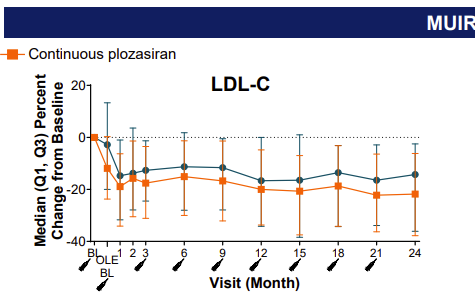
Is this a compound-specific or a class effect of the therapy?” he wondered. In the SHASTA-2 study of patients with severe hypertriglyceridemia, plozasiran (Arrowhead Pharmaceuticals), a small interfering RNA designed to reduce APOC3 in the liver, did not result in a significant increase in hepatic fat, he noted
English