Matan flank
425 posts


An eye-catching -- and curious -- detail from @LizzyLaw_'s FDA story: Nicole Verdun, the official who was marched out of the FDA earlier this year, "may be returning from administrative leave after [an] investigation apparently found that her behavior did not merit permanent removal." I do wonder if this is a signal that not being careful about legal matters -- something that has led previous FDA officials to walk on eggshells -- could lead to a lot of FDA decisions later being walked back. It's only one data point, of course. statnews.com/2025/10/31/vin…






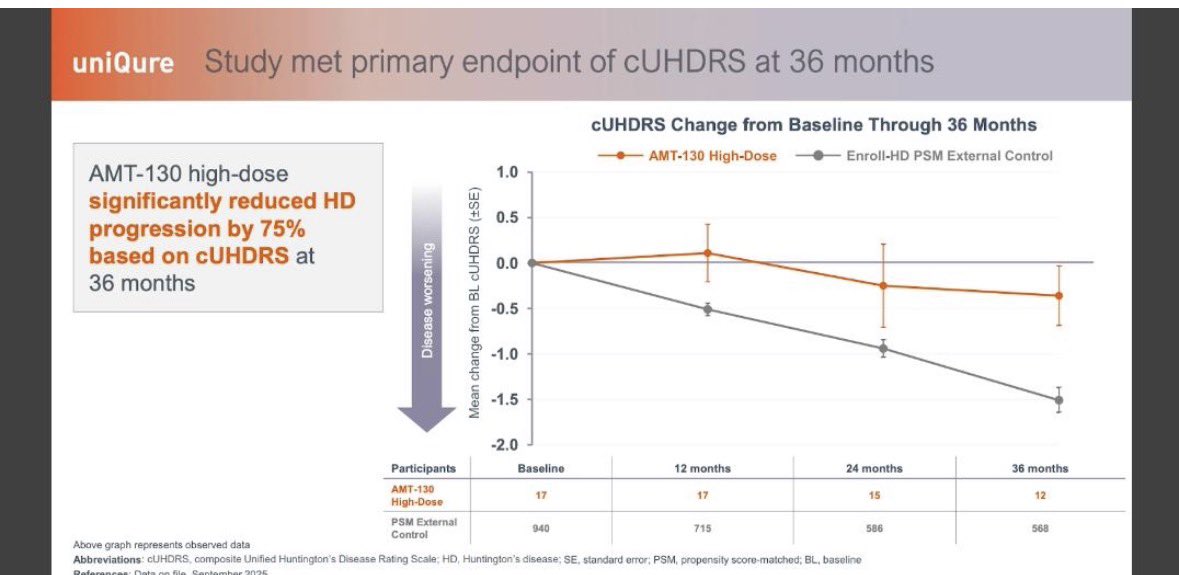
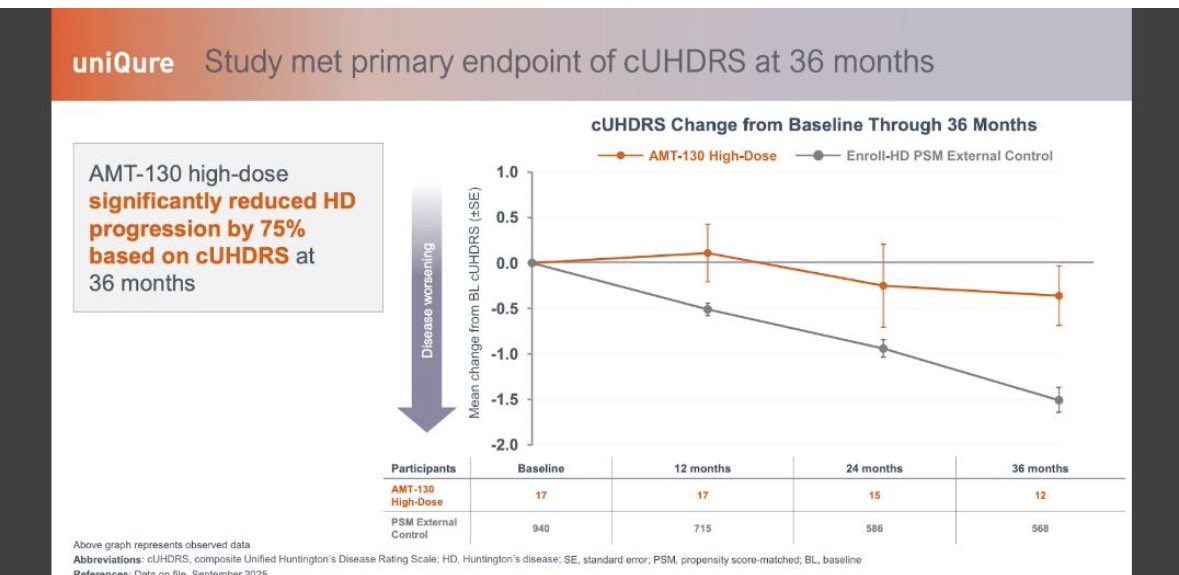
Yes and this is what I don’t get… I understand the criticisms against the data, but the moral calculus almost always seems unimaginable off and stubbornly sadistic. Like the whole safety argument is entirely speculative and goes something like well the data might be promising but it’s nowhere near certain and we can’t approve or even review this thing because we might be putting a drug that’s unsafe onto the market. There isn’t even an attempt to characterize what type of safety risks could manifest or any mention of the fact that after multiple years of treatment there have been no adverse reactions that have persisted. The moral framework should instead be looked at as something like a 50% probability of slowing disease progression that will significantly increase life expectancy and save lives vs a 50% chance of no true effect paired with a few non-life threatening adverse reactions (not even including the fact that the drug would get pulled if there was any noticeable safety flashpoints after approval). It’s just plain obtuse and morally ignorant to side with the skeptics here who are just being efficacy snobs with no consideration for moral practicality. And yes you are 100% right, time is qure’s best friend. FDA should just agree to something like filing upon the finalization of the 4 year data paired with a plan/initiation of a confirmatory trial.



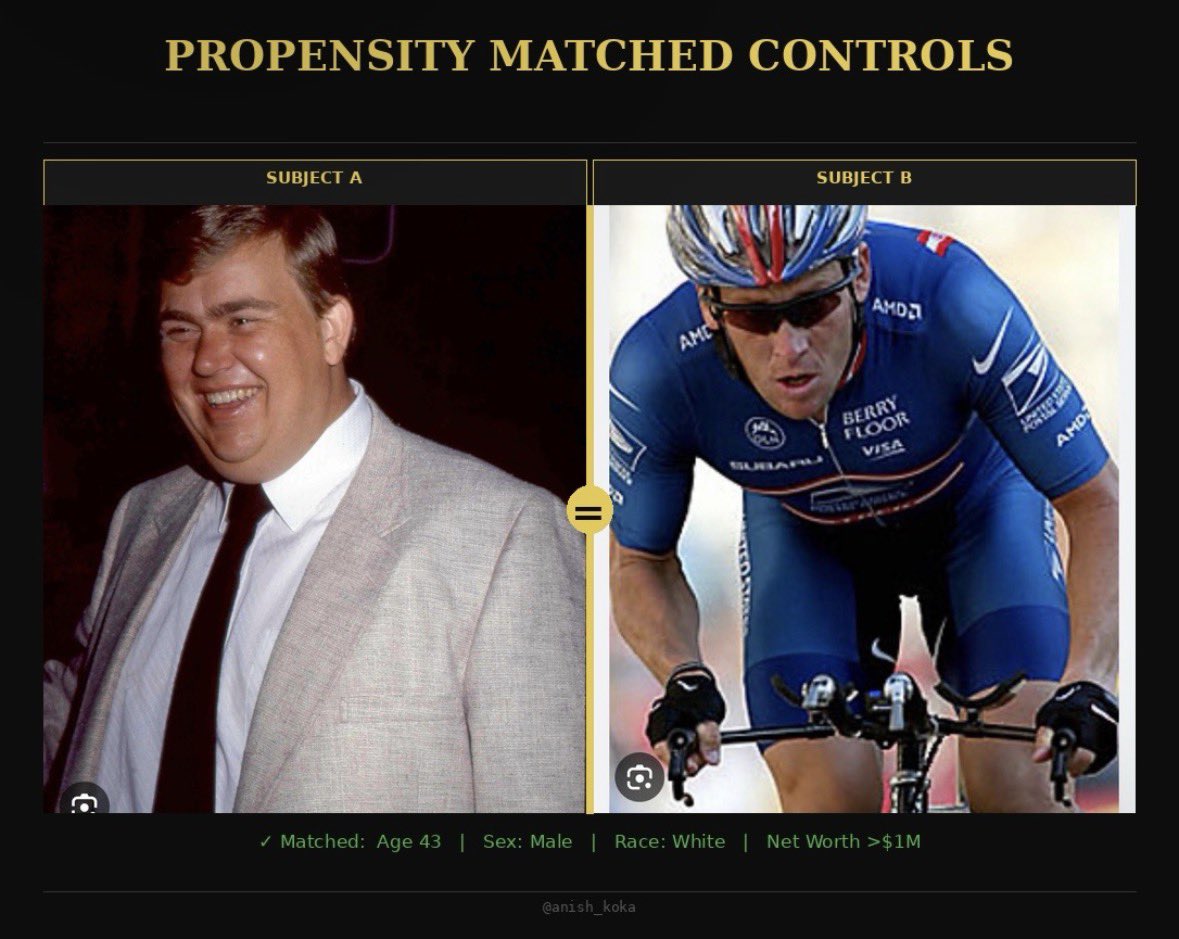
The recent apologia for former CBER Director Vinay Prasad by Dr. Anish Koka is a masterclass in narrative framing over scientific precision. $QURE 🧵




Will Vinay Prasad leave the FDA? $QURE #FDA




















