Aumilto Silva, MD.
46 posts


Aumilto Silva, MD.
@aumilto
Medical oncologist and clinical research focused in breast cancer. Hospital Santa Catarina Paulista.BCRI.



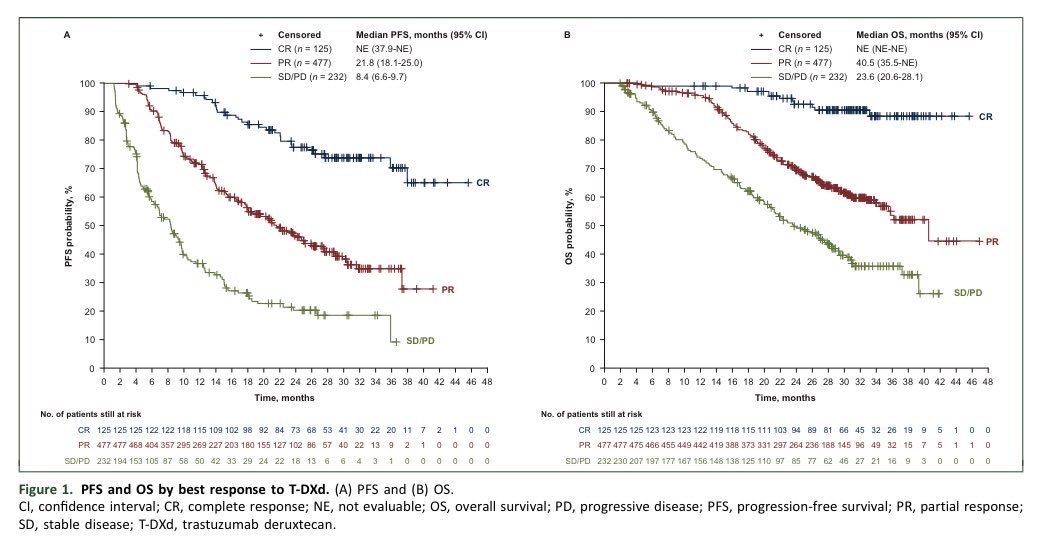
Phase II (n=6): atezolizumab + SRS in TNBC brain mets showed feasibility but minimal efficacy. Median PFS: 1.3 months Median OS: 9.7 months No objective responses Despite biologic rationale, outcomes remained poor. ➡️ CNS metastases in TNBC continue to be one of the hardest settings to treat, highlighting a critical unmet need for more effective systemic + CNS-active strategies. @OncoAlert @nlinmd @antgiorda 👉 link.springer.com/article/10.100…












New 📰: Outcomes of patients with de novo #oligometastatic #BreastCancer treated with curative intent at a single institution. pubmed.ncbi.nlm.nih.gov/41501299/ @jenniferbellon2 @DrHBurstein @stolaney1 @NabihahTayob @LiorBraunstein @nlinmd @drsarahsam #DrFainaNakhlis @hthrparsons