Sources of Truth || !Truth
18.5K posts


Sources of Truth || !Truth
@SourcesTruth
vincit omnia veritas | amor vincit omnia







NAD+ supplementation has become one of the most widely marketed longevity interventions—driven by the assumption that NAD+ levels decline with age and that restoring them can reverse aging processes. But a new study in Nature Metabolism directly measured NAD+ levels across seven independent human cohorts and found something unexpected. Whole-blood NAD+ levels don't decline with age. They remain remarkably stable across the lifespan. The research team used ultra-high-performance liquid chromatography coupled with high-resolution mass spectrometry—one of the most rigorous NAD+ quantification methods available—and measured NAD+ in over 1,100 human blood samples spanning ages 20 to 100. The cohorts included healthy young adults, elite athletes, individuals undergoing lifestyle interventions (resistance training, Mediterranean diet, multidomain interventions), participants in longevity studies, and elderly adults. Across all seven cohorts, NAD+ levels remained unchanged with age. No decline in the third decade. No decline in the seventh. No decline in centenarian offspring compared to age-matched controls. The stability persisted across lifestyle interventions as well. Six months of progressive resistance training in frail older adults: no change in NAD+. Mediterranean diet intervention: no change. Multidomain lifestyle intervention combining exercise, nutrition, and cognitive training: no change. Elite endurance athletes—individuals with exceptional mitochondrial capacity—had the same NAD+ levels as sedentary controls. The only intervention that altered blood NAD+ was direct supplementation with nicotinamide riboside, which increased NAD+ levels by ~40% during treatment—exactly what you'd expect from exogenous NAD+ precursor intake. This contradicts the central hypothesis driving NAD+ supplementation: that systemic NAD+ deficiency is a hallmark of aging that can be restored through lifestyle or dietary interventions. The disconnect appears to be this: tissue-specific NAD+ depletion may occur during aging—particularly in muscle, liver, and adipose tissue—but those changes don't manifest in whole-blood measurements. Blood NAD+ is a poor surrogate for tissue NAD+ status. NAD+ doesn't equilibrate across compartments the way glucose or lactate does. It's synthesized locally within tissues, and blood levels reflect the metabolic activity of circulating cells—primarily erythrocytes and leukocytes—not peripheral tissues where aging-related dysfunction occurs. This has significant implications. First, using blood NAD+ as a biomarker for aging or metabolic health appears to be fundamentally flawed. The stability across age and lifestyle suggests it's tightly regulated within a narrow homeostatic range, regardless of what's happening in metabolically active tissues. Second, it raises questions about whether NAD+ supplementation strategies—nicotinamide riboside, nicotinamide mononucleotide, niacin—are addressing the right problem. If the goal is to restore NAD+ levels in skeletal muscle mitochondria or hepatocytes, does elevating blood NAD+ by 40% translate to meaningful tissue-level repletion? Or are we measuring the wrong compartment and inferring benefits that may not exist? Tissue NAD+ measurements require biopsies—invasive, expensive, and logistically complex for large-scale human trials. Blood is accessible and scalable, which is why it's been used as a proxy. But this study suggests that proxy isn't valid. The third implication is about mechanism. If NAD+ doesn't decline systemically with age, then the benefits observed in preclinical NAD+ supplementation studies—improved mitochondrial function, enhanced autophagy, extended lifespan in model organisms—aren't operating through a simple restoration-of-deficiency model. They may be working through pharmacological activation of NAD+-dependent enzymes like sirtuins or PARPs, independent of baseline NAD+ status. That's a different biological target with different dose-response dynamics and different expectations for human translation. The study doesn't disprove tissue-specific NAD+ decline during aging. It doesn't invalidate NAD+ as a therapeutic target. But it does challenge the foundational narrative that's been used to justify widespread supplementation. If blood NAD+ doesn't decline with age, and lifestyle interventions that improve metabolic health don't alter it, then NAD+ may not be the universal aging biomarker it's been positioned as. The decisions made in the fourth and fifth decades about whether to supplement with NAD+ precursors—and at what doses—should be informed by whether the intervention is addressing a measurable deficiency or activating a pharmacological pathway. Right now, we're measuring blood NAD+ and inferring tissue status. This study suggests that inference is likely incorrect.








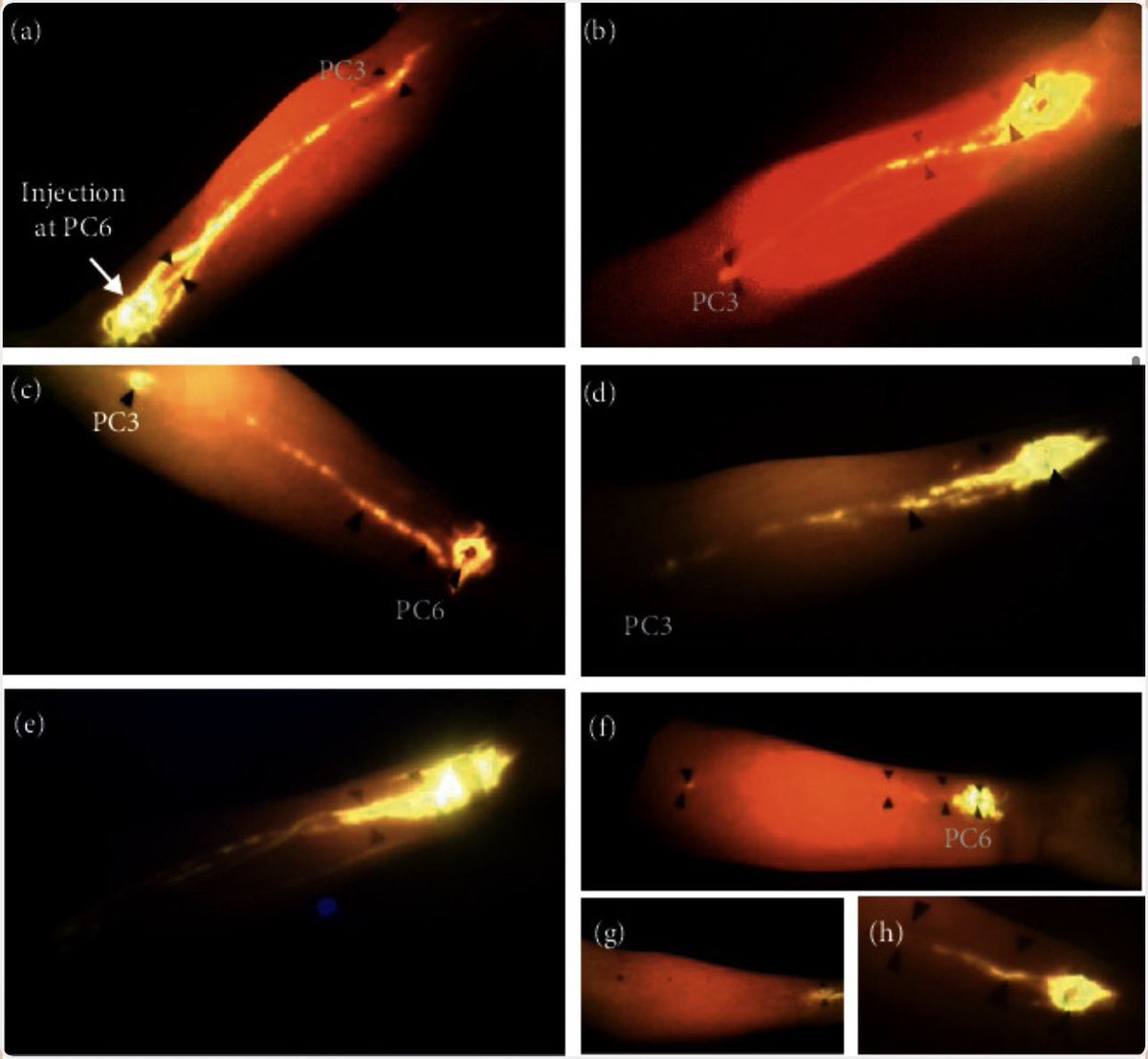
New research from the University of Helsinki reveals a significant connection between severe infections and increased long-term risk of dementia. Analyzing nationwide health registry data from over 375,000 Finnish adults, the study found that hospital-treated infections — including severe urinary tract infections (such as cystitis) and dental caries — were robustly associated with higher dementia incidence years later. For individuals under age 65, certain serious bacterial infections and pneumonia roughly doubled the risk of early-onset dementia. The associations held strong even after researchers adjusted for 27 other chronic conditions, including heart disease, stroke, and mental health disorders. On average, these infections were diagnosed about five to six years before dementia was identified. Scientists suggest that the systemic inflammation caused by severe infections may damage the blood-brain barrier, affect cerebral blood vessels, and accelerate brain changes that contribute to cognitive decline. These findings indicate that what are often viewed as temporary or localized health issues may have lasting effects on brain health. The researchers emphasize that better prevention and prompt treatment of common infections could represent an important and underutilized approach to reducing dementia risk in the population. [Sipilä PN, Korhonen K, Lindbohm JV, Kivimäki M, Martikainen P. The role of noninfectious comorbidities in the association between severe infections and risk of dementia in Finland: A nationwide registry study. PLOS Medicine. 2026;23(3):e1004688. doi:10.1371/journal.pmed.1004688]


Archaeologists have uncovered a remarkable 5,000-year-old artificial island — known as a crannog — submerged in Loch Bhorgastail on the Isle of Lewis in Scotland’s Outer Hebrides. The site significantly predates Stonehenge and challenges long-held assumptions about the origins of these structures. While most Scottish crannogs were previously thought to date to the Iron Age, this Neolithic example began as a large circular timber platform, roughly 23 meters (75 feet) wide. Over centuries, successive generations expanded it by layering brushwood, timber, and stone, creating a communal structure passed down through time. A submerged stone causeway once linked the island to the shore. Hundreds of Neolithic pottery fragments, many retaining traces of ancient food residues, were recovered around the site. This suggests the crannog served as a gathering place for feasting and possibly communal rituals or ceremonies during the Neolithic period. The discovery was achieved through innovative shallow-water archaeology techniques, including diver-operated stereo cameras and aerial drones that produced detailed 3D models merging underwater and terrestrial data. This approach is now enabling researchers to investigate other previously overlooked sites in Scotland’s lochs. [Blankshein, S., Garrow, D., & Sturt, F. (2026). Revealing the Neolithic origins of a crannog: High-resolution 3D recording at Loch Bhorgastail, Isle of Lewis. Advances in Archaeological Practice. University of Southampton]

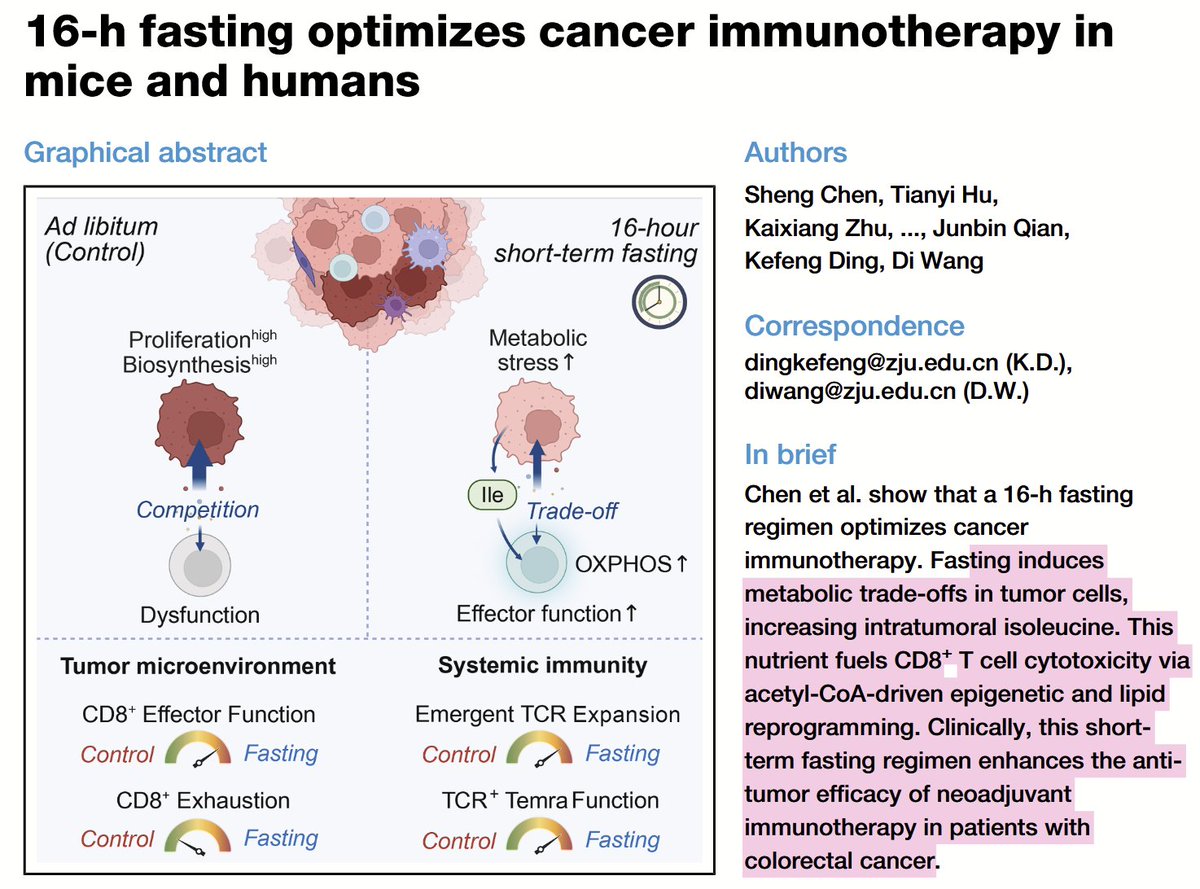



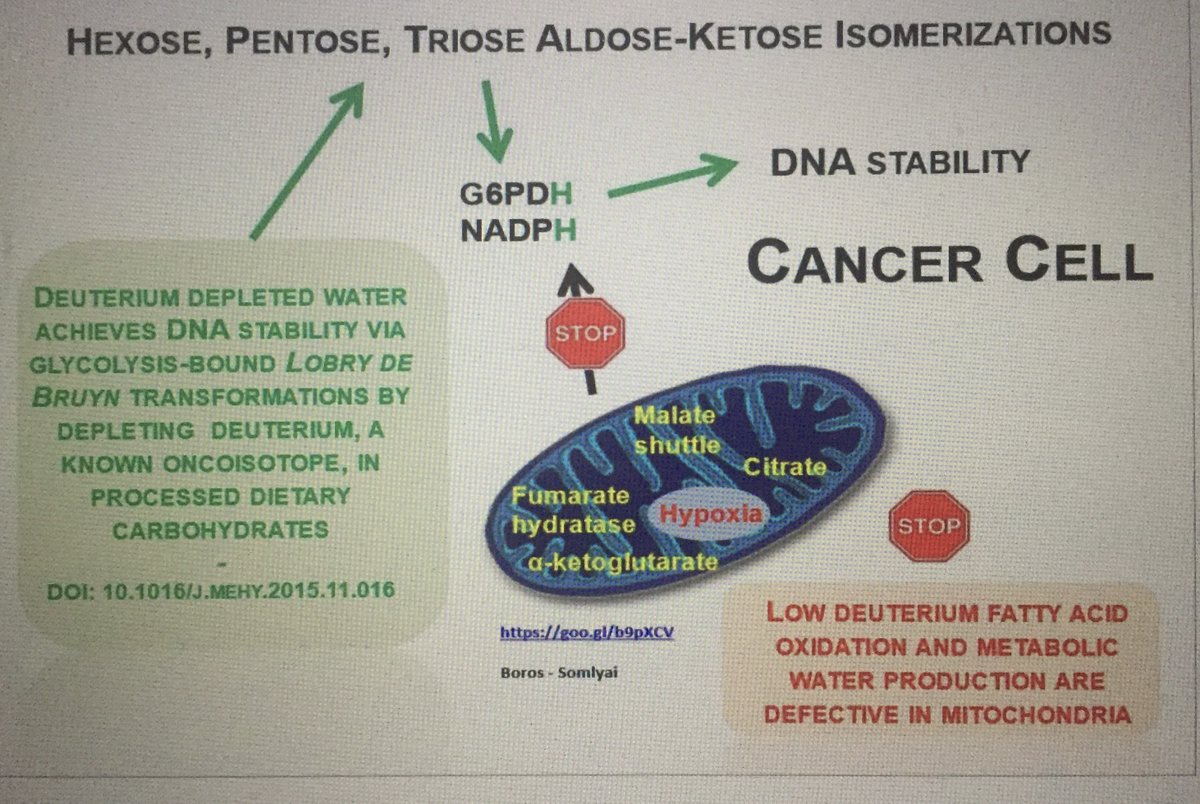
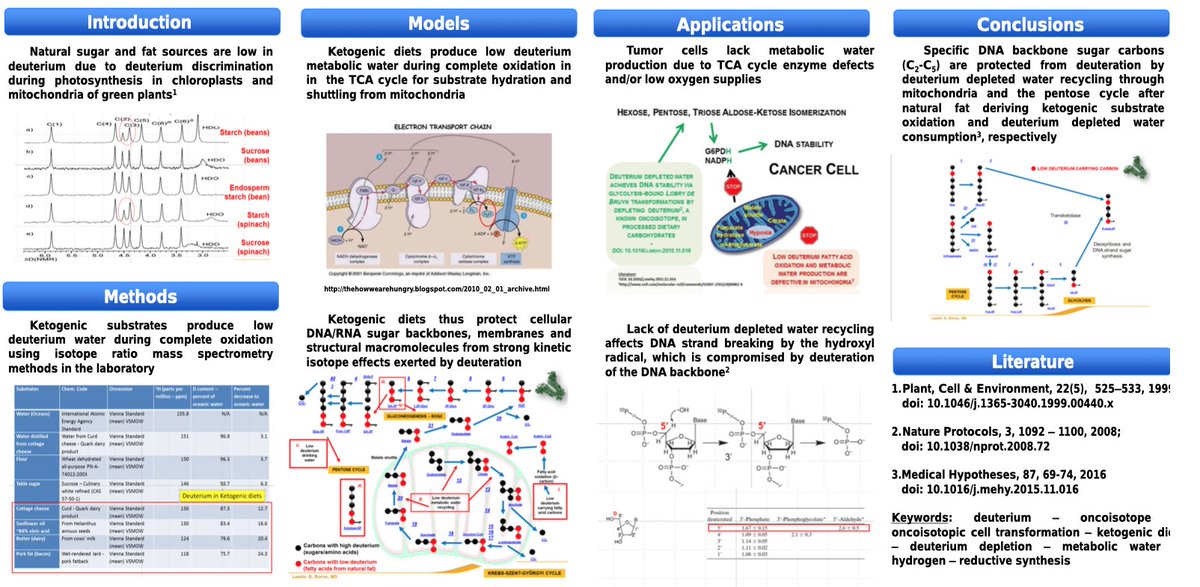


A 16-hour fasting regimen may boost cancer immunotherapy. Transient nutrient stress reshapes tumor metabolism, increasing isoleucine in the TME and enhancing CD8+ T cell function. In mice & patients, short-term fasting improved immune response—offering a feasible way to strengthen treatment. #Fasting, #immunotherapy, #cancer, cell.com/cell-metabolis…