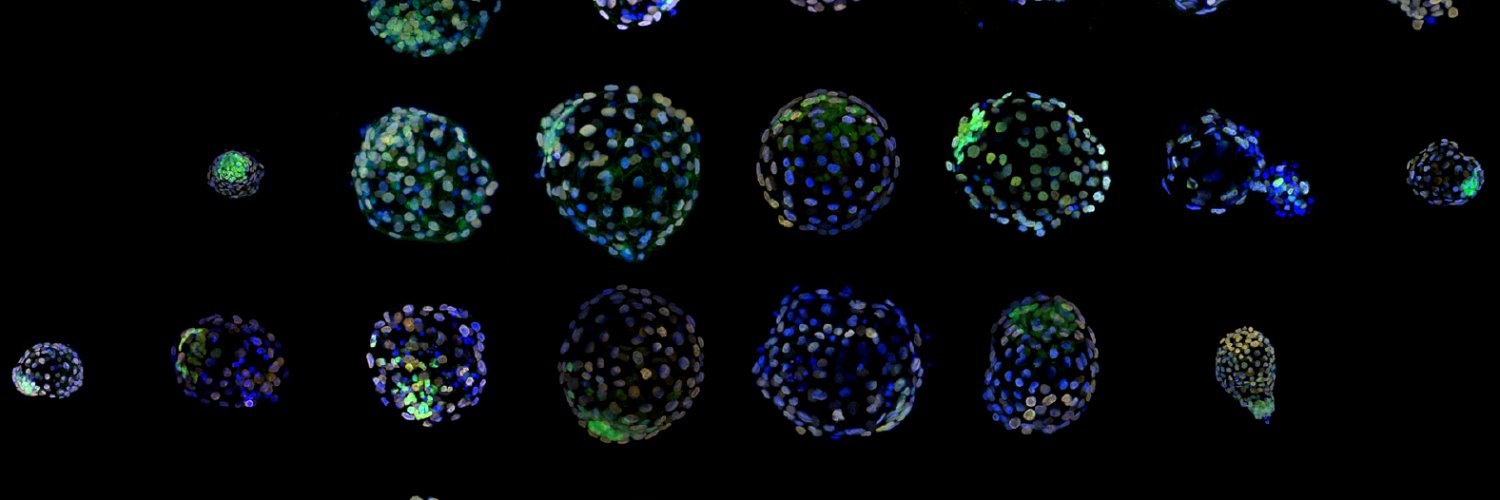
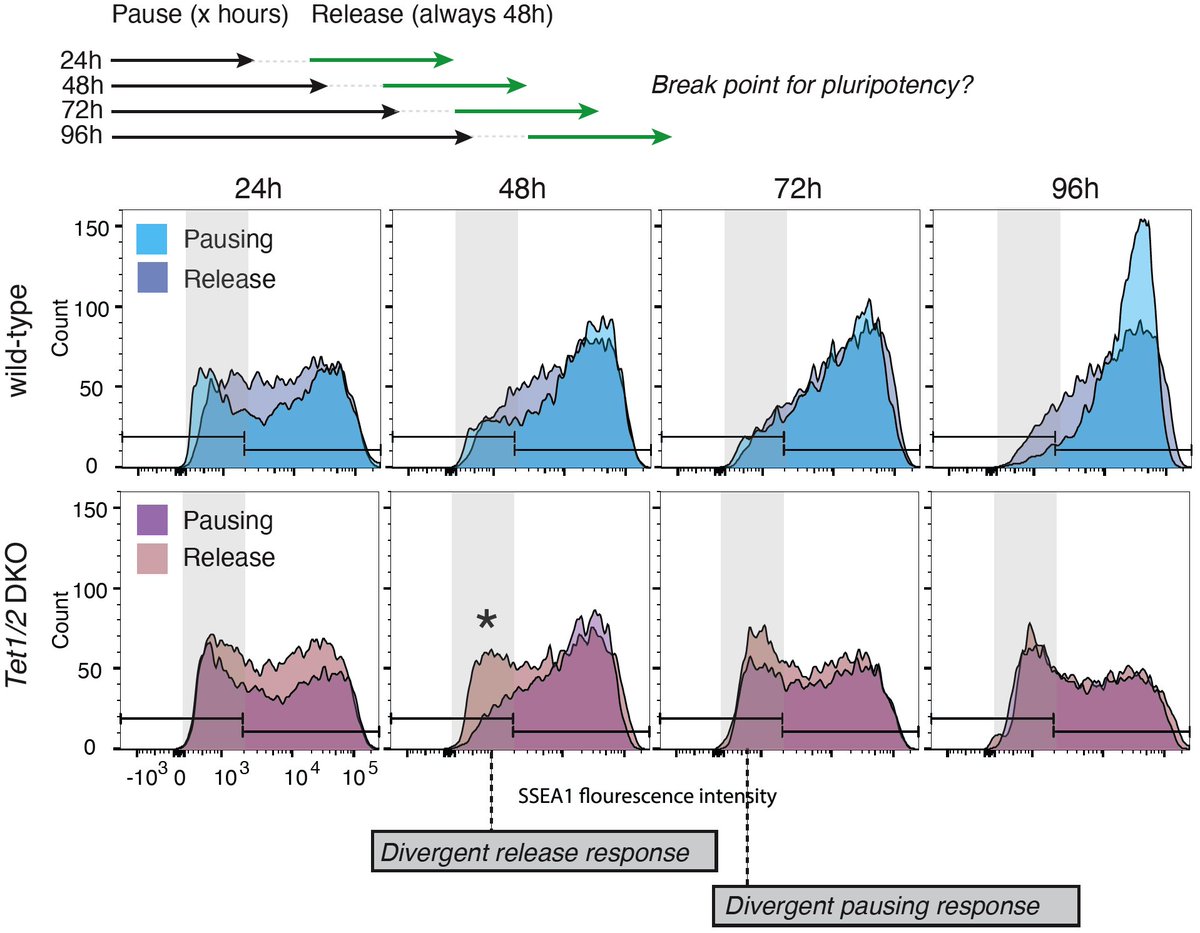
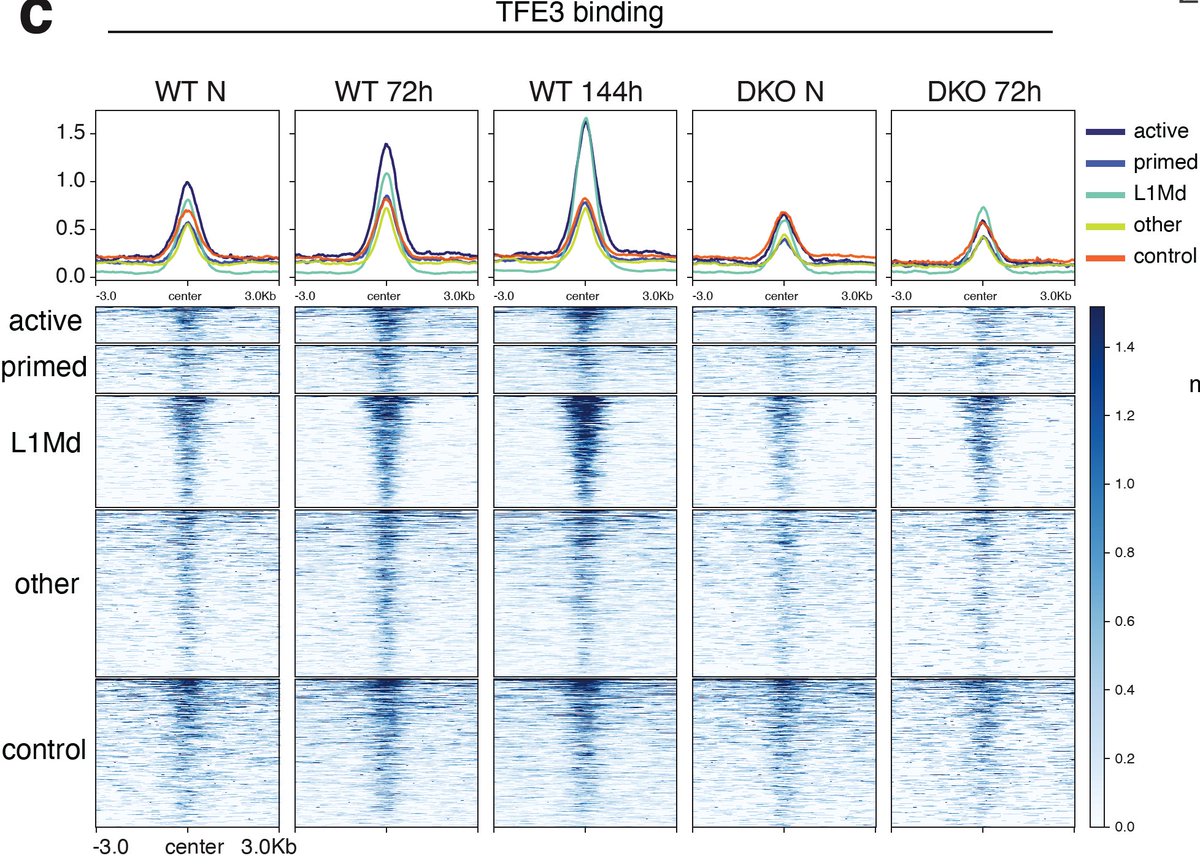
ทวีตที่ปักหมุด

How do embryos and ESCs safeguard pluripotency throughout a state of dormancy? A great collaborative effort of the @bulutkarslioglu -lab & the
@MPI_MolGen involving @helenekretzmer @AbhiSampKumar @_taktas_ @VvdWeijden & many more.
nature.com/articles/s4159…
English