ทวีตที่ปักหมุด
Avi Roy
7K posts


Avi Roy
@agingroy
Biomed Scientist in #longevity. Mentor & Investor in Health startups. Past CTO @BioVivaScience ,Researcher @CASMIORG ,Lead @oxfordscisoc @NewsInLongevity ,BGRF
Oxford, England เข้าร่วม Mayıs 2008
2.7K กำลังติดตาม3.9K ผู้ติดตาม

Sources:
@EurostatOffice LE release: ec.europa.eu/eurostat/web/p…
Eurostat excess mortality: ec.europa.eu/eurostat/stati…
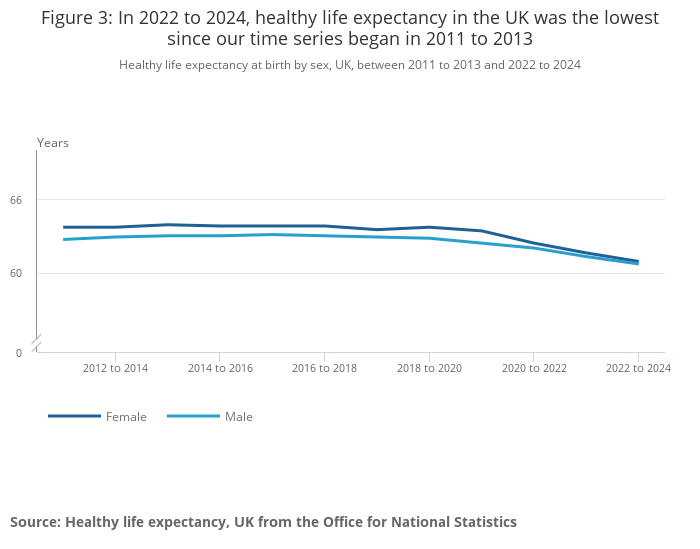
@ONS HLE release: gov.uk/government/sta…
The ONS data covers 2011-2013 through 2022-2024. Eurostat is also recalculating its older-age methodology (extending from 85+ to 95+ by single year), so historical baselines for the oldest cohorts will shift.
English

Laurus Labs is an API manufacturer, mostly for generics, including semaglutide copies launching in India now.
Retatrutide is Eli Lilly's proprietary molecule, still in Phase 3, not yet licensed to Indian manufacturers. The supply chain for next-gen GLP-1s like retatrutide is separate from the generic semaglutide story.
Two very different pipelines.
English

@agingroy @jaganmsna Isn’t Laurus a supplier to retatrutide?
English

Ozempic costs $936/month in the US. As of tomorrow in India, it'll cost $40.
The Ozempic patent expired this week in India. 50+ Indian generic makers are launching copies. India already supplies 20% of the world's generic drugs and just did the same thing to weight-loss medication that it did to HIV drugs two decades ago: made them affordable for billions.
Patents also fell in China (17+ generics in Phase 3), Brazil, and Canada. Combined, that's 40% of the world's population.
Meanwhile, @EliLillyandCo just dropped results for what comes next. Retatrutide hits three gut hormones instead of one.
In Phase 3, patients lost 28.7% of body weight (71 lbs average), the biggest weight loss ever recorded in a late-stage trial. A1C dropped 2% in a diabetes cohort.
The old guard goes cheap. The next generation goes bigger. 890 million adults with obesity are watching.

English

You're right that NVO's revenue exposure to India/Brazil is minimal. That's the whole point. A drug priced at $936/month in a country with median income of ~$2,500/year isn't a real market. It's a missed opportunity for public health.
The patent expiry probably doesn't hurt @novonordisk's earnings much. It opens access for hundreds of millions of people who were priced out entirely. Those two things aren't in conflict.
English

You're flagging the part of this story that I worry about too. The 39% lean mass loss figure from STEP 1 is real and doesn't get enough airtime.
Plastic surgeons and physiatrists are going to be busy. What I'd want to see in the generic era: mandatory resistance training protocols alongside prescription, not as optional lifestyle advice.
Right now that's not happening at scale. The access boom makes this more urgent, not less. What are you seeing in your clinic already?
English
@agingroy Many people aren’t discussing the day after . Ozempic face, massive abdominal pannus, profound muscle weakness . Benefit outweigh the risk but it is a large cohort of patient awaiting us.
English

You're right that the sequencing isn't accidental. But I'd push back slightly on the framing.
Retatrutide took 8+ years of trials to get to Phase 3, and the biology of triple agonism is genuinely harder than GLP-1 alone. It's not purely a timing play. The patent cliff on semaglutide was always coming.
What India did differently was refuse to let evergreening block it. Section 3(d) of their Patents Act is the part of this story that deserves more attention.
English

The Pharma industry always times this to perfection.
Like clock work, when the old drug is coming off patent, they will have the next drug take it's place.
The reality is that they could have developed both drugs at the same time. They purposely don't do that and instead make sure they time it when the other goes off patent!!
English

The 3m figure is astounding, and the mechanisms work.
GLP-1s reduce CV events (SELECT: 20% relative risk reduction, n=17,604), likely reduce cancer incidence, and address obesity-driven mortality across multiple pathways.
If India's generics make this a $14-40/month drug globally, we're not talking about a weight-loss story anymore. We're talking about one of the biggest shifts in preventable mortality in a generation.
English

@agingroy The key issue here is potential to save ~ 3m lives per year globally medrxiv.org/content/10.110…
English

@EliLillyandCo is in a strong position. The SURMOUNT-5 head-to-head trial showed tirzepatide outperformed semaglutide 2.4mg on weight loss: ~20% vs ~14% body weight reduction. But Indian generics are now entering both molecules.
Lilly's tirzepatide patents expire in India on a similar timeline. The clinical winner and the access winner may not be the same. In a price-sensitive market, $14/month beats superior efficacy for most patients.
English

@agingroy Interesting. Hasn’t eli Lilly already started their marketing their obesity portfolio ? Clinical analysis & prescriptions will drive who wins the race, because consumers will take everything they believe/ afford.
English

Exactly right. The STEP 1 trial extension showed ~two thirds of weight lost returned within a year of stopping semaglutide. The drug suppresses appetite but doesn't rewire hunger biology permanently.
What you're describing, building genuine behavioral infrastructure while on the drug, is the only thing we know reduces that rebound.
GLP-1s as a bridge, not a destination. That framing needs to be standard clinical practice.
English

@agingroy What actually matters long term isn’t just access, it’s how people cycle and exit. Using GLP‑1s as a bridge to normalize appetite while simultaneously training hunger cues, walking, and strength makes rebound weight gain far less likely once the script stops.
English

The subsidiary verification approach is smart. For semaglutide specifically, look for products from manufacturers with US FDA or EMA-approved facilities. Many Indian generics manufacturers (Sun Pharma, Cipla, Dr. Reddy's) already supply Western markets and meet those standards.
The risk isn't Indian generics broadly. It's unregulated compounders and gray market vendors with no oversight.
English

@agingroy Now along with Canadian & Mexican comes the turn of Indian Online Pharmacy for 🇺🇸s😆😆😆Luckily wheneverI need something like that I go to hub where I can see if the product is made by a subsidiary of the original drug maker like Abbot eg🤷
English

Those programs exist, but they're not easy to access. Income limits, paperwork, re-enrollment hurdles. The people who need it most are often least equipped to navigate it. And introductory pricing that jumps to $936 isn't access, it's a funnel. India's generic market just solved this with competition instead.
English

@agingroy Self-pay options through Novo Nordisk or programs offer lower direct prices (e.g., $349–$499/month depending on dose, or introductory $199 for new patients on lower doses).
English

@2000_vina Wow! NATCO's ₹1290/month works out to ~$14-15 at current rates. If that's right, the number I cited may reflect a different dose or early projections. Prices across 50+ generics will vary. But whether it's $14 or $40, the gap with the US $936 list price is the real story here.
English

Fair point on list vs. net price. But "most people don't pay it" only holds if you have good insurance. The 25 million uninsured Americans do pay something close to list. And even insured patients often face high copays on branded GLP-1s. The list price isn't irrelevant. It anchors everything downstream.
English

@agingroy The price you state is a half truth. It's the list price which most people don't pay.
English

ITP studies actually run for the full lifespan of UM-HET3 mice, roughly 2-3 years, which is their equivalent of a human 80+ year life. Treatment often starts at 4 months, 12 months, or 20 months specifically to test different life stages. That's not short.
It's one of the longest-running standardized aging trials in biology. The multi-site design is what makes it credible, not a waste.
English

@agingroy The ITP test has too short a longevity period and requires at least three years. The ITP should be abolished as it is a waste of money and authoritarian.
English

At 79 and still in the game, you've run the longest personal experiment here. The healthspan point is right, and the article probably underplayed it. Lifespan is the metric ITP can measure.
Healthy aging is what most of us actually want. Those aren't the same thing. I'd bet your decades of consistent habits, not luck, explain most of your trajectory. That's what the super-ager data shows too: consistency compounds.
English

As a supplement guy since the late 1970s, you are missing the point here. Some of us are more into healthy aging than extended lifespan. What is the point of living longer if you aren’t healthy. At 79 I have one more year to qualify as a super ager. Was it all that exercise, my unique diet, luck/randomness or maybe that supplement regimen over the decades.
English

That tracks with what we see, and I'm glad you caught it. Liver enzyme elevation on rapamycin is real, usually reversible, but definitely something your prescribing doctor needs to know about now.
Did it normalize after stopping? Either way, this is exactly why lab monitoring before and during is non-negotiable.
English

We spent weeks building the most complete public database of every longevity drug ever tested by the @NIHAging's Interventions Testing Program.
54 compounds. 30,000+ mice. 20 years of data. 3 independent sites.
Seven drugs extended life. Most popular supplements failed completely.
Here’s the ranked data, the mechanisms, and the database:
Avi Roy@agingroy
English

Lifestyle is the foundation, I don't argue with any of that. But the ITP mice aren't sedentary junk food addicts: they're on standardized diet with normal activity. The drugs extend life on top of that baseline.
Rapamycin started at the equivalent of human age 60 still added roughly 10% more lifespan. That's not replacing sleep or exercise. That's a separate mechanism.
The question isn't lifestyle vs. drugs, it's whether you want both working for you.
English

Partly yes, and the field knows it. Acarbose and canagliflozin both work through glucose metabolism, so they might be correcting a sugar-heavy chow effect.
But rapamycin's mTOR pathway works in calorie-restricted animals too, and glycine's benefit is probably independent of diet composition.
The ITP uses identical chow across all sites specifically to control for this.
The honest answer: diet interacts with these drugs, and we don't fully know the human equivalent.
English

Tetracyclines weren't in the ITP database we built, but that's not necessarily an NIH decision.
The ITP tests nominated compounds: researchers submit candidates, the committee evaluates them. If tetracyclines weren't tested, someone didn't nominate them, or the proposal wasn't selected.
The mitochondrial angle is interesting though. Do you have a published lifespan study you'd nominate? Genuinely curious.
English