

David Filmore
2.7K posts


@Filmoreorless
Really only On Here now for #Knicks and #NBA twitter





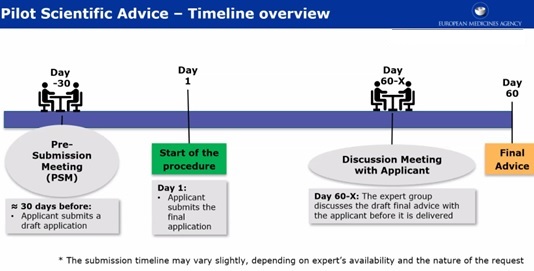
In this month’s spotlight from Pathways’ Document Depot: EU organizations, including the European Commission and TEAM-NB, rolled out resources and arguments engaging with the recently extended Medical Device #Regulation transition: bit.ly/3Rswi83 #medtech




@US_FDA @JAMA_current @CesarCaraballoC @jsross119 @hmkyale @JAMAInternalMed (9/15) Lastly, we matched recalled predicates to contemporaneously authorized recall-free controls, and compared recall rates among descendants. The risk of a Class I recall was increased *sixfold* for devices that used a recalled predicate.






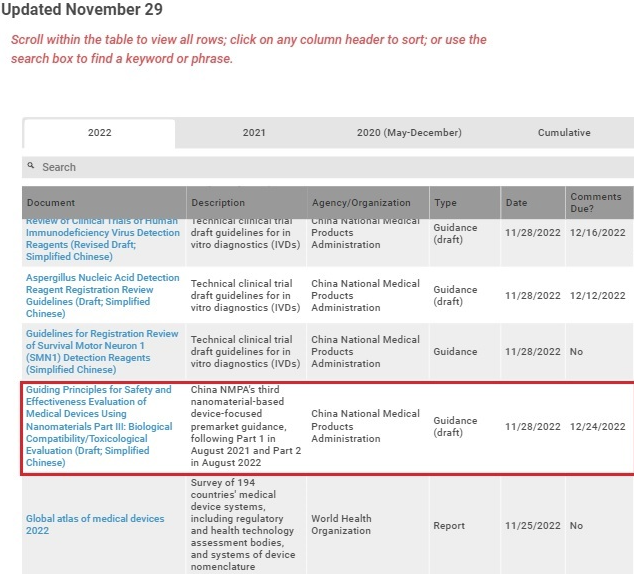




#Regulatory Doc of the Week: “Developing and Responding to Deficiencies in Accordance with the Least Burdensome Provisions: Guidance for Industry and #FDA Staff” @US_FDA, October 26, 2022 Here are 3 reasons why this matters. 🧵⬇️








