelawbiz.com/patent/delhi-h… #stocks #investing
English

Every week the $NVO thesis gets bigger.
Nature Reviews (2026): GLP-1 receptor agonists associated with reduced risk of liver, oesophageal, endometrial, ovarian, and prostate cancer.
The indication list for semaglutide:
• Obesity ✔️ FDA approved
• Diabetes ✔️ FDA approved
• Cardiovascular ✔️ FDA approved
• MASH/liver disease ✔️ FDA approved
• Depression — 44% risk reduction (Lancet data)
• Anxiety — 38% risk reduction
• Anti-aging — under investigation (Einstein College)
• Cancer risk — reduced across 5 types (Nature)
One molecule. One daily pill. No oral competitor.
Protected by SNAC until 2032. A patent that covers the only way to deliver this orally at therapeutic levels.
The market prices $NVO as a weight loss company at 14x.
It's becoming a daily health platform with 8+ indications, recurring use, and an oral monopoly.
$NVO $LLY #GLP1 #SNAC #oncology
English

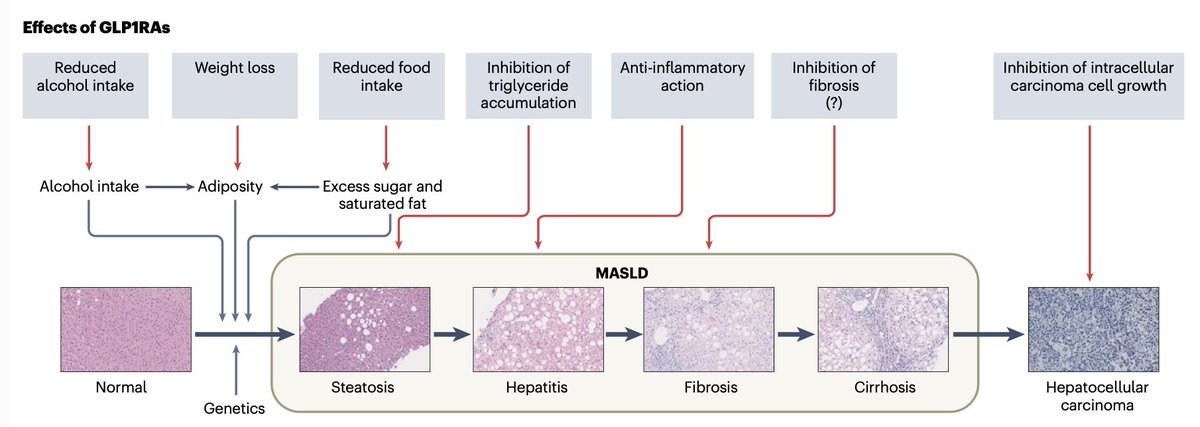
🚨 GLP-1RAs and cancer risk.
GLP-1RAs in T2DM/obesity show heterogeneous associations with cancer: ↓ hepatocellular, oesophageal, endometrial, ovarian, prostate; concern persists for thyroid; no confirmed pancreatic signal.
Biases and limited RCT follow-up remain key limitations. Net benefit favourable in high cardiometabolic risk. Role in oncology remains investigational.
📖 @NatRevClinOncol
DOI 👉🏻 doi.org/10.1038/s41571…
#CánCare #oncology #cancer #GLP1 #obesity #diabetes



English

The $NVO indication universe keeps expanding.
New data: semaglutide reduces risk of worsening depression by 44% and anxiety by 38% in patients with obesity/diabetes. Strongest effect of any GLP-1 in the dataset.
So the Wegovy pill now touches:
• Obesity (100M Americans)
• Cardiovascular risk (FDA-approved indication)
• MASH/liver disease (FDA-approved)
• Depression (280M people globally)
• Anxiety (300M globally)
• Potential anti-aging (Einstein College research)
A daily pill. You take it forever. No oral competitor. SNAC patent until 2032.
And semaglutide specifically outperforms every other GLP-1 on mental health — which means $LLY tirzepatide and orforglipron can't assume equivalent benefit.
The market is pricing $NVO at 14x for a weight loss story. The reality is a multi-indication oral platform with recurring revenue and no rival.
Follow the filings. Follow the science.
$NVO $LLY #GLP1 #SNAC #mentalhealth
English

GLP-1 as an anti-aging drug.
Albert Einstein College of Medicine, Future Cardiology (2025): investigating GLP-1 receptor agonists as tools against aging, frailty, and age-related disease.
Obesity was the market. Aging is the universe.
And $NVO owns the only oral delivery platform (SNAC) that can put this in a daily pill. Patented until 2032. No competitor.
A daily pill you take forever. For weight. For your heart. Maybe for aging.
At 14x earnings.
$NVO #GLP1 #SNAC #longevity
English

GLP-1 receptor agonists and SGLT2 inhibitors: new anti-aging tools?
tandfonline.com/doi/full/10.10…

English

Think about what $NVO actually owns.
A pill for obesity. FDA approved. No competitor. 600K prescriptions in 10 weeks.
But here's the part nobody says out loud:
If you stop taking it, the weight comes back.
This isn't a cure. It's a subscription. Recurring revenue. Monthly. For life.
Viagra changed pharma. This is bigger. Obesity affects 100M+ Americans. Every one of them is a potential lifetime subscriber.
And the technology that makes the pill work — SNAC — is patented until 2032. Six years of legal monopoly on the oral delivery platform. No generic can replicate it. Delhi High Court confirmed it 2 days ago.
Approved worldwide. No alternative. Recurring use. Patent-protected until 2032.
This is the Holy Grail of pharma. And it trades at 14x earnings.
$NVO #GLP1 #SNAC
English

Exactly. This is the part the market hasn't priced in.
Delhi High Court (March 20) confirmed it: Torrent Pharma forced to use 0.53 mmol SNAC — below $NVO's patented 0.6-2.1 mmol range.
Lower SNAC = lower absorption = lower efficacy. It's a different product. Would need its own trials, its own approval, its own label.
So the "semaglutide going generic" narrative is really:
• Injectable generic? Yes.
• Oral generic with equivalent efficacy? No. Legally impossible without infringing SNAC.
That's the moat nobody is discussing. Semaglutide expires. SNAC doesn't.
$NVO #GLP1 #SNAC
English

Yes it will be interesting to see how they will market their oral semaglutide with half the amount of SNAC vs $NVO 1. Generation.
Bioavailability will be lower than NVO 1G 3/7/14mg Rybelsus = less effect
So its should be a new product that needs to go through ordinary clinical trials to be approved.
And there’s no prices for their oral semaglutide. Only their injection
English

Why no one can copy the pill:
SNAC — the patented absorption technology that makes oral semaglutide work.
Yesterday (March 20), Delhi High Court confirmed it: Indian generic Torrent Pharma forced to reformulate BELOW the effective SNAC range.
If you can't copy it in India, you can't copy it anywhere.
$NVO
English

Important context missing here.
ACHIEVE-3 compared orforglipron vs oral semaglutide at 7mg and 14mg — the old Rybelsus doses for T2D.
It did NOT compare against the 25mg Wegovy pill, which is the actual obesity product driving 600K+ prescriptions in 10 weeks.
The obesity comparison that matters:
• $NVO Wegovy pill 25mg: 16.6% weight loss
• $LLY orforglipron 36mg: 11.2% weight loss
48% efficacy gap. Different trial, different dose, different result.
On convenience: yes, no fasting is a real advantage. But patients and doctors choose outcomes over convenience. You don't switch from 16.6% to 11.2% to skip 30 minutes of fasting.
And your amycretin mention in tweet 4 is the key nobody connects: amycretin is $NVO's next-gen dual agonist. Oral delivery? Via SNAC — the same patented platform. Every future Novo peptide gets an oral version. Orforglipron is one molecule with no platform behind it.
SNAC is the moat. Not semaglutide.
$NVO $LLY #GLP1 #SNAC
English

1/ ACHIEVE published. First head-to-head Phase 3 trial: oral orforglipron vs oral semaglutide in T2D (ACHIEVE-3) . A non-peptide GLP-1 RA that doesn't need SNAC — no fasting, no water restrictions. This could reshape oral GLP-1 access worldwide. doi.org/10.1016/S0140-…
English

This is the data point people are missing.
NRx +4.5%. TRx +6.7%.
TRx growing faster than NRx means refills are accelerating. Doctors are writing multi-month scripts. Patients are staying on the pill.
Week 10. No oral competitor. Retention building.
And $NVO guided -13% for 2026 while this was happening.
At some point the prescription data and the guidance will collide. Q1 earnings is that moment.
$NVO #GLP1 #Wegovy
English


Parity with tirzepatide. That's the headline.
But here's what matters more: $NVO now has best injection AND best pill. No one else has both.
Wegovy HD: 20.7% — matches Zepbound.
Wegovy pill: 16.6% — crushes orforglipron's 11.2%.
On pricing: at $349 they're already undercutting Zepbound while offering an oral option $LLY can't match. The pill at $149-$299 opens an entirely new patient population that won't inject.
And the pill is protected by SNAC — a patent no generic can touch. Delhi HC confirmed it 2 days ago.
The market is pricing $NVO as if it has nothing. It has everything.
$NVO #GLP1 #SNAC
English

$NVO strengthens its position in obesity with FDA approval of Wegovy HD.
A higher-dose version designed to push efficacy further, with ~20%+ weight loss in trials.
The strategy is clear:
• Extend the semaglutide franchise rather than wait for next-gen drugs
• Raise the efficacy bar to stay competitive with $LLY
• Leverage existing scale, manufacturing, and physician familiarity
This isn’t just a product update. It’s lifecycle optimization of a blockbuster.
Bigger picture:
• Higher efficacy could drive more prescriptions and reimbursement
• Obesity is increasingly treated as a chronic disease, not lifestyle
• Competition is shifting from access → performance
Link to article: business-news-today.com/novo-nordisk-s…
#stocksinfocus #investing
English

20.7% vs Zepbound's ~22%. Injectables are now at parity. That narrative is over.
But the real question isn't injectable pricing. It's this: why is nobody talking about the oral gap?
$NVO Wegovy pill: 16.6% weight loss. Approved. 600K Rx in 2 months.
$LLY orforglipron: 11.2%. Not approved. Delayed.
48% more efficacy in oral. No competitor on the market. Protected by the SNAC patent that Delhi High Court just confirmed untouchable (Mar 20).
Everyone is debating injectable pricing. The oral monopoly is where the margin lives.
At $149-$299/month with no injection, no cold chain, no needle aversion — that's how you reach the 100M+ Americans with obesity who haven't started treatment yet.
$NVO at 14x P/E. $LLY at 32x. With inferior oral data and no approved pill.
$NVO $LLY #GLP1 #Wegovy #SNAC
English

Wegovy 7.2mg just approved by FDA. Will be launched in April.
Patients in Wegovy 7.2mg trial reached 20.7% WL. With 31% losing more than 25%.
This should make wegovy more competitive vs Tirzepatide.
What about the price……? $NVO does not mention anything about that. I think it will be either same price as 2.4mg ($349) or $399
$LLY $VKTX
English

Something to think about with $NVO.
The Novo Nordisk Foundation already controls 77% of votes and 28.1% of share capital.
Under Danish law, 90% of capital = compulsory squeeze-out. Minority shareholders forced to sell. Company delisted.
The Foundation's net worth? $220 billion. They could buy the remaining 72% tomorrow.
Now consider:
• The company guided -13% — stock crashed
• Next day: $2.1B buyback at historic lows
• Every cancelled share raises the Foundation's % for free
• In 2023-2024, the Foundation sold shares during buybacks to stay at 28%
• In 2025, they stopped selling
Behind the guidance, behind the buybacks, sits the most valuable oral drug platform in history: SNAC. A patented absorption technology that no one can replicate. The pill that delivered 600K prescriptions in 2 months.
If the Foundation reaches 90% and delists, every future dollar from the Wegovy pill, Wegovy HD, oral amycretin, and any peptide delivered via SNAC stays inside the Foundation. Forever.
The world's richest foundation. The world's best oral drug platform. A stock price 75% below ATH.
Connect the dots.
$NVO #GLP1 #Wegovy #SNAC
English

It makes perfect sense once you follow the filings instead of the guidance.
Feb 3: $NVO guides -13%. Pill framed as cannibalization.
Feb 4: $2.1B buyback launched at historic lows.
Feb 8: First Super Bowl ad in 103 years. $30M+.
76K TRx/week. All new patients. Week 11. No oral competitor.
And the stock goes down.
Now ask: who benefits from a depressed price during a $2.1B buyback?
Novo Holdings (28.1% of capital, 77% of votes). Every cancelled share raises their ownership at zero cost.
The pill isn't failing. The price is being managed.
$NVO
English

You're asking the right question but missing the answer.
Sema going generic in India is irrelevant to the real $NVO thesis. Here's why:
The injectable compound patent expired Mar 20. Yes, generics will flood India. That's priced in.
But the oral pill — the actual growth driver — depends on SNAC, a completely separate patent covering the absorption technology. Without SNAC at 0.6-2.1 mmol, oral semaglutide doesn't work.
Delhi High Court confirmed this on March 20: Torrent Pharma was forced to reformulate below the effective SNAC range. They can sell an oral semaglutide tablet. It just won't work as well.
As for "something better than sema" — they already have it. Wegovy HD (20.7% weight loss, FDA approved Mar 19) is at parity with $LLY Zepbound. And the Wegovy pill (16.6%) crushes orforglipron (11.2%).
The market keeps debating semaglutide generics. The real moat is SNAC. Nobody is talking about it.
$NVO $LLY #GLP1 #SNAC
English

There are dozens of boring posts on Sema getting generic in India. Not sure why is big deal for $NVO ? How much India contributed in 2024-2025 ? It does have large diabetes population though. And there are humans there who want better products such as $LLY TZP ( seen its rise there ..) and there is an emerging middle class as well that can afford $100-$200 critical drugs ?
Why not novo focus on getting something better than Sema rather than just competing with generic companies in India and elsewhere. Something competitive with TZP ?
English

Bråse nails it on SNAC. But there's more:
Delhi High Court (March 20) just confirmed $NVO's SNAC patent is untouchable — Indian generic Torrent Pharma forced to reformulate below the effective range. If you can't copy it in India, you can't copy it anywhere.
Plus: Wegovy HD (20.7% weight loss) FDA approved March 19. Injectables now at parity with $LLY Zepbound.
Best pill. Best injection. SNAC moat. 14x forward P/E.
And Novo didn't issue a single press release about the Delhi ruling. The silence is deafening.
$NVO #GLP1 #Wegovy
English

The other day I mentioned that Swedish fund Protean bought back $NVO. Here’s why, according to portfolio manager Richard Bråse:
“Sometimes you have to change your view when the facts change.”
Three key reasons:
• The Wegovy pill
Strong US launch with 300k+ prescriptions in ~2 months. Novo is currently the only one offering a real alternative to injections.
• Technology edge
He highlights SNAC as critical for absorption and believes Novo still has the best product.
“Novo is the only one who can do this… the pill is magical.”
• Valuation
After the selloff, the stock looks too cheap to ignore.
“At DKK 250, it’s an 8% free cash flow yield. You don’t see many like that.”
He adds:
“Novo may not be the most trusted capital allocator, but at this level the stock is too cheap to ignore.”
Protean has rebuilt a larger position than before.
#stocksinfocus #Investing
English