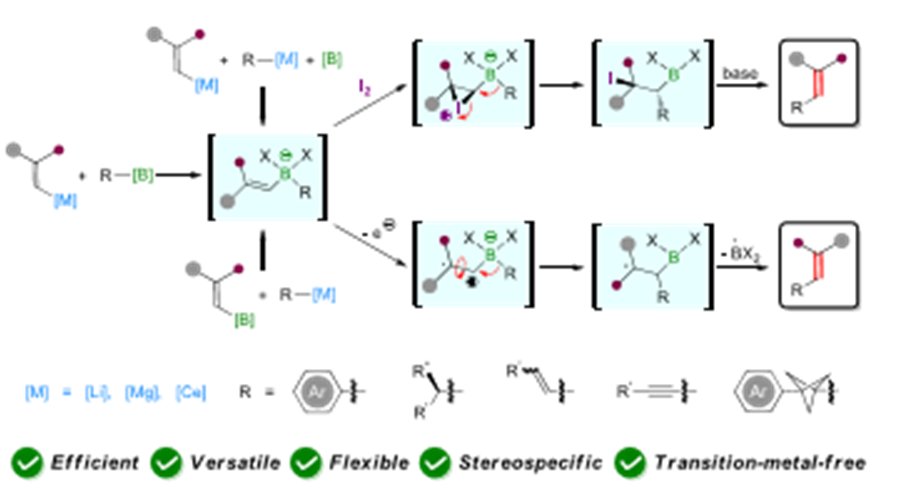
Excited to share our latest work on 4-to-5 skeletal editing towards halogenated pyrroles, and first 1st author paper for Rahma! Congrats 💪 chemrxiv.org/engage/chemrxi…
English
DidierResearchGroup
221 posts


@didier_group
Research group in Darmstadt. Organic and organometallic chemistry. Synthesis of strained ring systems. Electro-coupling.




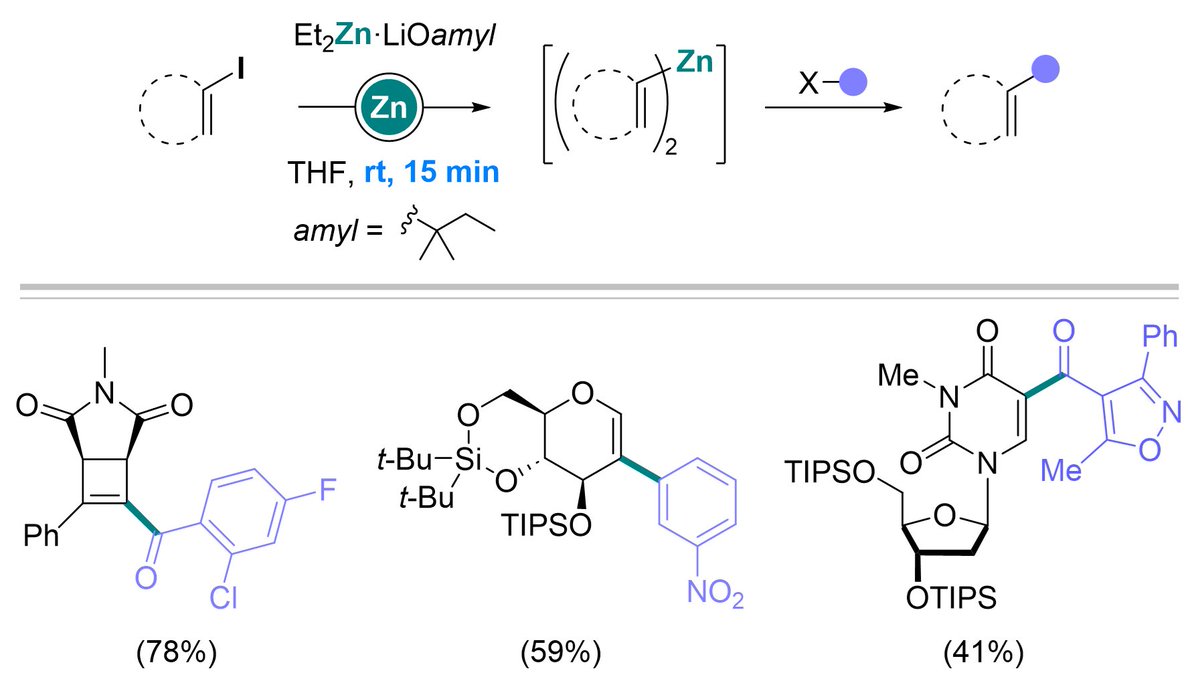

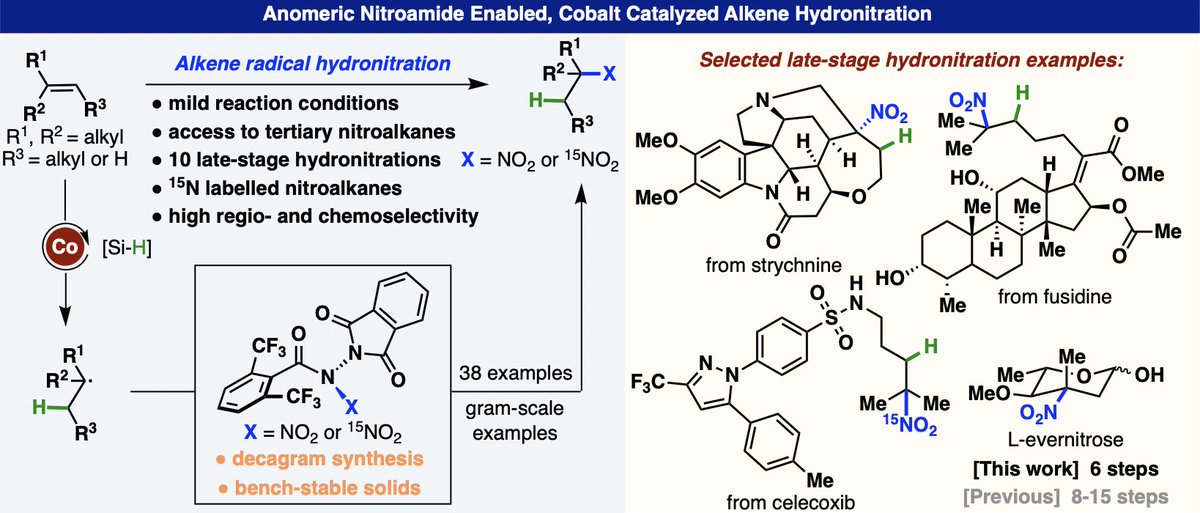


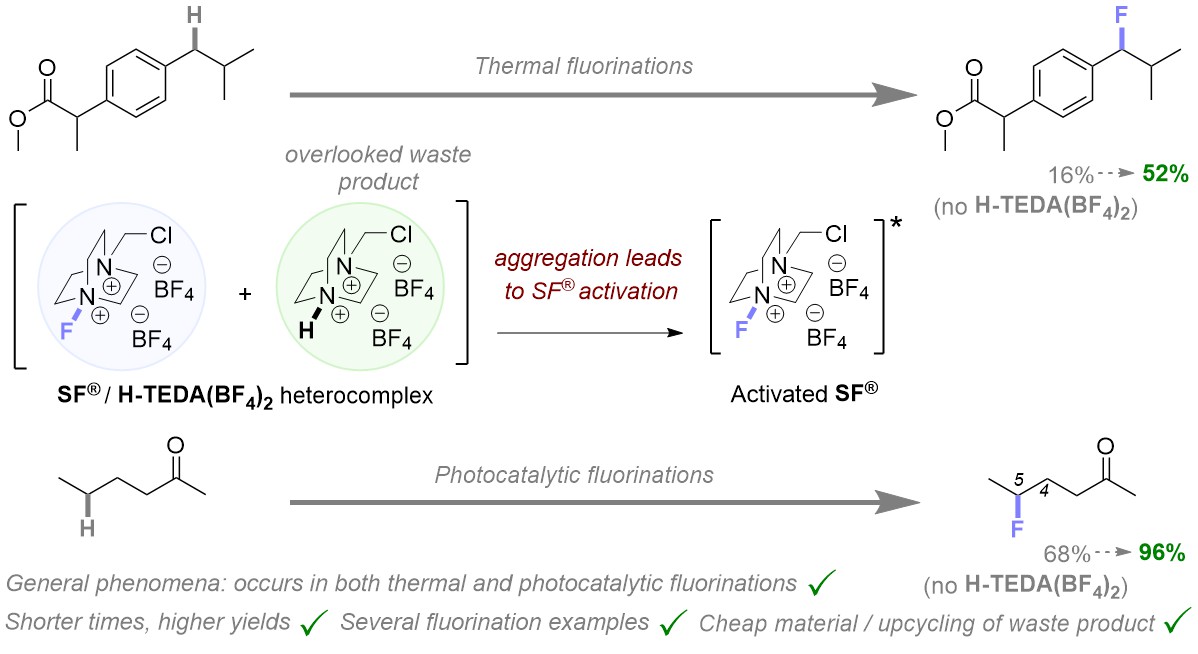








Delighted to join @tuBraunschweig @OCTUBS from today and looking forward to the exciting times ahead! This follows a fantastic 4.5 years @RWTH... I am incredibly grateful to @Schoenebeck_Lab for such strong support during this time.