Feng Tao Ning
660 posts

Feng Tao Ning
@ftning
chesterton's fence needs a california exemption




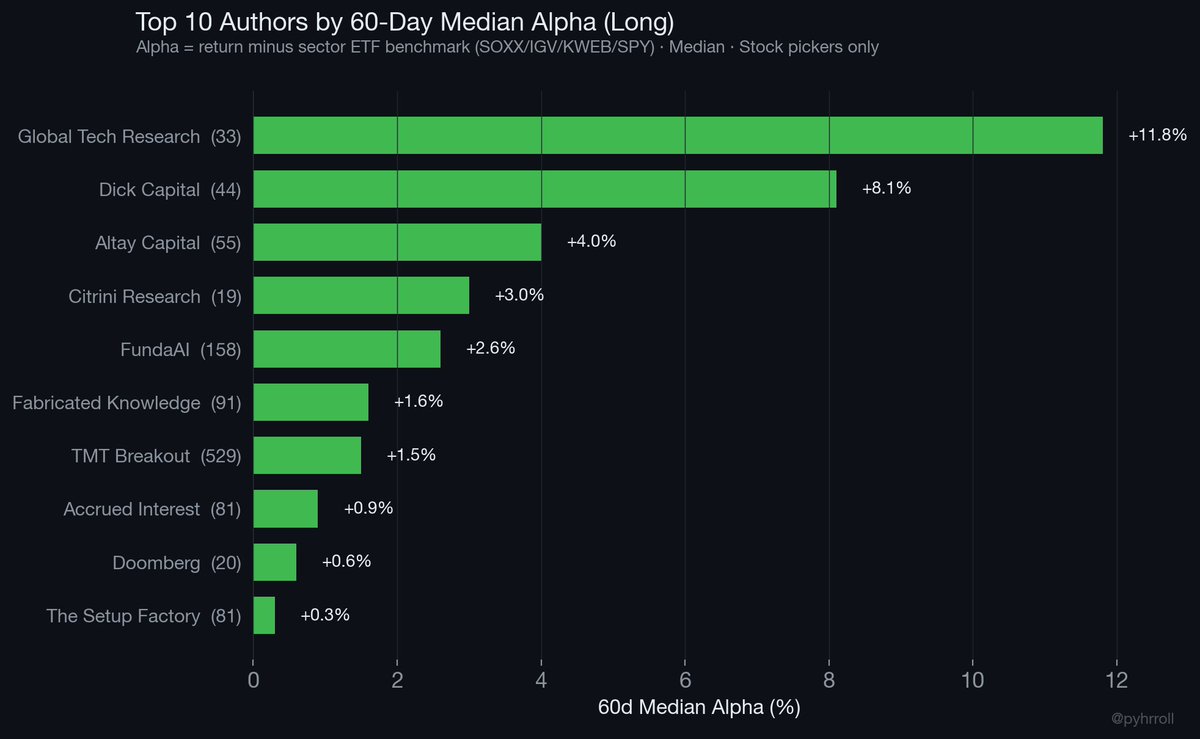
There are almost no original planned-economy socialist states left. We are down to Cuba and North Korea. Everyone else adopted market reforms. Looking at this it might not be very long until only North Korea is left.



Our company trains AI on tumor tissues. My partner had cancer and it was difficult for us to even get her samples to our lab for profiling.






Am I interpreting this Tweet correctly - individualized n=1 mRNA cancer vaccines are trivial to make but are never used in people because you can't do an RCT with one person? This seems like a gigantic regulatory failure.


this is actually insane > be tech guy in australia > adopt cancer riddled rescue dog, months to live > not_going_to_give_you_up.mp4 > pay $3,000 to sequence her tumor DNA > feed it to ChatGPT and AlphaFold > zero background in biology > identify mutated proteins, match them to drug targets > design a custom mRNA cancer vaccine from scratch > genomics professor is “gobsmacked” that some puppy lover did this on his own > need ethics approval to administer it > red tape takes longer than designing the vaccine > 3 months, finally approved > drive 10 hours to get rosie her first injection > tumor halves > coat gets glossy again > dog is alive and happy > professor: “if we can do this for a dog, why aren’t we rolling this out to humans?” one man with a chatbot, and $3,000 just outperformed the entire pharmaceutical discovery pipeline. we are going to cure so many diseases. I dont think people realize how good things are going to get





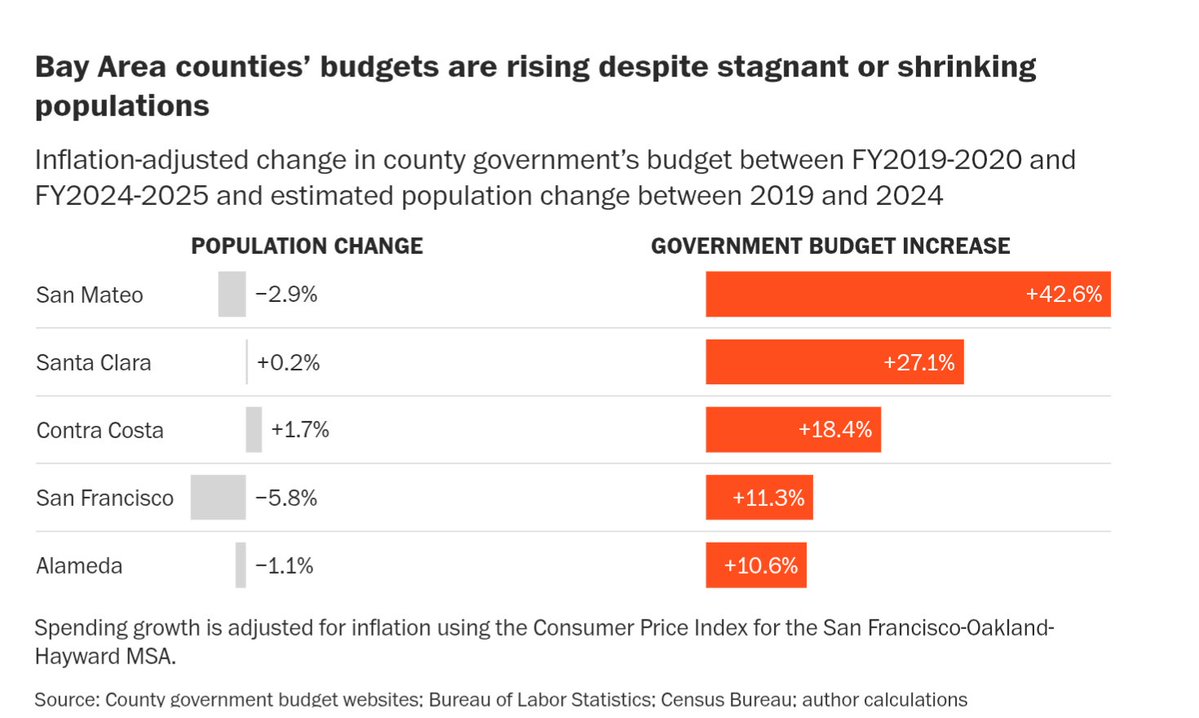
This is wild. theaustralian.com.au/business/techn…

I received a classified briefing from the administration. It is categorically false that they did not plan for Iran closing the Strait of Hormuz. Lawmakers and national security officials have known for years that this was Iran’s plan once their backs were against the wall.











