
Prof Peter Collignon
36.6K posts

@CollignonPeter
Infectious Diseases Physician and Microbiologist. Professor Medical School. Australian National University. Views are my own.



This is sad but unsurprising. Antipsychotic drugs were created in the 1950s not to heal, but to manage unruly patients. They were called a “chemical lobotomy”. That logic persists today.

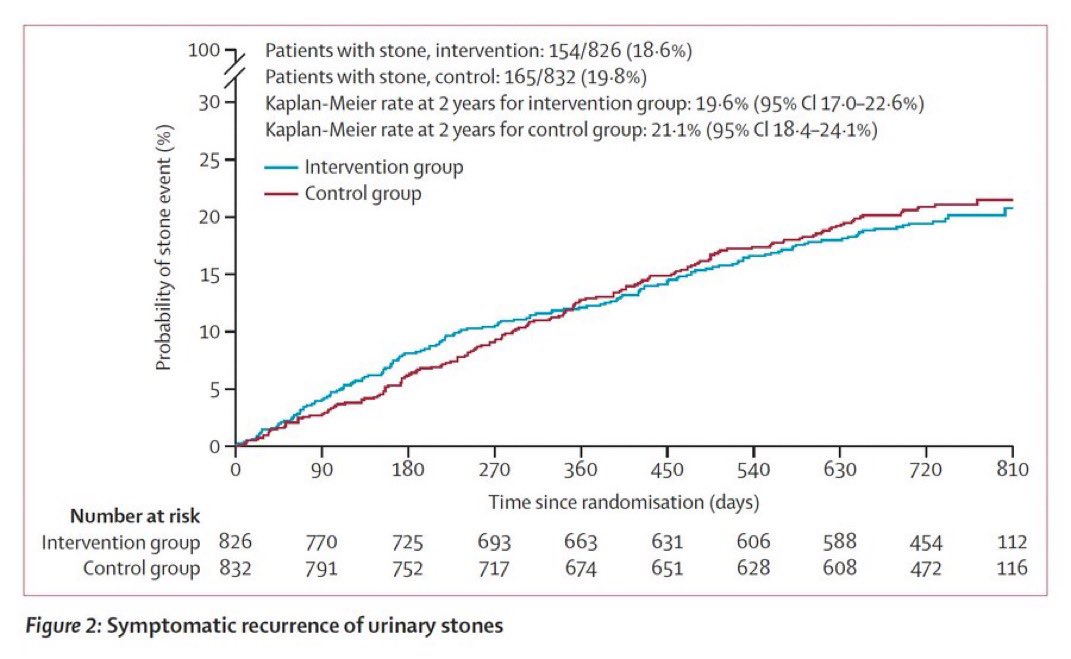
Evidence from randomized trials suggests that calcium supplements may not significantly reduce fracture risk in well-nourished adults. mdsc.pe/4bAVwuK

@PatMcGorry That’s generalisation. Not everyone responds to or benefits from meds. Practitioners assume everyone does & force them on everyone. Programs that help people recover without meds don’t get attended to. My journey trying to find my way to stay out of madness was hampered by this


The NYT is out with an analysis that TrumpRx does not have better prices than Europe. That's only part of the story - TrumpRx does not have better prices than elsewhere in the U.S.! nytimes.com/2026/03/18/wor…



🚨MAJOR NEW PAPER 🚨 just out in @JAMAPsych : Psychedelic Therapy vs Antidepressants for the Treatment of Depression Under Equal Unblinding Conditions (tinyurl.com/yu2rbtaf). I am very proud of this one, was a lot of work for me - both co-first and last author! Eternal gratitude to co-first @QuantPsychiatry and twitterless Hannah Barnett! The premise is that it is biased to compare open-label trials (=where patients know what treatment they are getting) to blind trials (=where patients do NOT know what they are getting). Open-label trials would gain an unfair advantage by higher placebo response. Even formally blinded psychedelic trials are practically open-label as its obvious to distinguish placebo from 25mg of #psilocybin. In contrast, traditional antidepressants (SSRIs/SNRIs) trials are are close to be truly blind (Lin 2022). Given the bias of open-label vs. blinded comparison, we compared the efficacy of psychedelic-therapy (which is practically always open-label) vs. open-label antidepressants for the treatment of major depression. We tested 3 prior hypothesis: - There will be a significant difference between psychedelic-therapy vs. open-label antidepressants, favoring psychedelic-therapy. - There will be a significant difference between blinded and open-label antidepressants trials, favoring open-label. - There will NOT be a significant difference between blinded and open-label psychedelic-therapy, as practically they are always open-label. In contrast with our prior hypothesis, we did not find psychedelic-therapy to be more effective than open-label antidepressants (H1). Not only was the difference not clinically meaningful, but practically there was no difference at all. This finding means that antidepressants administered knowingly to patients, which is the case in real-life medical practice, is as effective as psychedelic-therapy. This result was robust across variations in study selection, including when we removed psychedelic-therapy trials on treatment-resistant depression. We also assessed the impact of blinding in both psychedelic-therapy and antidepressants trials. We found that for antidepressants (H2), but not for psychedelic-therapy (H3), open label is associated with better outcomes than blinded treatment. However, even in the case of antidepressants, the difference was practically small (~1.3 HAMD units). How come hypothesis 1 failed, i.e. that psychedelic-therapy is no ore effective than open-label antidepressants, given that antidepressants trials are famous for small drug-placebo difference (~2.4 HAMD units), while psychedelic-therapy trials reported large effects (~7.3)? The key factor is that in psychedelic trials the placebo response is about 50% relative to antidepressants, ~ 4 vs 8 HAMD units (Hsu 2024, Hieronymus 2025). This suppressed placebo response leads to an inflated between-arm difference, as the treatment arm is measured against a lower floor. The suppressed placebo response in psychedelic-therapy trials is likely attributable to the ‘know-cebo’ effect, i.e. the disappointment when patients realize they are in the control group. In psychedelic-therapy trials, this placebo suppression accounts for 4.0 / 7.3 ~ 55% of the specific treatment effect. In other words, ~55% of psychedelic-therapy’s effect is not explained by patient improvement after the treatment, but rather by the lack of improvement in the placebo group. In summary, we found that for the treatment of depression, psychedelic-therapy is no more effective than open-label SSRIs/SNRIs. Our results for psychedelics are twofold: psychedelic-therapy demonstrated a robust and large therapeutic effects (~12 HAMD units), which justifies optimism. On the other hand, psychedelic-therapy’s lack of superiority compared to open-label SSRIs/SNRIs highlights the influence of blinding integrity and argues against overly optimistic narrative's about psychedelic-therapy's potential.


Are psychedelics better than antidepressants? New study says no eurekalert.org/news-releases/… jamanetwork.com/journals/jamap… With an innovative approach, scientists try to get around the problem of participant expectation in tests of psychedelics. Psychedelic-assisted therapy may be no more effective than traditional antidepressants when patients know what drugs they are actually taking, according to a first-of-its kind analysis that compared how well each type of drug worked for major depression. Psychedelic-assisted therapy has resisted placebo-controlled testing methods — the gold standard in clinical trial design. Due to their powerful subjective effects, nearly everyone in the trial knows whether they received a psychedelic or the placebo even if they are not told. But in trials of antidepressants, participants may not figure out whether they have received the drug or a placebo, which makes it hard to compare them with psychedelics. To get around this problem, researchers from UC San Francisco, UCLA, and Imperial College, London tried a different approach. They compared the results from psychedelic therapy trials to the results from so-called open-label trials of traditional antidepressants, in which the participants all knew they were getting an antidepressant. That way, both treatments benefitted equally from the positive effect of patients knowing that they were being given a drug instead of a placebo. The findings both surprised and disappointed them: there was virtually no difference. “Unblinding is the defining methodological problem of psychedelic trials. What I wanted to show is that even if you compare psychedelics to open-label antidepressants, psychedelics are still much better,” said Balázs Szigeti, PhD, a clinical data scientist at UCSF’s Translational Psychedelic Research Program, who led the study. “Unfortunately, what we got is the opposite result — that they are the same, which is very surprising given the enthusiasm around psychedelics and mental health.” Szigeti is the co-first author of the paper with Zachary J. Williams, MD, PhD, of UCLA; Hannah Barnett, MSc, of Imperial College, London is also an author. The study appeared March 18 in JAMA Psychiatry.







If you ever wonder whether we have a utilization problem or price problem in US healthcare This chart should help Here are four procedures by rates and prices, in the US, Australia, Switzerland and Germany We don't use more healthcare We just pay much higher prices

🫀 Inflammation vs cholesterol: which residual risk matters most in ASCVD? A large real-world study explored how cholesterol risk (LDL-C) and inflammatory risk (hsCRP) relate to cardiovascular outcomes in patients with established atherosclerotic cardiovascular disease (ASCVD). Researchers analyzed 39,638 patients with ASCVD in routine healthcare in Stockholm between 2007–2021. Patients were stratified into four groups based on LDL-C ≥1.8 mmol/L (≈70 mg/dL) and hsCRP ≥2 mg/L: • Low risk • High cholesterol risk • High inflammatory risk • Combined high cholesterol + inflammatory risk The primary endpoint was major adverse cardiovascular events (MACE), with additional outcomes including cardiovascular mortality, all-cause mortality, and heart failure hospitalization. 📊 Key findings 🔥 Inflammatory risk mattered more than cholesterol alone!!! Patients with high inflammatory risk (hsCRP ≥2 mg/L) had significantly higher rates of: • MACE • cardiovascular mortality • all-cause mortality • heart failure hospitalization 🧬 High cholesterol alone was associated with only a modest increase in MACE and was not strongly linked to other adverse outcomes. ⚠️ Another striking observation: ~39% of ASCVD patients were not receiving lipid-lowering therapy, highlighting persistent gaps in secondary prevention in routine care. 🧠 Why this matters The study reinforces a growing concept in cardiometabolic medicine: ➡️ Residual inflammatory risk remains a major driver of cardiovascular events even when cholesterol is treated. This helps explain why therapies targeting inflammation (e.g., colchicine, IL-1 pathways) are increasingly explored alongside lipid-lowering strategies. 📌 Take-home message ASCVD risk is not just about LDL-C. In real-world populations, inflammation appears to be an equally important — and often overlooked — driver of recurrent cardiovascular events. Future prevention strategies will likely need to target both cholesterol and inflammation.