Dirk Haussecker
41.3K posts


Dirk Haussecker
@RNAiAnalyst
Wannabe biotech billionaire with a stock trading focus on nucleic acid-based and -targeted biotech. Fiercely independent. https://t.co/dfLqOuHFJP
Germany Katılım Kasım 2011
550 Takip Edilen21.4K Takipçiler

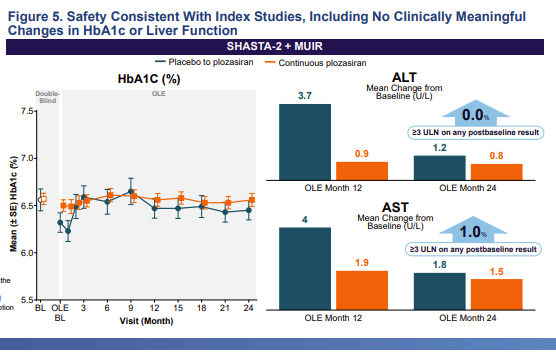
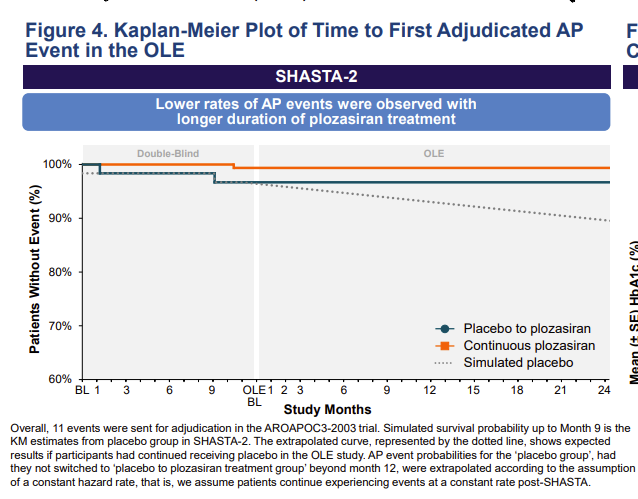
$arwr super-impressive: in the 200+ sHTG subjects treated with ApoC3 RNAi #plozasiran and followed for another 2 years in the open-label portion of study....ZERO experienced an acute pancreatitis event.
$ions
ir.arrowheadpharma.com/static-files/4…
English

@GeneInvesting It is not just the market realizing the AATD data, but also sickle cell and other data and biz dev. Of course, Trump's narcissism may have put an and to humanity by then, but go bold or go home. $beam
English

@RNAiAnalyst Wow 5x in a year is bold in this twilight zone of a biotech market. But I can definitely see 3x.
English

This would have been a great review if not for the glaring omission of the actual toxic molecular species in Huntington's: exon1a mRNA.
It is why seemingly similar knockdown approaches will strikingly differ in their efficacy:
$ptct/nvs and $ions/roche should fail (if not worsen HD by increasimg exon1a), while $qure and $alny should and are delivering on their promise.
It is the details that matter.
English

“Huntington’s Disease and The Triad of Therapeutic Conviction”. Debut blog from Eric Green, CEO of Trace Neuro, explaining why and how advances are happening in HD. Genetics, cellular understanding, and enabling modalities.
lifescivc.com/2026/03/huntin…
English

@ShahramSN @RocketPharma @CivilizationVC Congrats. Getting a drug approved for commercialization is a big achievement. Curious, but what was your financial reasoning for backing LAD-I gene therapy given its ultrarare nature?
Can it be 'passively commercialized' without it becoming a cash drain? $rckt
English

Today is a BIG day for @RocketPharma — and for my firm @CivilizationVC!!! An FDA approved drug. 🚀🧬
The FDA granted accelerated approval to Rocket gene therapy for the severe form of a rare immune disorder called leukocyte adhesion deficiency-I (LAD-I).
"LAD-I is caused by mutations in the ITGB2 gene and leads the immune system to stop working properly. Patients with severe disease face serious and potentially deadly bacterial and fungal infections. Currently, the only potential cure is stem cell transplant from a donor, but that bears its own serious risks.
The gene therapy involves extracting a young patient’s own blood stem cells and modifying them in the lab to introduce functional copies of the gene. Patients then receive conditioning to clear out their bone marrow, after which the modified cells are infused back in hopes of giving them a working immune system."
Congratulations to the Rocket team on this milestone!
FDA approves Rocket's gene therapy for ultra-rare immune disease - endpoints.news/fda-approves-r…
English

Still floored by #FDA exploring 'collaboration opportunities' with Arnold Ventures whose main healthcare objective is to reduce expenditures for rare disease drugs.
@johnarnold has not only been main financial backer of Prasad's academic work (as well as funding Makary), we now also know that he was involved in FDA staffing decisions and to this month keeps meeting with FDA leadership to influence policy.
Time to drain the swamp.

English

Shivers down the spines of the #rare disease community.
On the official calendar: Ex-Enron chief trader #Arnold Ventures continues to meet with its henchmen Makary and Prasad to give instructions.
Zach Brennan@ZacharyBrennan
Couple interesting meetings for FDA Commissioner Makary: met with Arnold Ventures on Mar. 3 about "collaboration opportunities" fda.gov/news-events/pu… and with $MNPR on "establishing a regulatory mechanism" on Mar. 13 fda.gov/news-events/pu…
English

@ABD1232121 There MUST be a sell-the-news, it's a rule in biotech. See also $dnli approval.
$rckt
English

$rckt after getting its PRV worth $150M+ after
1) much angst around Prasad and uncontrolled trials and
2) in this current environment marked by fears of #FDA delays due to staffing shortages
Stock down -7% without even having had a runup prior.
OK, now I've seen it all.
English

Catalyst scorecard:
$beam ✔️
$wve 👎 no apparent dose response a disappointment, but market clearly overreacted and tripling my share count in low to mid $5s was right decision
$srpt not much data released really, absence of splicing data raised my eyebrows; so no score here (had no position)
$rckt ✔️
Any more catalysts I'm right on and I'm broke. $pepg next.
English

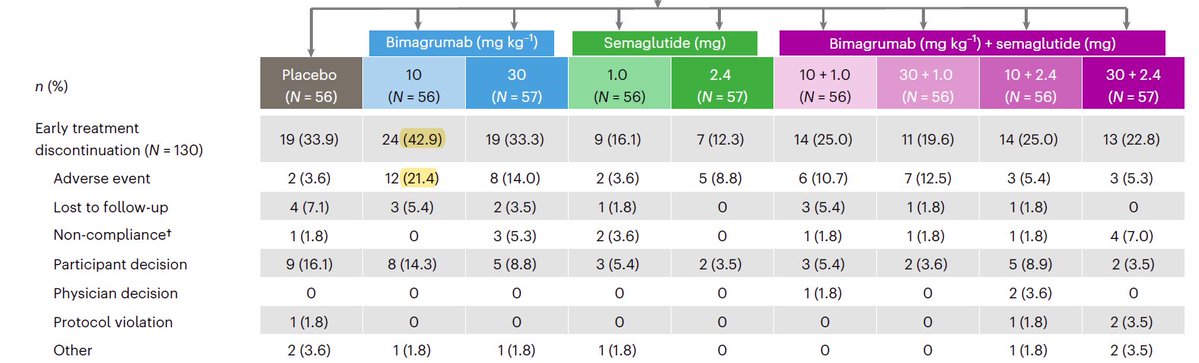
30mg/kg may be a bit more potent, but much quarterly infusions (vs annual subQ) and what about the high discontinuation rate for the 10mg/kg mono group here? What did $lly change to lower it in subsequent cohorts?
Also, $lly tested in much higher bmi and diet/exercise requirement (see pbo response).
$wve

English
Dirk Haussecker retweetledi

I returned from Beijing earlier this week, where @Pfizer has been on the ground for 36 years.
I heard from our team there about how they're using AI to fundamentally rethink how we develop and deliver medicines, not just incremental improvements but a different way of working entirely.
Globally, we intend to be the most AI-forward company in biopharma. This will take the best minds, in every market we serve, working together. That is how breakthroughs happen.

English