jason ryals
352 posts







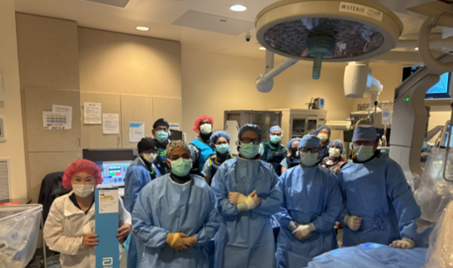


Proud to share that we completed our first and some of the first cases in the nation post fda approval of the Abbott VOLT Pulsed Field Ablation (PFA) system at St. Bernards Medical Center in Jonesboro, Arkansas as part of the #VOLT LMR, following VOLT’s FDA approval in late December. What stood out in our early experience: - Zero fluoroscopy workflow in these cases - PVI, with additional workflow as clinically needed - Very low microbubble visualization on ICE, and no clinical hemolysis observed in any of our cases We also appreciate the platform design elements that support efficiency and precision: - Balloon-in-basket design (8 splines) that conforms to anatomy and supports targeted lesion sets - Real-time contact visualization (impedance-based) for lesion tracking - Single-catheter , zero exchange workflow enabling mapping, pacing, and ablation while minimizing catheter exchanges - The ability to customize energy delivery by selectively delivering from electrodes in good contact Our cases were performed under general anesthesia, and it’s encouraging that PFA workflow characteristics may support future pathways that reduce reliance on GA in selected patients. Huge thanks to our incredible St. Bernards EP Lab team and the outstanding clinical support team from @AbbottCardio. Teamwork made this LMR launch a success.




















