William J. Rapp
234 posts


@michaelkboston I was yelling to throw the ball to him on those crossing patterns all day.
English

@a_russell_2027 @frantroyan @dtroy44 @Lehighsoftball @PAChaos16UNat @Get2CollegAth @DiamondAddicts @Softball_Home @LegacyLegendsS1 @USASoftballREC Congratulations!! Your mother is so proud of you, and deservedly so.
English

Next 4🤎🤍! So beyond excited to be a Mountain Hawk! @frantroyan @dtroy44 @Lehighsoftball @PAChaos16UNat


English

@gman5180 You are a clueless idiot. I'm sure your parents are disappointed. Life is too short to follow you. BLOCK!
English
William J. Rapp retweetledi

@TheEXECUTlONER_ Representative Wheeler you look and sound like the type of representative that could have a promising career in the Republican Party.
English

A young New Hampshire Democrat representative, 22 year old Jonah Wheeler (who is black), was accused of of discrimination by fellow Democrats for standing up for women's rights and keeping transgender "women" out of their restrooms and other spaces.
His Democrat colleagues got up and walked out while he was giving his speech.
I have to give credit where credit is due, even if it is from a Democrat. That was a great speech Jonah and I agree with you 100%. Thank you for protecting our women.
English
William J. Rapp retweetledi

eagle8 $nwbo @alphavestcap
Thursday, March 20, 2025 4:31:15 AM
Lets go approval decision!
Assuming we don't have an approval before then, the upcoming 10K (before month's end) should have an interesting MD&A section.
[1]nwbio.com/northwest-biot…
[2]investorshub.advfn.com/boards/read_ms…
[3]sec.gov/ix?doc=/Archiv….
➡️Potential Post Approval PRs⬅️
I believe $NWBO is holding off on possible PRs until after approval news to have a greater impact (...assuming we survive to the approval decision 😉). In the meantime we are seeing the minimum disclosure requirements in the 10Qs.
What PRs could we see post approval?
1?Flaskworks
2?Reimbursement of DCVax-L
3?Agreements with Private Clinics
4?Combo Trial Collaboration
5?Immune Booster Agent Collaboration
6?DCVax-Direct Restart
7?Roswell Park Expansion of Scope
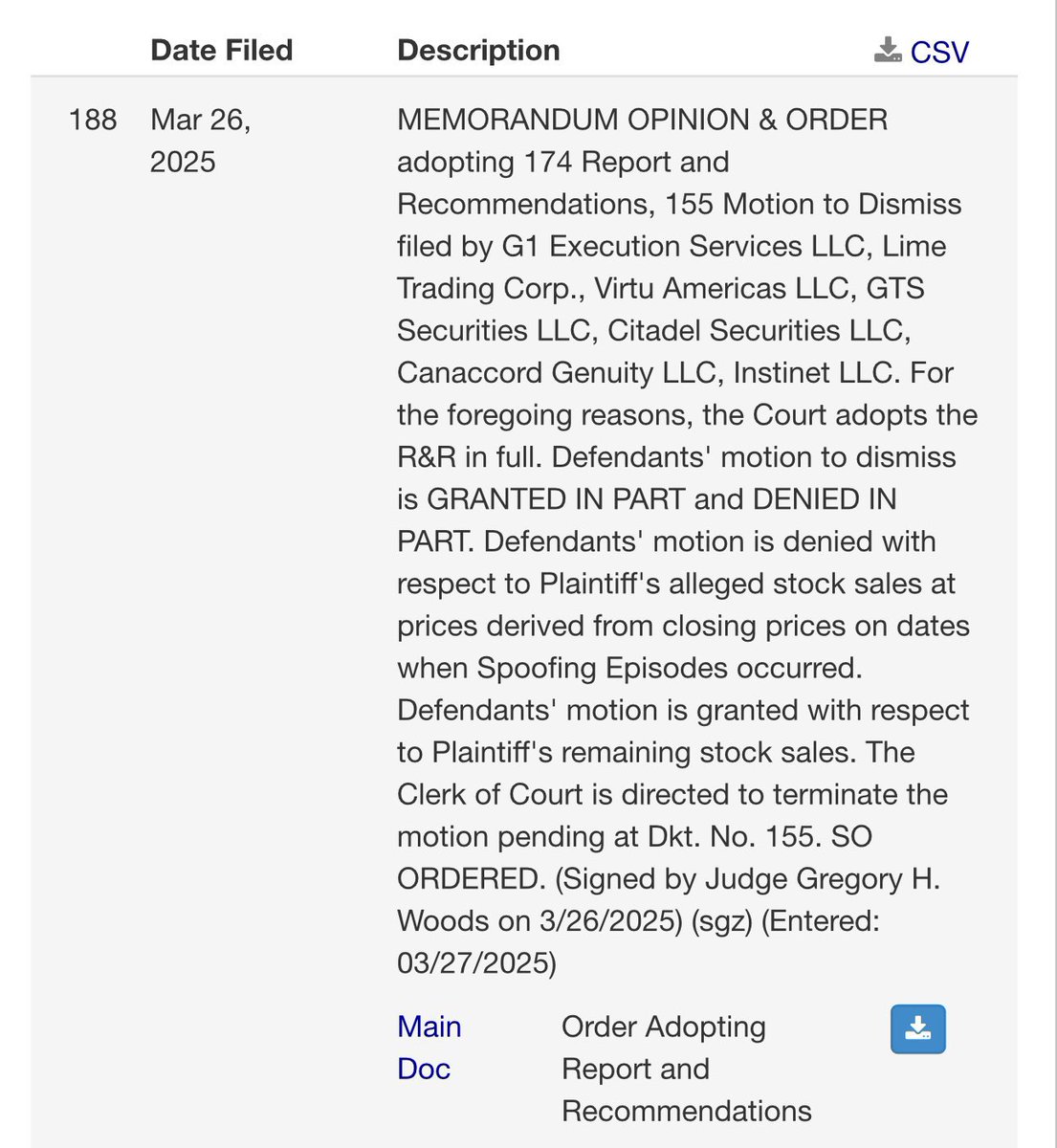
8?Judge Woods MTD Decision*
Some more detail as to each possible news item.
1?Flaskworks
February 6, 2024:
“The next step is to have GMP-compliant (i.e., clinical grade) versions of the prototype Flaskworks machine made with GMP grade materials. A specialized contractor has been engaged to do so. This process is expected to take several months…” [1]
“As soon as the GMP-grade units are delivered, Advent BioServices will undertake final qualification and validation of the GMP units and use engineering runs with the system to collect data in order to submit an application to regulators to approve the use of the system for production of DCVax-L for patients.” [1]
Basically, $NWBO issued a PR saying they picked their final version of Flaskworks and are ordering GMP compliant parts to then run validation tests before ultimately applying for Flaskworks approval with the MHRA.
The next update we see is the June 29, 2024 ASM where Linda Powers states:
“So that adaptation work for GMP was recently finished. There are a couple of improvements that we are doing to streamline, and then those units are about to be ordered shortly. So that's the that's the clinical grade machines.” [2]
I speculate that $NWBO is now doing validation runs of Flaskworks in anticipation of submission for approval. I believe $NWBO files for a variation on the hopefully soon to be approved DCVax-L MAA.
Linda Powers believes that Sawston, with fully built out lower grade clean rooms and an approved flaskworks device, could have an annual capacity of 15,000 personalized vaccines. [2]
2?Reimbursement of DCVax-L
June 29, 2024 ASM:
"What we will need to do is, we'll need to engage specialized consultants to develop, what's referred to, as a health economics model. We have to make an economic model about the cost benefits of the DC vaccine treatment, and how it fits with their policies, and that sort of thing. So, that for sure will be, in our grouping of top priority activities, over the, as we look forward, over the coming 18 month period, 12 months, whatever." [2]
November 12, 2024 10Q:
“The Company has evaluated and selected specialized, highly experienced consultants with whom to undertake preparations for a potential reimbursement review and evaluation process. The Company has executed the relevant contracts and is under way with the first stage of the process with the consultants." [3]
$NWBO stated that they will need to engage consultants to prepare an "economic model" to submit with NICE to determine the reimbursement for DCVax-L. A few months later they had apparently only just recently engaged said consultant. I believe $NWBO is not close to a NICE reimbursement recommendation. Which brings me back to my first point, a Flaskworks variation submitted post MAA approval. I believe $NWBO plans to get flaskworks approved and then submit their economic model to NICE so that the lower prices via automated manufacturing helps $NWBO obtain reimbursement approval of DCVax-L. These updates could closely follow an approval decision PR.
3?Agreements with Private Clinics
November 12, 2024:
“These include joint efforts by the Company and Advent BioServices to establish collaborations with several private clinics in the U.K. The collaborations with private clinics are designed to achieve several goals. One goal is to establish dedicated leukapheresis units which will be available for NWBio patients at all times. This is quite important for scale-up of the numbers of patients who can be handled for NWBio’s treatments, because there is a country-wide shortage of leukapheresis capacity in the U.K.” [3]
It appears $NWBO, as of Nov. 12th, had agreements, or were close to agreements, with three clinics. These clinics will apparently be used for compassionate use patients, the scale up of DCVax-L and future trials.
One clinic that has been following a lot of $NWBO shareholders lately is x.com/ClinicNeurology
4?Combo Trial Collaboration
$NWBO appears to be collaborating with a company that has a "complementary immunotherapy agent".
IMO, this would likely be a big pharma with a checkpoint inhibitor based upon the encouraging data with DCVax-L + $MRK's Keytruda in rGBM.
November 12, 2024:
“The Company completed negotiations and entered into a contract with a company that has a complementary immunotherapy agent, and the two companies have been engaged for a number of months in joint development of trial designs and selection of candidate cancers for a combination treatment clinical trial. The companies are particularly focusing on trial designs in which the primary endpoint will be tumor response (shrinkage), with potential timeframes in months, rather than time-to-event survival outcomes, with timeframes in years. The Company anticipates potentially being ready to submit an IND for such a combination trial to regulators during Q1 of next year.” [3]
5?Immune Booster Agent Collaboration
November 12, 2024:
“The Company has also completed negotiations and entered into a contract with a company that has an immune booster agent that may have the potential to further increase the potency of DCVax products. The contract provides for material supply and in vitro testing of the booster agent with DCVax products. If the in vitro testing is successful, the parties plan to enter into a license of the booster agent to NWBio. The parties jointly designed the program of experiments, and the experiments were carried out and completed by the Flaskworks team. The parties are now evaluating the results and developing plans for further experiments.” [3]
IMO, think Oncovir, the maker of Poly-ICLC.
6?DCVax-Direct Restart
“The Company currently anticipates that the DCVax-Direct program will be able to restart by the end of January. For the same reasons as described above in connection with the planned combination treatment trial, the Company plans to focus DCVax-Direct clinical trial designs on tumor response endpoints with potential timeframes of months rather than survival endpoints with timepoints of years.” [3]
This seems a little further out than the combination trial listed in #5 above as that trial gives a date for filing an IND whereas this trial does not.
7?Roswell Park Expansion of Scope
November 12, 2024:
"These Phase 2 trials are ongoing and one of them has been approved for a significant expansion in scope by the grant funding agency…The expansion of the trial scope that has been approved is to add refractory lung, breast and bladder cancers to the trial in addition to refractory melanoma." [3]
8?Judge Woods MTD Decision
*This isn't dependent on the approval decision, but with how things are playing out, we might see Judge Wood's decision on the MTD after the MHRA's approval decision.
For more information on the MTD:
English

@Patriotmom717 Yes! And every other federal spending cut recommendation from Musk.
English
William J. Rapp retweetledi

TRUTH 💣 💣‼️ What’s going on transcends Zelenskyy, Ukraine, Putin—it's bigger than all that. The Democratic Party has masterfully brainwashed a chunk of America. They’ve conditioned people to despise Trump, patriotism, anything pro-America and everything that makes sense. They’ve opposed progress, development, anything Trump touched, and trained their followers to hate it too. If Trump cured cancer, these blue-haired women, bald-headed men with piercings would rally for cancer, all screaming for cancer just to spite him. It’s insane, but it’s our reality. And because he’s a Black man who dare call it out, he gets labeled every name in the book when he is just a patriot who hates watching our country crumble. He said he doesn’t care, call him what you will, just don’t call him a Liberal.
English