Sabitlenmiş Tweet
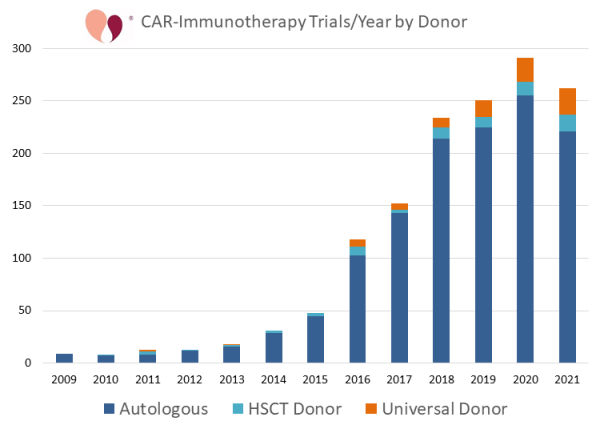
@celltrialsdata Forecasted Growth of Allogeneic CAR-Immunotherapy celltrials.org/news/forecaste… Allo- CAR cell trials increased by 30% or more from 2018 to 2019 and again to 2020, but by 14% from 2020 to 2021... @fverter
English
Alexey Bersenev
23.1K posts
@cells_nnm
Cell trialist.




'Dead End': Radical 20-Year Study Reveals Genetic Cloning Hits a Limit sciencealert.com/dead-end-radic…















Calling all early-stage professionals! Don't miss this opportunity to gain practical experience on an ISCT expert committee and collaborate on real-world projects alongside Key Opinion Leaders from across the globe. Learn more and apply now: buff.ly/3ZgCNyh