Letitride1012
10.5K posts


CANCER HAS BEEN CURED
Ivermectin & Fenbendazole cure cancer.
Pass it on.
BREAKING NEWS: First-in-the-World Ivermectin, Mebendazole and Fenbendazole Protocol in Cancer has been peer-reviewed and published on Sep.19, 2024!
The future of Cancer Treatment starts NOW.
My thanks to lead authors Ilyes Baghli and Pierrick Martinez for their incredible inspired work, FLCCC’s Dr.Paul Marik for his extensive work on repurposed drugs and every co-author who worked hard to bring this paper to life.
I hope that this peer-reviewed paper lays the groundwork for a brand new future for Cancer Treatment.
Many of you know that I have been helping thousands of Cancer patients with high dose Ivermectin, Mebendazole, and Fenbendazole
FOLLOW ME, THE NEXT DROP WILL BE SHOCKING

English

@DonutsBaga I thought they had a new chief that was going to take them in a different direction. Same shit different day.
English

💥Canton has been paying Sean Goode for (22 weeks), while he’s on Admin Leave for allegedly exchanging racist texts with disgraced former trooper Michael Proctor. What are they hiding?
JoeyBagaD🍩nuts@DonutsBaga
💥IT HAS BEEN (10 WEEKS) SINCE CPD SERGEANT SEAN GOODE WAS PLACED ON ADMIN LEAVE FOR MISCONDUCT! 💥WHAT KIND OF SHADY GRIMEY SHIT IS @CantonMAPolice UP TO? 💥MAYBE CANTON CAN HIRE 5 STONES INTELLIGENCE TO GET GOODEY OUT OF THIS MESS!
English
Letitride1012 retweetledi
Letitride1012 retweetledi

Update on Andrew’s Condition — God Is Amazing!
Here is the latest update on Andrew’s condition. We are so grateful—God is truly amazing! @bullishbruk
English
Letitride1012 retweetledi

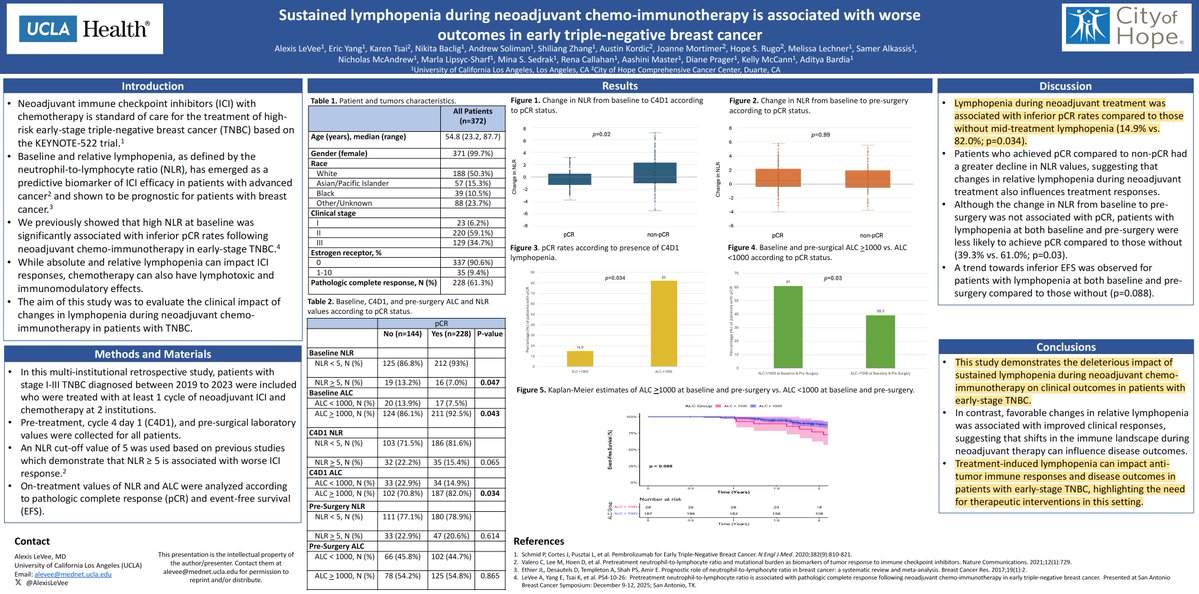
The evidence generated by UCLA is compelling. This presentation entitled, "Sustained lymphopenia during neoadjuvant chemo-immunotherapy is associated with worse outcomes in early triple-negative breast cancer" was presented at ASCO-IO (Feb 2026).
#3 ASCO-IO (Feb 2026): Early Stage Triple Negative Breast Cancer (TNBC)
In a 372 patient study, the data shows that only 15% of patients with early stage TNBC have a pathological complete response (pCR) when the ALC is less than 1,000 cells while 82% of patients with NK and T cells (ALC greater than 1,000 cells) have a complete response after therapy. This difference is stark and significant (p=0.034).
Dr Bardia states:”Sustained lymphopenia and the development of relative lymphopenia during neoadjuvant chemo-immunotherapy may reflect impaired anti-tumor immune responses in patients with early-stage TNBC, highlighting need for intervention therapeutics in this setting“
Conclusion: Treatment-induced lymphopenia can impact anti-tumor immune responses and disease outcomes in patients with early-stage TNBC, highlighting the need for therapeutic interventions in this setting
aacrjournals.org/cancerimmunolr…

English
Letitride1012 retweetledi

Dr. Patrick Soon-Shiong dropped a cancer paradigm shift that could change everything:
“Cancer isn’t the disease — it’s the symptom. The real disease is the collapse of your immune system.”
Instead of destroying the body with aggressive treatments, he says we should focus on rebuilding and supporting the immune system so it can actually fight back.
No single magic bullet, but the science is finally moving fast — and he’s calling for more clinical trials to turn this new understanding into real results.
This one idea flips the entire approach to cancer treatment on its head.
What do you think — could shifting from attacking cancer to strengthening the immune system be the breakthrough we’ve been waiting for, or is it still too early to tell?
English
Letitride1012 retweetledi

$IBRX Three months ago, a Saudi government official said a phrase on camera that does not appear in any ImmunityBio press release:
"First in the world."
He was talking about Saudi Arabia's approval of ANKTIVA for lung cancer post-checkpoint therapy. First regulator on Earth to approve the IL-15 plus checkpoint combination. Not the US FDA. Not the EMA. Not the MHRA. Not Japan. Not China.
The quote is Ammar Altaf, Assistant Deputy at Saudi Arabia's Ministry of Investment, speaking at the inaugural U.S.-Saudi Biotech Alliance summit on January 20, 2026. (LA Times Studios)
What he said next is the reason today's press release matters.
WHAT HAPPENED TODAY
April 21, 2026. ImmunityBio announces ANKTIVA is commercially available in Saudi Arabia. Two indications live: bladder cancer (NMIBC CIS) and metastatic non-small cell lung cancer post-checkpoint. Distribution through Biopharma and Cigalah Healthcare. First patients identified; dosing begins soon. (ImmunityBio press release, Apr 21 2026)
Sixty days from partnership to patient.
A new pharma launch into a fresh emerging market is usually a multi-quarter operation. This one went end-to-end in two months.
That by itself is a PR. What makes it a story is what's inside the PR, and what was said three months ago in San Francisco.
THE HIDDEN LINE IN THE LABEL
Buried in the Saudi approval language, one sentence:
"This indication is approved under accelerated approval based on the increase of ALC associated with overall survival in single arm study."
Translate that.
ALC is absolute lymphocyte count - the number of lymphocytes in a patient's blood. Every oncologist reads it before every follow-up visit. It is the most ordinary lab value in cancer medicine.
What that sentence says: a regulator just formally accepted ALC movement as a surrogate for overall survival.
That is the first time any regulator has done this for an IL-15 therapy.
Twenty-four hours earlier, Patrick Soon-Shiong surfaced a UCLA / City of Hope SABCS study by Bardia et al. - a 372-patient analysis showing that baseline lymphopenia (low ALC) predicts lower pathologic complete response and worse event-free survival in early-stage triple-negative breast cancer. (AACR abstract PS4-10-26)
The abstract's own conclusion ends:
"Develop potential therapeutic strategies to improve lymphopenia in patients with early-stage TNBC."
Saudi just made ALC-movement an accepted endpoint for lung cancer. Bardia just showed ALC predicts outcome in breast cancer. Two dominoes landing together in public, on the same biomarker.
ANKTIVA is the FDA-approved IL-15 superagonist designed to restore lymphocytes by mechanism - the first of its kind in oncology.
THE MANUFACTURING PARTNER IS A GOVERNMENT
Back to Altaf. He went on to explain how Saudi Arabia plans to manufacture ANKTIVA locally:
"We can actually produce at scale this hopefully new product through a new robotic system that currently has been developing with the Saudi entity, which is the National Guard healthcare ecosystem."
Re-read that.
The manufacturing partner is not a commercial contract distributor. It is the Saudi Arabian National Guard healthcare ecosystem - a state institution that operates a major hospital network across Riyadh, Jeddah, and Dammam.
Altaf also confirmed the relationship is not new:
"We've been working with ImmunityBio for almost a year... we've met with Dr. Patrick Soon-Shiong at the beginning of 2025."
Twelve months of engagement. A state-run healthcare ecosystem as manufacturing partner. An "almost a year" build-up to today's commercial launch.
That is not distribution. That is sovereign industrial policy.
WHAT A SAUDI CEO POSTED A MONTH AGO
March 21, 2026. Mohammed H. Al Qahtani, CEO of Saudi Arabia Holding Co., published a post on LinkedIn titled "Riyadh: The New Global Frontier for the NK Cell Revolution."
The post names the facility: NantG-Force.
The specs: "sample processing and activation in record time (as fast as 30 minutes logistically)... produce billions of active NK cells at a fraction of current costs."
And the scope: "Saudi Arabia will host the second robotic factory in the world (after the US), based in Riyadh... the primary distributor and hub for exporting these advanced biological therapies across the MENA region." (LinkedIn, Mar 21 2026)
Al Qahtani listed four therapeutic targets in his post: bladder cancer. Lung cancer. Triple-negative breast cancer. Pancreatic cancer.
Bladder and lung are approved.
Breast cancer is what @DrPatrick has been posting about for the last forty-eight hours.
Pancreatic is the QUILT-88 overall-survival benefit ImmunityBio has reported before.
The Saudi CEO published the roadmap thirty days before the American audience saw PSS's breast cancer drumbeat.
WHAT SAUDI SAID NEXT
The public Saudi posture is not "one drug, one indication." It's a pipeline.
PSS on Sean Spicer Ep 631: "Saudi approved for bladder cancer AND lung cancer, committed to additional cancers." Saudi leaders also stated ambition to bring ANKTIVA access across the Middle East and Africa. (Sean Spicer Ep 631, Jan 20 2026)
Al Qahtani's LinkedIn post listed four therapeutic targets explicitly - two approved (bladder, lung), two "showing highly promising clinical results" (triple-negative breast cancer, pancreatic cancer).
And today's PR closes with the company's own stated intent: "We continue to work with the same level of diligence and commitment to expand access to ANKTIVA for eligible patients across the Middle East and North Africa."
Three independent sources - the inventor, a Saudi CEO, and ImmunityBio's own press release - saying the same thing in different words: bladder and lung are the first two doors. Breast and pancreatic are already in the queue. The stated ambition extends to the whole region.
Each new indication is its own SFDA filing, its own review, its own timeline. Nothing is automatic. But the direction is public, on record, and corroborated from three sides.
SIXTY DAYS FROM HANDSHAKE TO PATIENT
The timeline is clean:
- February 2026: ImmunityBio announces MENA partnership with Biopharma and Cigalah Healthcare.
April 21, 2026: ANKTIVA commercially available, first patients identified.
Two months, end to end - within the stated deadline.
Inside those sixty days, a wholly-owned Saudi subsidiary was activated, a distribution network was stood up, two indications were loaded into formulary, and patients were identified in at least two tumor types.
In the middle of what the CEO called "a fluid situation in the region."
This is not the speed of a company waiting for permission.
WHILE WASHINGTON HELD TEN-MINUTE MEETINGS
On January 20 - the same day Altaf spoke in San Francisco - Patrick Soon-Shiong appeared on The Sean Spicer Show, Episode 631, and told the story of the FDA meetings.
The Saudi FDA, he said, reviewed "1.7 million pages" before issuing approval.
The U.S. FDA Commissioner, Dr. Marty Makary, gave him "a 10 minute meeting. 'I've gotta go.'" (Sean Spicer Ep 631, Jan 20 2026)
One review reviewed a million and a half pages. The other ended in ten minutes.
The contrast is not an editorial. It is a document trail.
PSS also described the scene in Riyadh at the Trump-MBS state visit, May 2025: "He turned to MBS and said, this is important for both our countries."
Trump invited PSS to the White House. Miller, Wiles, Vance, Kennedy - all met. JD Vance, walking in: "Hi doc, have you cured cancer yet?"
And yet. "It stuck at levels beyond my pay grade."
Saudi approved in January. Commercial launch in April. The U.S. is still reviewing additional indications since 2024.
WHAT THE COMPANY IS NOW CALLING ITSELF
In today's press release, the "About ImmunityBio" paragraph has changed.
The company is no longer describing itself through bladder cancer, or even through ANKTIVA. The lead is now:
"At the core of our strategy is the Cancer BioShield platform, which is designed to stimulate critical lymphocytes, including natural killer (NK) cells, cytotoxic T cells, and memory T cells via our proprietary IL-15 superagonist."
A platform to stimulate lymphocytes.
That is the same thing Bardia's abstract called for. That is the same thing the Saudi label accepted as an endpoint. That is the same thing the National Guard healthcare ecosystem is now building a factory to produce at scale.
The company has caught up to its own thesis.
THE CLOSE
Sixty days, partnership to patient.
One Saudi CEO listing the roadmap in March.
One academic team presenting the biomarker data in December.
One regulator writing the ALC surrogate into a drug label today.
Five regulators in twenty-four months. A second factory on Earth. A platform, not a drug.
Today was Saudi and MENA, bladder and lung. Yesterday's post mapped the $50 billion global checkpoint-failure population (IL-15 post, Apr 20) - one front of many. Sepsis Phase 3. Lymphoma CAR-NK Phase 2. Pancreatic QUILT-88 overall-survival benefit. Long COVID Phase 2. Glioblastoma pilot. Lymphopenia BioShield expanded access (570+ patients). Triple-negative breast cancer per Bardia's drumbeat. Same biology. Different battles.
Today's PR is one line in a much longer ledger. The ledger is real; the lines are arriving one at a time.
Saudi Arabia spent 1.7 million pages and a sovereign factory to deliver this.
The U.S. spent 10 minutes.

English
Letitride1012 retweetledi

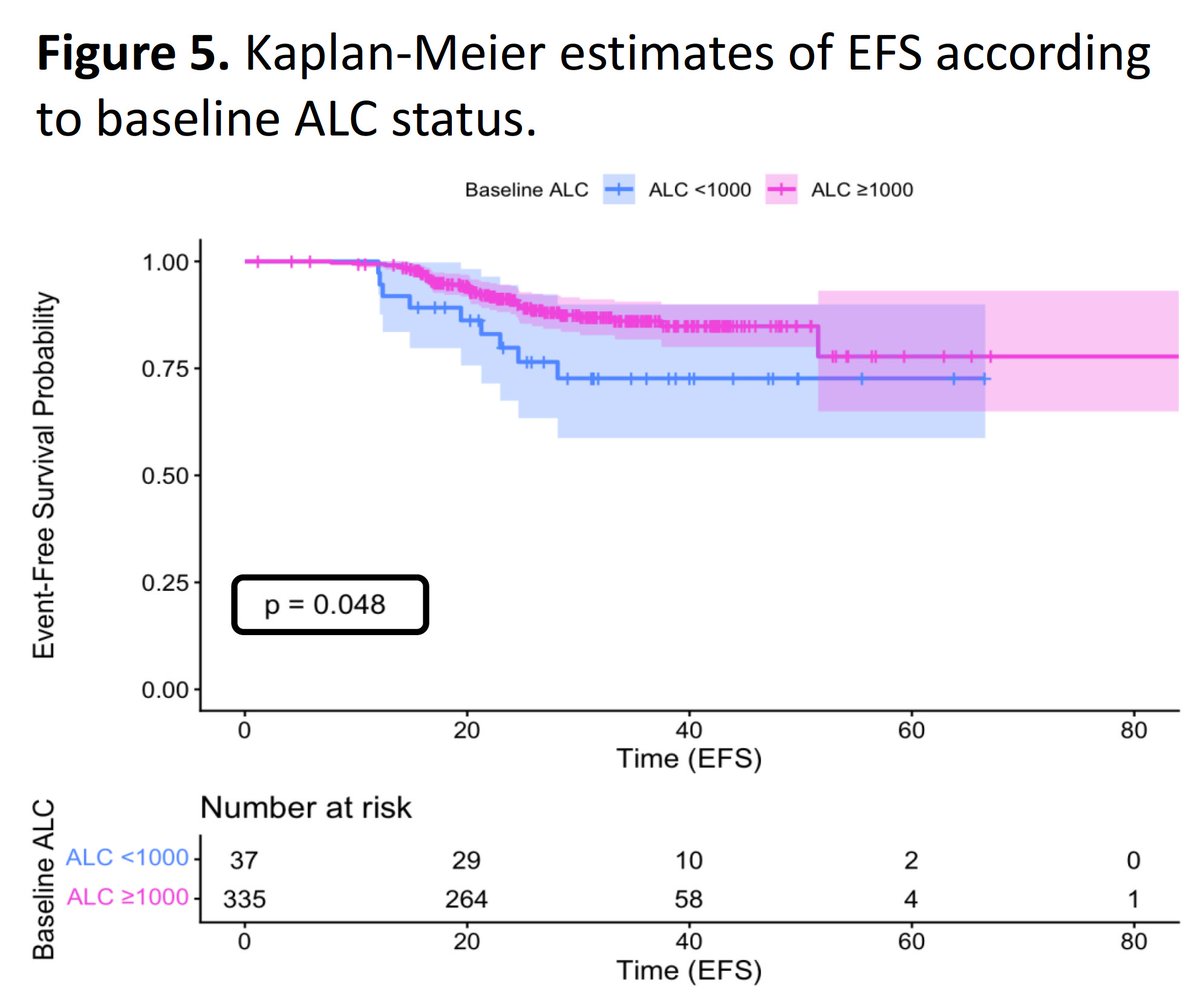
Here we go with the drumbeat of evidence of how lymphopenia results in poorer prognosis in breast cancer, regardless of type and regardless of stage by Dr. Aditya Bardia, a renowned UCLA breast cancer oncologist.
#1 Dec 2025:
Early stage TRIPLE NEGATIVE BREAST CANCER at San Antonio Breast Cancer Symposium (SABCS): A study demonstrating that lymphopenia is associated with statistically LOWER pathological complete response (pCR) rates in patients with early-stage triple negative breast cancer (TNBC) treated with neoadjuvant chemo-immunotherapy.
News tomorrow at @AACR on Stage 1, 2, and 3 breast cancer.
aacrjournals.org/clincancerres/…

English
Letitride1012 retweetledi

$IBRX Thirty-six days. Same FDA.
March 13: warning letter to Patrick Soon-Shiong for citing peer-reviewed IL-15 evidence. April 18: Makary, beside Trump, calls two veteran testimonies and a Rogan text scientific data. Executive order signed.
One agency. Two rulebooks.
APRIL 18, 2026.
Oval Office. Saturday morning. President Trump signs an executive order expediting FDA review of psychedelic therapeutics. The target drug is Ibogaine. Schedule 1 under the 1970 Controlled Substances Act. Zero FDA approvals in American history.
Trump, from the opening remarks, on how the executive order came to be:
"I got a call from a number of people, including the great Joe Rogan. And he said, we have to do something about this. I looked into it. I called Bobby, I called Oz, I called Marty and Jay. It was uniform support. And I said, so why would we wait three or four years to get it done? Well, for ten years, frankly. Let's get it done immediately."
"This has probably never been anything happened so quickly."
Two Navy SEAL testimonies served as the scientific case. Marcus Luttrell: "Five years ago I got my life back." Rob O'Neill: "It saved my life and then it saved my wife."
FDA Commissioner Marty Makary, standing next to the President: "The stories of those individuals with dramatic results, that is data. That is scientific data, even from a single individual."
Trump's moral case for the speed, from the same remarks: "Since 9/11, we've lost over twenty-one times more veteran lives to suicide than on the battlefield."
MARCH 13, 2026.
The same FDA's Office of Prescription Drug Promotion sent Patrick Soon-Shiong a warning letter. The violation: citing efficacy data in a @seanspicer podcast, and a television commercial that ImmunityBio had voluntarily submitted to the FDA for pre-airing review. The ad never aired. Instead of review notes, the agency issued a public warning.
The evidence he cited was the compounding record of IL-15 across four diseases.
Bladder cancer: ANKTIVA FDA-approved since April 2024 for non-muscle invasive disease that had failed BCG. Now approved in thirty-four countries.
Lung cancer: Saudi Arabia granted first-line approval for non-small cell lung cancer in January 2026. QUILT-2.023 Phase 2 showed a statistically significant increase in absolute lymphocyte count versus checkpoint inhibitor alone, p=0.0065.
Sepsis: Hotchkiss lab at Washington University, 2010, Journal of Immunology - IL-15 reverses immune cell apoptosis and improves survival. Two hundred thousand Americans die of sepsis every year. No approved immunotherapy exists. Hotchkiss followed with a 2014 call to action in the New England Journal of Medicine. Nobody built it.
Long COVID: UCLA and Mount Sinai clinicians presenting at AACR 2026 - COVID-induced lymphopenia driving cancer recurrence in patients previously in remission. The same collapse mechanism. The same molecule that restores it.
Four diseases. One molecule. Documented since Sinkovics in 1969. Cloned by the NIH in 1995. Ranked number one by the NCI in 2007. Buried by the American government for fifty-seven years.
The same FDA that declared single-patient stories are scientific data six weeks later called this evidence misleading.
Same agency. Same commissioner. Opposite standards.
In that same Oval Office, a Stanford researcher praised the administration's goal: "The United States will be the first country in the world to treat mental health like we do cancer."
The one American company treating cancer with the precision standard Stanford was praising had been sanctioned by the same FDA thirty-six days earlier.
THE MOLECULE THE GOVERNMENT KNEW
Joseph Sinkovics was running cytotoxicity assays at MD Anderson in 1969. The experiment needed a healthy control. He used himself. His own lymphocytes attacked and killed a patient's chondrosarcoma cells. Not slowly. Not partially. They surrounded the tumor cells and lysed them.
On December 3, 1969, he photographed what he saw under the microscope. The first images in human history of NK cells attacking human cancer.
The National Cancer Institute sent site visitors. They could not comprehend how immune reactions could exist without pre-immunization. They called it an in vitro artifact. They canceled his grant.
Fifty-seven years later, on April 18, 2026, the successor to that same FDA declared a single veteran's Ibogaine testimony scientific data. Thirty-six days earlier, the same agency had warned Patrick Soon-Shiong for citing published evidence of exactly what Sinkovics had photographed.
He died on January 24, 2022, at ninety-seven. He had spent half a century proving them wrong. He never saw the day the FDA admitted he had been right.
THE PROPHECY
1995: the NIH cloned IL-15. The American government owned the molecule.
July 12, 2007: the National Cancer Institute gathered America's top cancer immunologists for one question: of 124 candidate molecules, which would cure cancer?
The molecule ranked number one was IL-15.
Above anti-PD-1 - the class that would become Keytruda, the top-selling drug in the world.
Above every molecule that would become a multi-billion-dollar cancer drug over the next seventeen years.
The workshop wrote its own failure condition into the manuscript, before any ranking was discussed: "If a year or two goes by and the list remains substantially unchanged, it would be a signal that the current system for developing immunotherapeutic agents is not working optimally."
Seventeen years passed.
In 2010, Richard Hotchkiss at Washington University published in the Journal of Immunology that IL-15 reverses immune cell apoptosis and improves survival in sepsis. In 2014, he wrote a call to action in the New England Journal of Medicine: greater collaboration required between oncologists, infectious-disease specialists, translational immunologists, and pharmaceutical companies.
Nobody built it.
The NIH knew. The NCI knew. Hotchkiss knew. The United States federal government knew what IL-15 did in 1969, 1995, 2007, and 2010.
It refused to build the drug for fifty-seven years.
On April 18, 2026, the same federal apparatus - the same FDA, the same NIH, the same president who ten years ago had considered Patrick Soon-Shiong for NIH Chief - gave Ibogaine its first-ever FDA clearance to test on American patients and three National Priority Vouchers in a single Oval Office signing, on the strength of two veteran testimonies and a Joe Rogan text message.
The NCI's number one molecule is still waiting.
THE BUILDER
In 2008, one man read Joseph Sinkovics's chapter. He had made his fortune developing Abraxane. He had the resources. He read the 2007 NCI workshop manuscript. He understood that manufacturing IL-15 at clinical scale was the bottleneck.
@DrPatrick started building.
2012: N-803 development begins. 2013: first patient dosed. April 2024: the FDA approves ANKTIVA for non-muscle invasive bladder cancer. Seventeen years after the 2007 workshop. Twelve years of clinical trials. Two decades of peer review.
The same FDA would grant Ibogaine its first-ever clinical-trial clearance in one Oval Office signing.
January 2026: Saudi Arabia approves ANKTIVA for non-small cell lung cancer, the first country outside the United States. Part of Crown Prince Mohammed bin Salman's Vision 2030 biotech strategy.
February 2026: the Saudi National Guard Healthcare Ecosystem announces a robotic manufacturing partnership with ImmunityBio. Sovereign-scale IL-15 production.
April 2026: Portugal. The Health Minister personally requests a meeting. A new integrated next-generation immunotherapy center treating a thousand patients a day. PSS on-site: "Never seen anything like this."
April 15, 2026: Cleveland Clinic partnership. CAR-NK cell therapy infused with ANKTIVA - the next chapter of the Bioshield Platform, built with America's most-trusted hospital system.
Greece calling. UAE moving fast. The United Kingdom next.
Today ANKTIVA is approved in thirty-four countries, from the United States to Macau.
The country that built it sent its developer a warning letter for saying so.
TWO CASES
On April 18 the President accelerated a drug with zero FDA approvals, supported by Phase 2 observational data and two veteran testimonies delivered on camera.
In that same Oval Office, an advisor told the room of reporters: "In 2016, the lead professor at Johns Hopkins said this was the most promising therapy he's seen in his career. There's been a hold. There's been incentives against this. And the bravery and leadership of President Trump." A reporter asked what he meant. "Artificial barriers," he answered.
ANKTIVA is already approved. Already in thirty-four countries. Already the product of fifty-seven years of federal government research conducted at the NIH, the NCI, and academic medical centers funded by taxpayers. Already tested in Phase 3 for sepsis and first-line lung cancer.
Already the molecule the American government unanimously ranked number one in 2007 and then failed to build for seventeen years while one man built it alone.
For Ibogaine, the hold broke on April 18. For IL-15, the hold is an OPDP warning letter dated March 13.
THE RELATIONSHIP
November 2016: President-elect Donald Trump interviewed Patrick Soon-Shiong for NIH Chief during the first presidential transition. Discussed a health care czar role with broad portfolio over the entire U.S. healthcare system.
May 2017: PSS was placed on the Health IT Advisory Committee.
May 2025: Patrick Soon-Shiong met President Trump and Crown Prince Mohammed bin Salman in Saudi Arabia. He addressed them both directly. The curing of cancer, he told them, is the best foreign policy a nation can have. Not left. Not right. A humanitarian tool that is also a diplomatic instrument.
November 18-19, 2025: Trump and MBS held their White House summit.
January 2026: Saudi Arabia approved ANKTIVA.
The President did that.
THE SIGNAL - Part 1
April 18, 2026. The pen is down. The room is still full.
FDA Commissioner Marty Makary, on camera, answering a reporter about timeline:
"The fastest FDA approval was forty-two days during the HIV epidemic. On our national priority voucher program under President Trump, we are now down to that timeframe on a routine basis instead of a year."
"In my opinion, we need the same level of urgency we had with HIV for PTSD and depression and anxiety and our mental health disorder and cancer and heart disease and so many other conditions."
"You're going to see decisions out from the FDA later this summer as soon as we get the applications."
Thirty-six days after the warning letter, the same Commissioner committed his agency on live television to forty-two-day cancer approvals.
THE SIGNAL - Part 2
Hours later, Patrick Soon-Shiong posted nine words:
"The President can make huge advances in transforming the health of Americans."
That evening, Commissioner Makary posted on X:
"FDA is moving proactively to advance potential therapies. It's time for new approaches."
Patrick Soon-Shiong retweeted it.
Before midnight, he posted one more:
"News will break tomorrow. Evidence of an increase of over two times the risk of distant recurrence of breast cancer in patients in remission with lymphopenia. ALC matters."
Then one more again - quote-tweeting the WIN Consortium's rare-cancer personalized-medicine meeting:
"So glad to see that awareness of this IL-15 superagonist is beginning - yet it had awareness through evolution 450 million years ago, then again in 2007, almost 20 years ago by NCI and NIH. And now 2026. Science enlightenment takes time."
A global oncology consortium openly discussing ANKTIVA in rare cancers. The 2007 NCI moment being re-litigated by the medical establishment itself. On the record.
ALC is absolute lymphocyte count. The molecule that restores it is ANKTIVA. The Commissioner committed to forty-two-day decisions.
The applications are the only thing missing.
THE RECKONING
The FDA that blocked IL-15 for fifty-seven years just declared that single-patient stories are scientific data.
The President who opened Saudi Arabia for Patrick Soon-Shiong just demonstrated on live television that he can accelerate the FDA at will.
The man the NCI's own 2007 workshop unanimously ranked above Keytruda is already approved, already in thirty-four countries, and already running two Phase 3 trials.
The buried scientists - Sinkovics, Hotchkiss, Berzofsky, Monneret - documented IL-15 for five decades. The NIH cloned it. The NCI ranked it. Hotchkiss called for it. Nobody built it.
One man built it.
More to come.

English
Letitride1012 retweetledi

My husband was diagnosed with Glioblastoma brain cancer in October. He has now had 2 normal brain scans.
You can be a deplorable person. I personally having our lives turned upside down and now being given a chance at life will continue to fight for others.
I hope you are never in the situation of a death sentence. The first brain scan is before, the second after. Jesus is Lord.


English
Letitride1012 retweetledi

News will break tomorrow.
Evidence of an INCREASE OF OVER 2TIMES THE RISK of distant recurrence of breast cancer( in patients in remission ) with lymphopenia ( low TCells and low NK cells). So ALC matters in patients in remission with early stage (1 to 3 ) breast cancer . An easy test to monitor lymphopenia and to address this increased risk.
English
Letitride1012 retweetledi

North Andover Massachusetts select board made the choice to have a CLOSED door meeting last night to address concerns about Cheif Gray for NAPD. Gray could not be bothered to even show up in person to this meeting. He then made the executive decision himself to place HIMSELF on paid administrative leave….
NAPD now has their own Cheif as well as Patrick Noonan who’s been on paid leave for months now and was the cop who shot #KelseyFitzsimmons on PAID administrative leave.
Oh and just in case you forgot Justin 👽Alyaian is still suspended WITH pay from the @NorthAndoverFD after hundreds of videos surfaced of him doing drugs ON VIDEO ….
Below is a group picture of the members of your select board North Andover who are facilitating this! THE WORLD IS WATCHING 🎪 🤡 @NoAndoverPolice
#NorthAndover
#FreeKelseyFitzsimmons
#JustinAlyaian
#CheifCharlesGray #NorthAndover
@north_andover @NorthAndover @NAndoverPatch @NorthAndoverHS @justice4kelseyf @MazzaMedia @lauriemullenesq @KyleSharkey8 @KYMaverick1 @monk_understudy @JusticeServedTV @KenneyBaden @reblavoie @BakerBroken @melrevision @DonutsBaga @TheEmilySee @RealNurseKim @defense_diaries @AlilawMotta @BostonGlobe @NBC10Boston @TedDanielnews

English

@HustleBitch_ Antiva needs to get approved !!! Antiva will save many lives.
English

🚨 “WHAT THE F*CK IS GOING ON?” — KATIE COURIC WARNS 17 CANCERS ARE SKYROCKETING… AND NO ONE CAN EXPLAIN WHY
Katie Couric says doctors are now seeing something they can’t ignore:
• A 21-year-old with stage 4 colorectal cancer — no family history
• Patients in their 20s, 30s, 40s being diagnosed late… already metastatic
• 17 different cancers increasing among people under 50
And even specialists are struggling to explain it.
Possible factors being discussed:
• Ultra-processed food
• Microplastics + “forever chemicals”
• Antibiotic overuse
• Environmental exposure
Couric: “It’s not just lifestyle… something is going on.”
And the most unsettling part?
Many cases are being caught too late.
So what changed?
What do you think is actually behind this spike?
English
Letitride1012 retweetledi
Letitride1012 retweetledi
Letitride1012 retweetledi

🚨Dr. Patrick Soon-Shiong - “I have focused my efforts on the Natural Killer cell... but once you have an infection or covid or chemotherapy, radiation, you wipe out these natural killer cells, which makes completely no logic”
“This confirms our greatest fears... the S protein even from the [injection] is immunosuppressive resulting in lower T cells”
English

@seanspicer @DrPatrick @ImmunityBio @TheFirstonTV @seanspicershow FDA is unfortunately controlled by big pharmaceutical companies. It needs to stop and allow good drugs to go to market
English

The Immune System's Secret Weapon Against Cancer | Ep 691 @DrPatrick @ImmunityBio @TheFirstonTV @seanspicershow
Today's show is sponsored by: Beam - shopbeam.com/SPICER to receive 40% off your order Are you tossing and turning at night and running on fumes during the day? If so, then you are missing out on the most important part of your wellness, sleep. If you want to wake up refreshed, inspired and ready to take on the day then you have to try Beam's Dream powder. This best-selling blend of Reishi, Magnesium, L-Theanine, Apigenin and Melatonin will help you fall asleep, stay asleep, and wake up refreshed. So if you're ready for the best night of sleep you ever had just head to shopbeam.com/SPICER to receive 40% off your order.
English









