
mark1eddie
87 posts







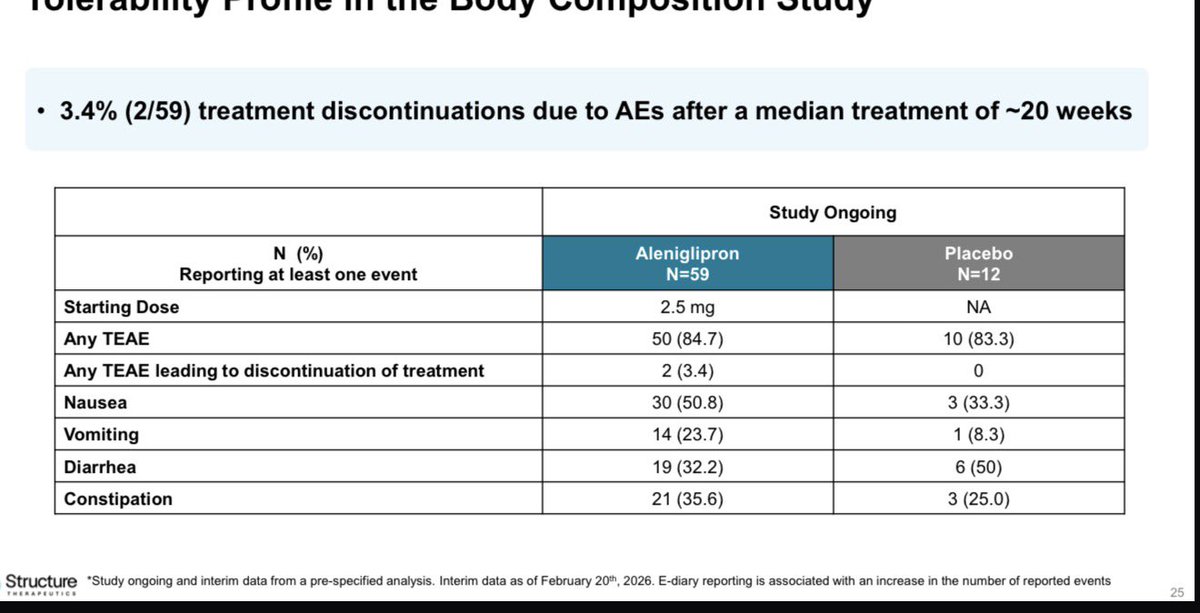

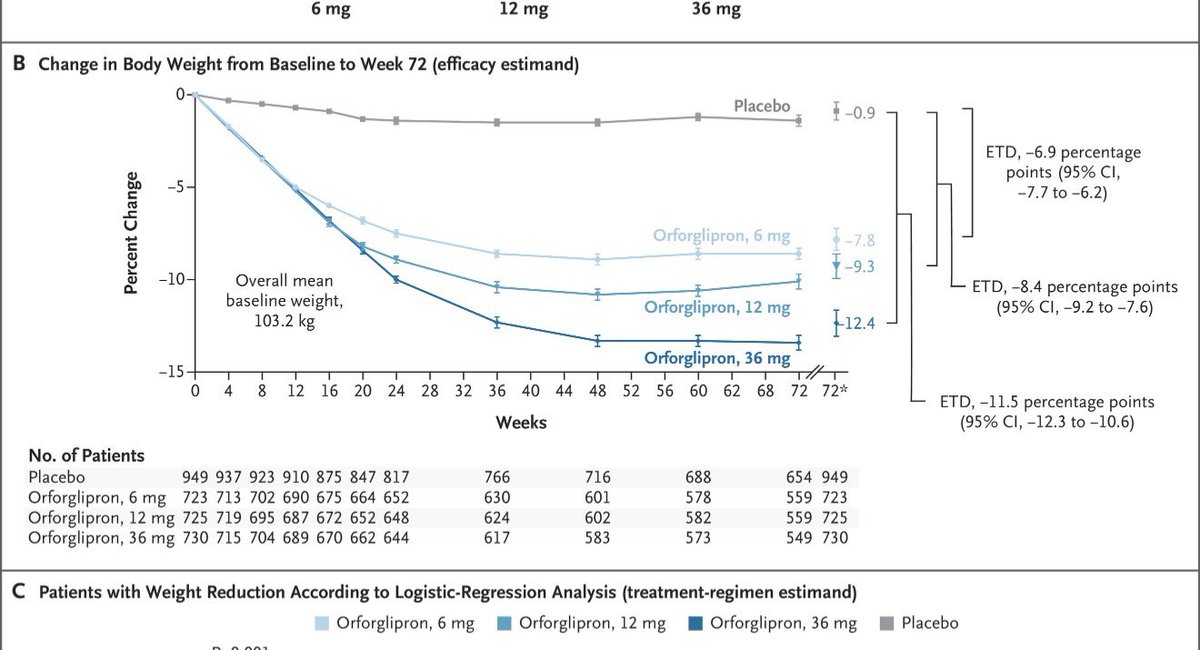
From HC wainwright on $GPCR “Our modeling and KOL feedback validate the 2.5 mg titration strategy, projecting "injectable-like" efficacy of ~17.5-19.0% in Phase 3 without the plateau seen in competitors, and with a discontinuation rate below 5%.” Not clear if projections are based on KOLs or analyst opinion but beware of the KOLs opinions in many cases in biotechs. Another example is in $SMMT lung ca OS prediction , if you exclude the top 1% lung ca real experts ( like the Emory dude ) , you see a lot of excitement among oncologists including ASCO people.

$VKTX autoinjector in phase 3 trials being implicated now & has been awhile


Laidlaw reiterated $VKTX Buy/$110: $NVO $GPCR LLY AMGN PFE Morgan Stanley⬇️the PT to $99 from $102 and reiterated at Overweight: Laidlaw said: VKTX reported 4Q25 financial results and corporate updates today after the close. Our take is that VKTX has provided clearer pathways for a more comprehensive VK2735 in obesity advancement on many fronts. Specifically, in addition to the completion and the later data readout of subQ VK2735 VANQUISH studies, oral VK2735 is expected to enter Phase III study in 2H26 and we estimate topline readout possibly in 2028. The potential to incorporate maintenance regimen(s) as a treatment option could also add more versatility for treating obesity. The addition of seasoned management on the commercial side can generate more optionality to maximize VK2735's potential for VKTX shareholders. Morgan Stanley said: Momentum continues as oral VK2735 advances straight to Ph3 (vs. Ph2b) based on FDA feedback, notably shortening time to market, while data from the Ph1 maintenance study, highlighting flexible dosing options, remains on track for 3Q26. Reiterate Overweight rating. Key Takeaways 🟩Oral VK2735 trials advancing directly into Ph3 (vs Ph2b) from Ph2a based on positive FDA feedback, with study expected to begin in 3Q26. 🟩Ph3 VANQUISH-2 study for VK2735 is nearing full enrollment, with completion expected this quarter, and continued strong interest in VK2735. 🟩Planning to introduce autoinjector in 1Q26 for all participants in VANQUISH; expected to reduce clinic visits by 75% and lower discontinuation rates. 🟩Ph1 maintenance dosing study data on track for 3Q26, with same GLP-1/GIP molecule for both routes of administration expected to support seamless transition. 🟩Ended 4Q25 with $706M in cash which fully funds the Ph3 VANQUISH studies and Ph1 maintenance study as well as the oral Ph3 studies.



$ABBV raising $8 B. Can’t be all for debt repay $VKTX vs kailera. ..













This was my post predicting $ABBV entry to obesity prior to GUBRA deal , but never heard management this aggressive with their obesity ambition as they were at JPM today. Read below X community focus now on $NVO @maziardoustdar statement about potentially massive BD .. but people under estimate ABBV will to spend particularly if $VKTX BL shares emerging blinded data from phase 3 that point to BIC tolerability and efficacy. Abbv is all about tolerability. Now ready to invest $100 B in 10 years with a lot in obesity infrastructure Can’t get more clear. They could become big contender to $LLy with right assets.



$VKTX CEO highlighted at JPM better PK profile of Vk2735 vs $LLy tirzepatide.. this consistent profile from preclinical ( mice , primates ) to phase 1-2 in humans is predictive of excellent phase 3 outcome.

