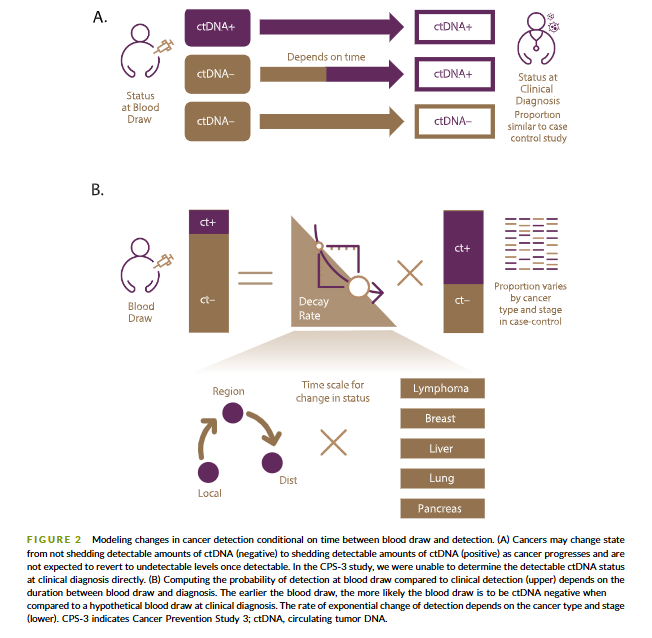
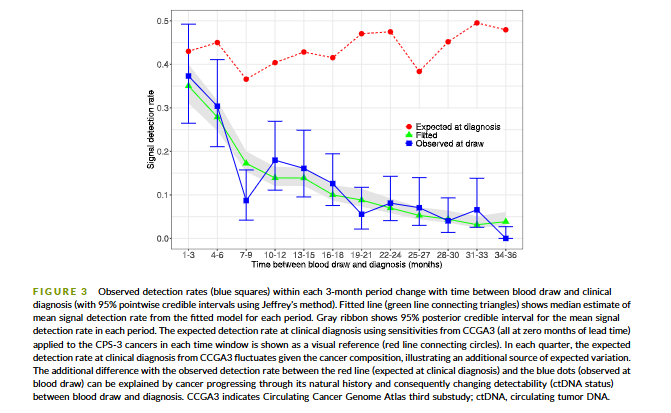
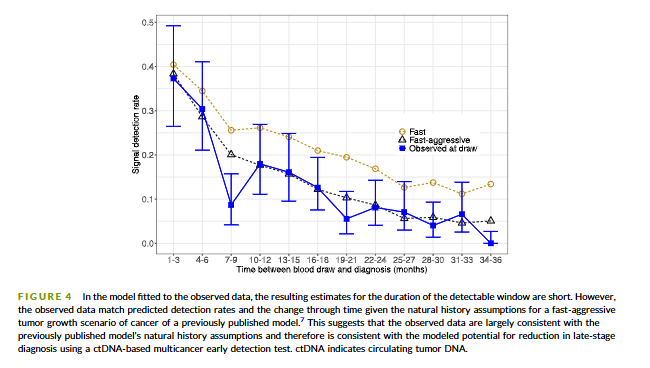
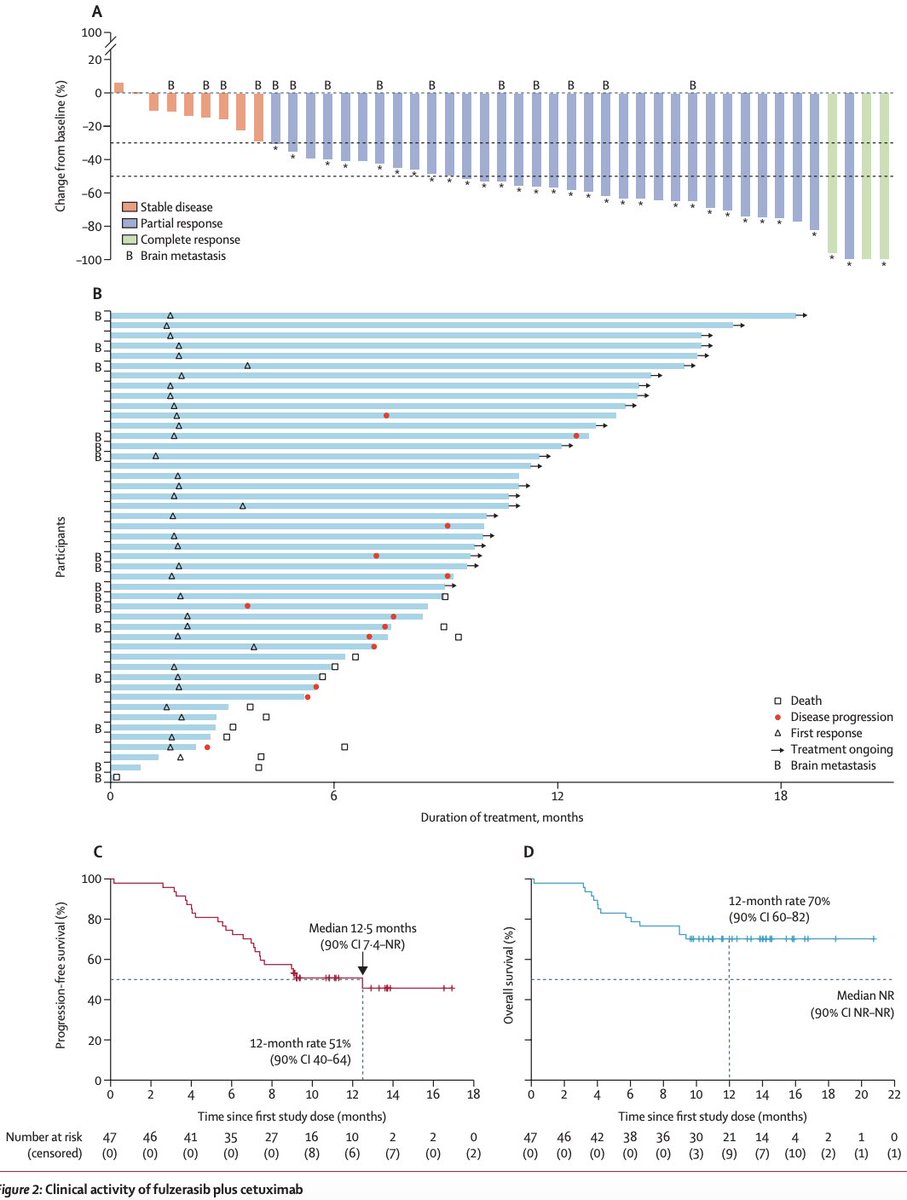
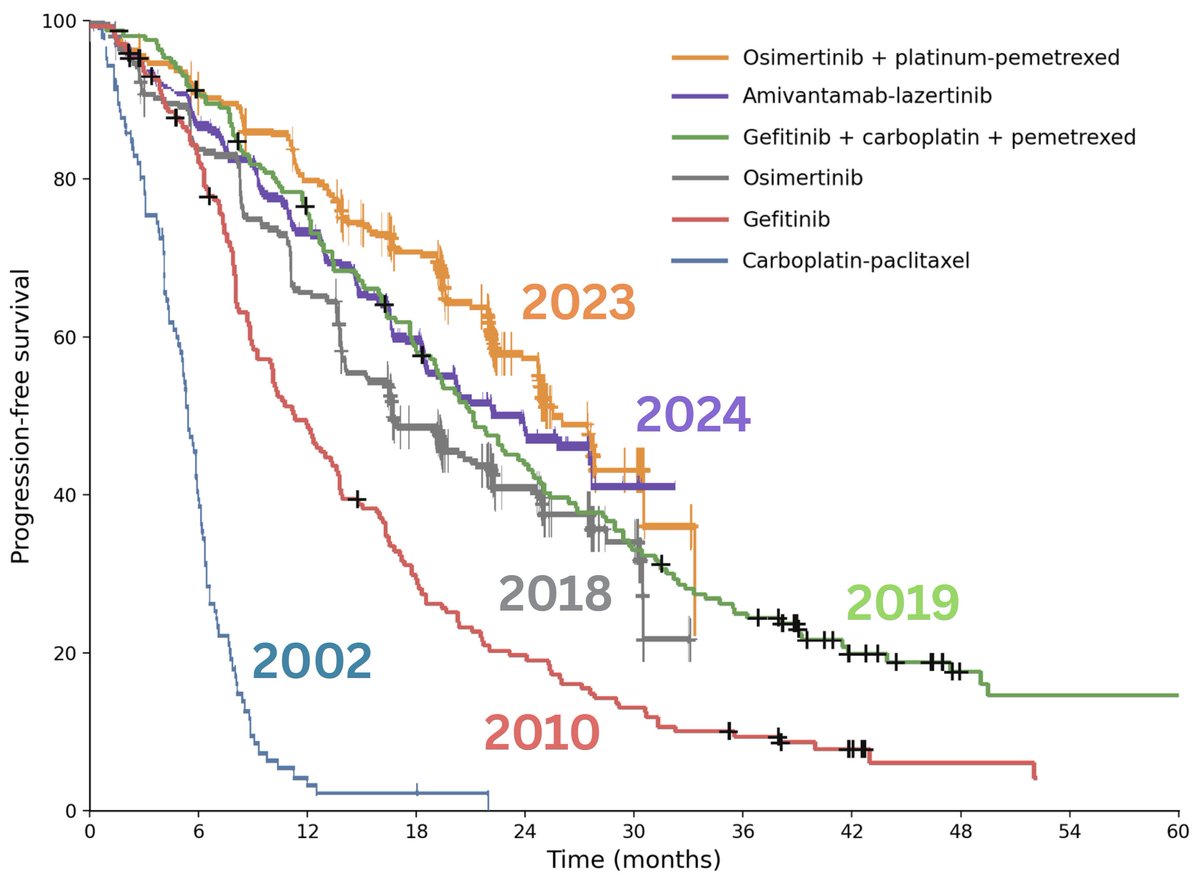
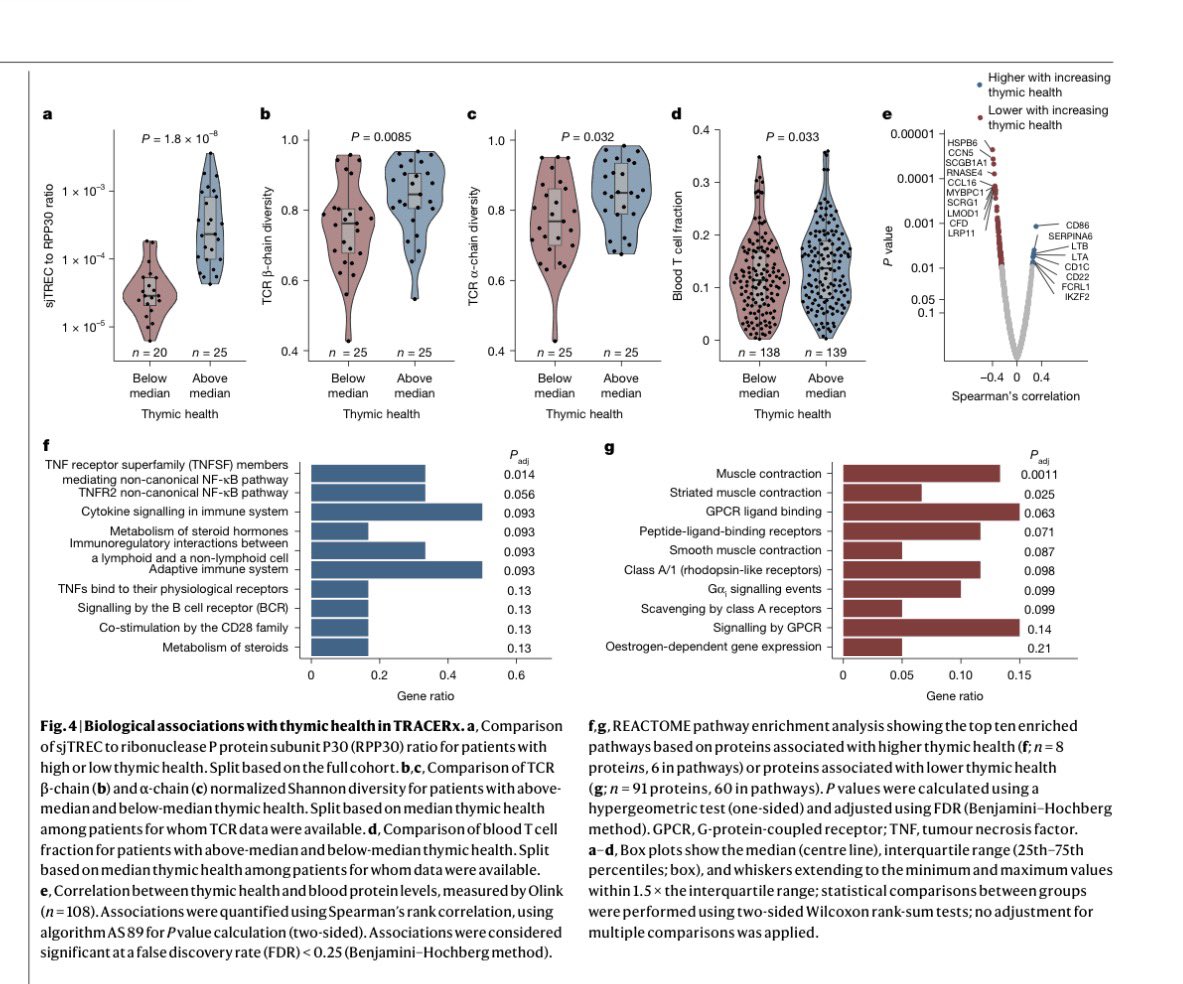
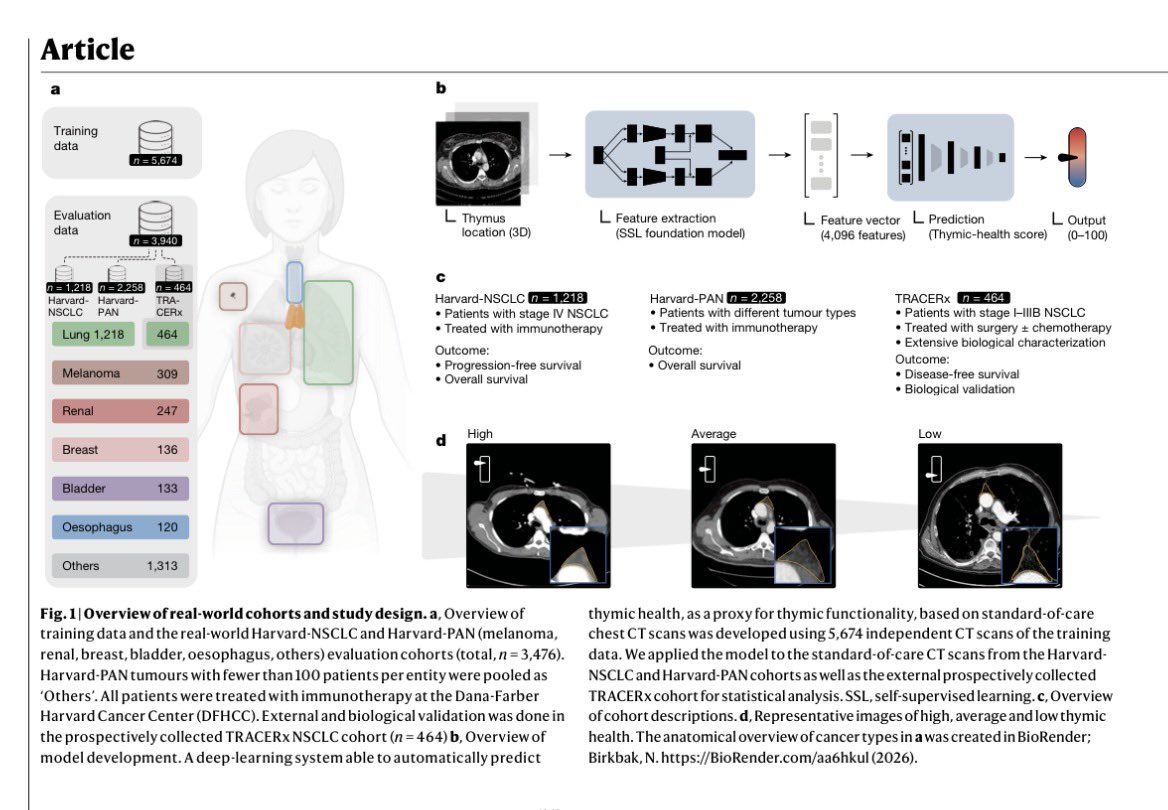
@myESMO Recommendations for clinical reporting of genomics - now published in @Annals_Oncology. We're aiming to advance rational, evidence-based use of genomics in patient care and harmonize biomarker interpretation in cancer treatment annalsofoncology.org/article/S0923-…