
Thomas Hutson, DO, PharmD, PhD FACP
997 posts


@DrTHut
I am a follower of Jesus, husband, father, brother and physician | Pers Views Only |










Voting for the ASCO election is now open! I am honored to be running as one of the candidates for the @ASCO Nominating Committee seat. Visit asco.org/election to learn more about the candidates and place your vote! Thank you!!




Join me - Take 5 mins this wknd to cast your vote in the @ASCO election! Counts for #FASCO points! Fantastic choice leaders on the ballot - @KimrynRathmell for Board, @neerajaiims for nominating committee, @herbloong for international seat on Board. asco.org/about-asco/asc…












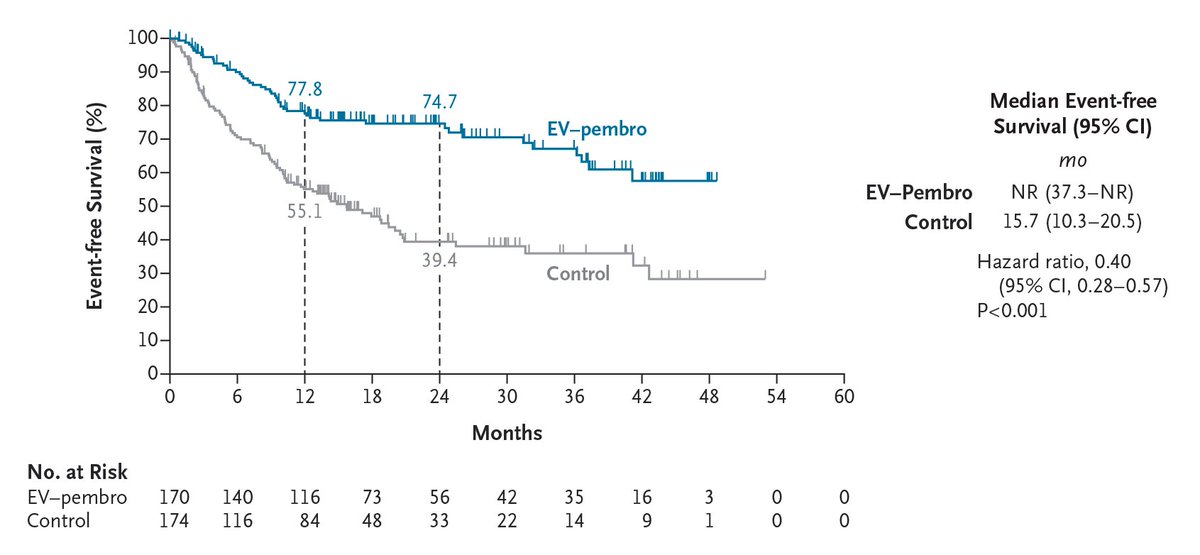
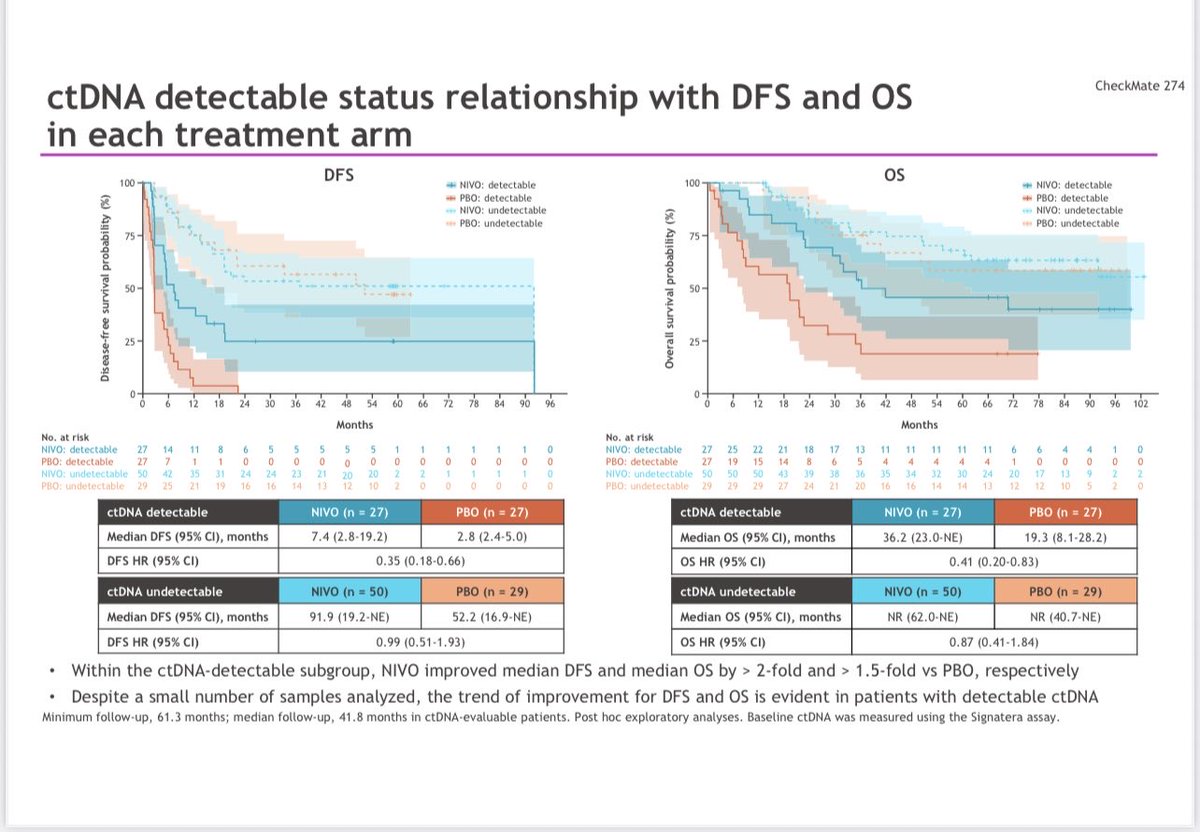
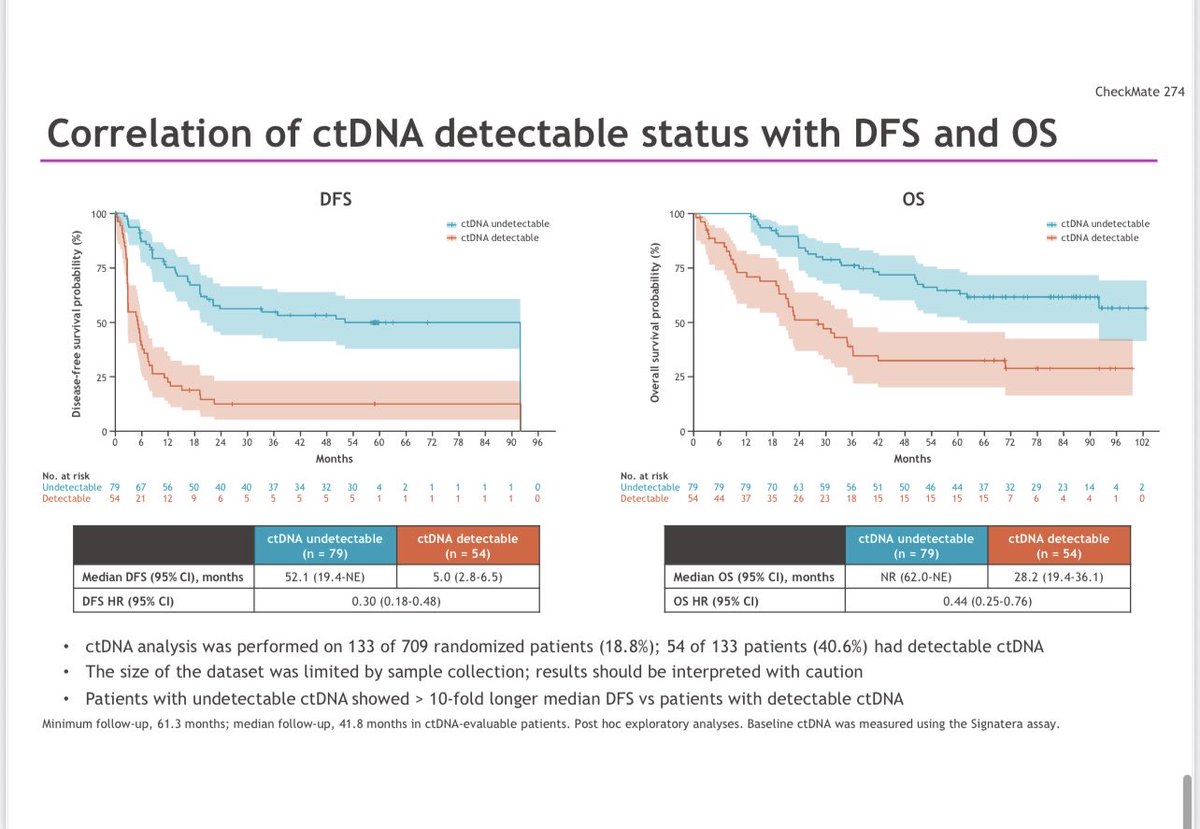
Influential, state-of-the-art, as always presentation by @tompowles1 : Biomarker constructive positive trial on ctDNA-guided adjuvant win in MIBC. Atezolizumab improved DFS (HR 0.64; 9.9 vs 4.8 mo) and OS (HR 0.59; 32.8 vs 21.1 mo) vs placebo in ctDNA+ pts. #ESMO25 #BladderCancer @myESMO @DanaFarberNews @DanaFarber @OncoAlert @OncLive

📣 #ESMO2025: n=565 🇬🇧🇪🇸🇦🇺🇫🇷 RAMPART RCT; Durvalumab + tremelimumab 📈boosts DFS in resected RCC #kidneycancer (HR 0.65, 2-yr 84% vs 78%), esp. in leibovich high-risk pts (HR 0.52, 81% vs 67%). G3+ AES 8% monitoring versus 40% with durva+treme. OS data eagerly awaited! #kcsm




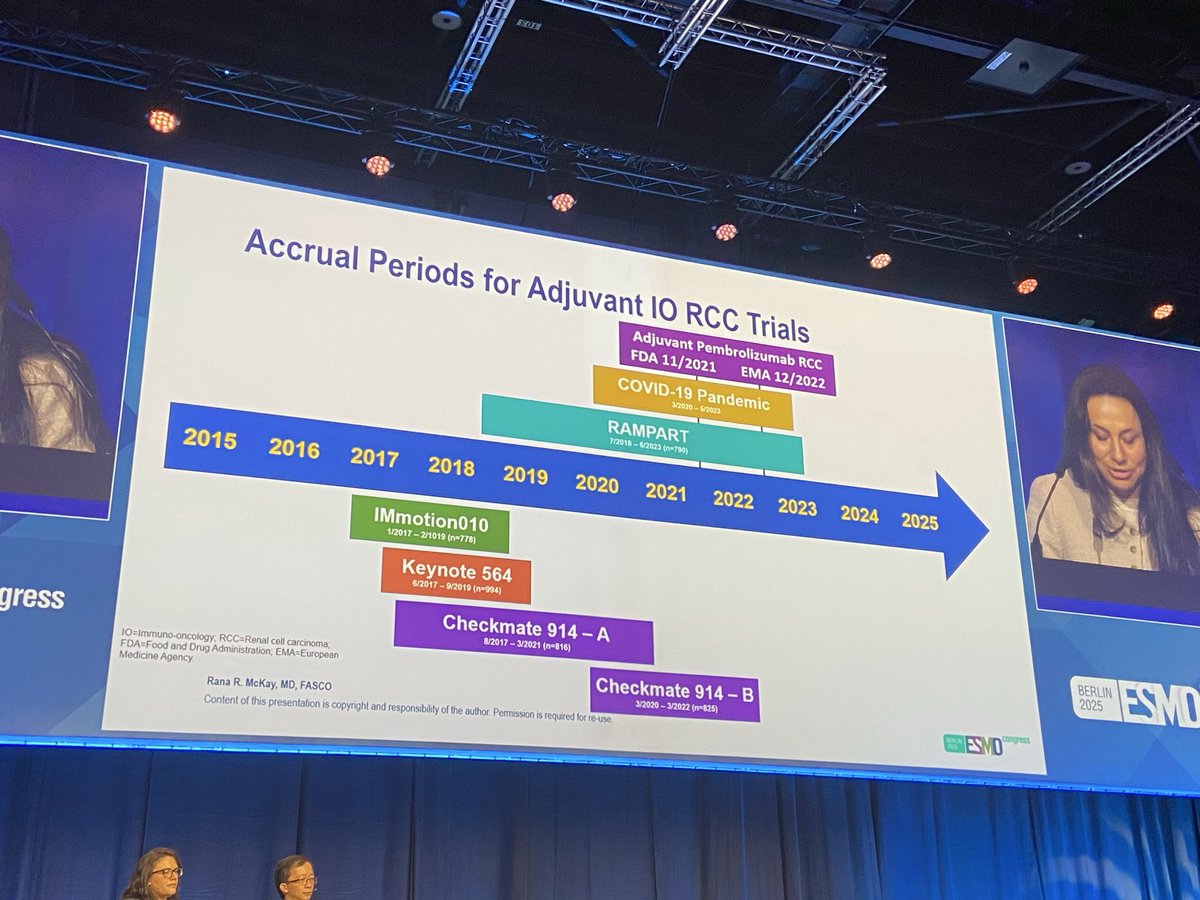



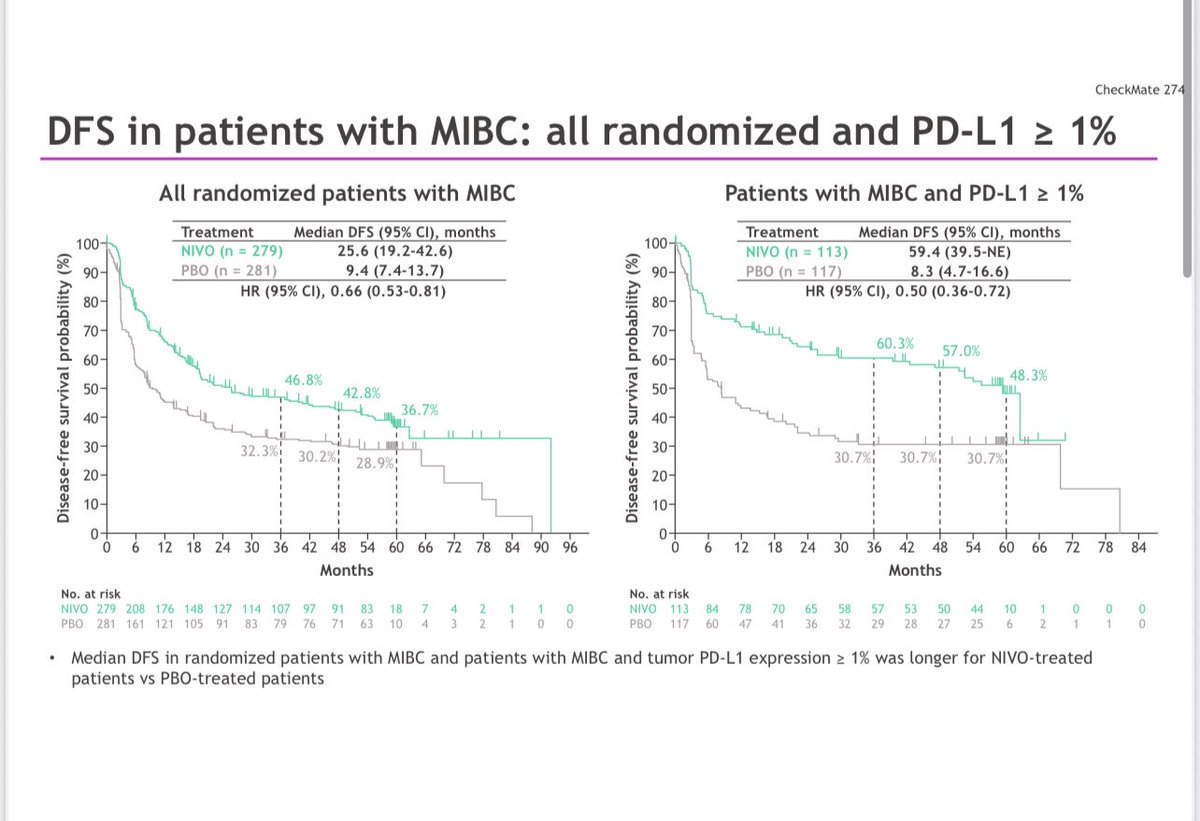
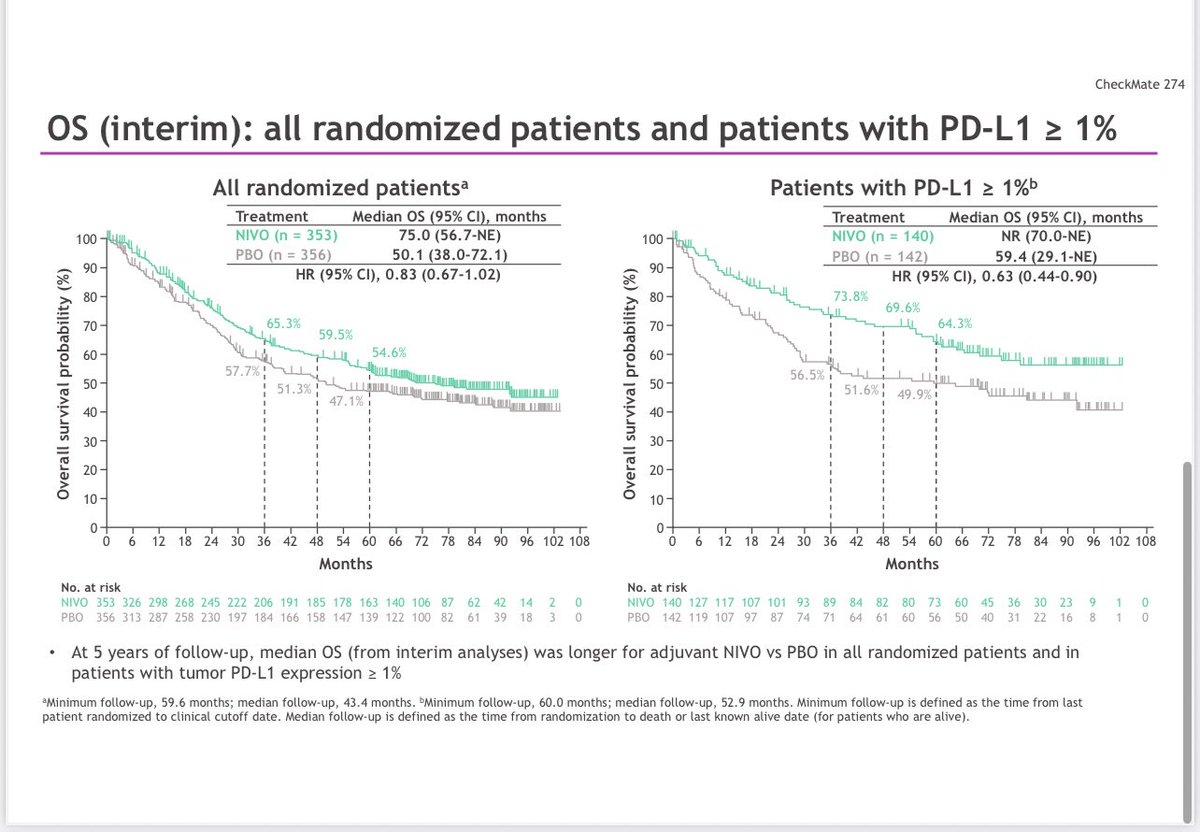

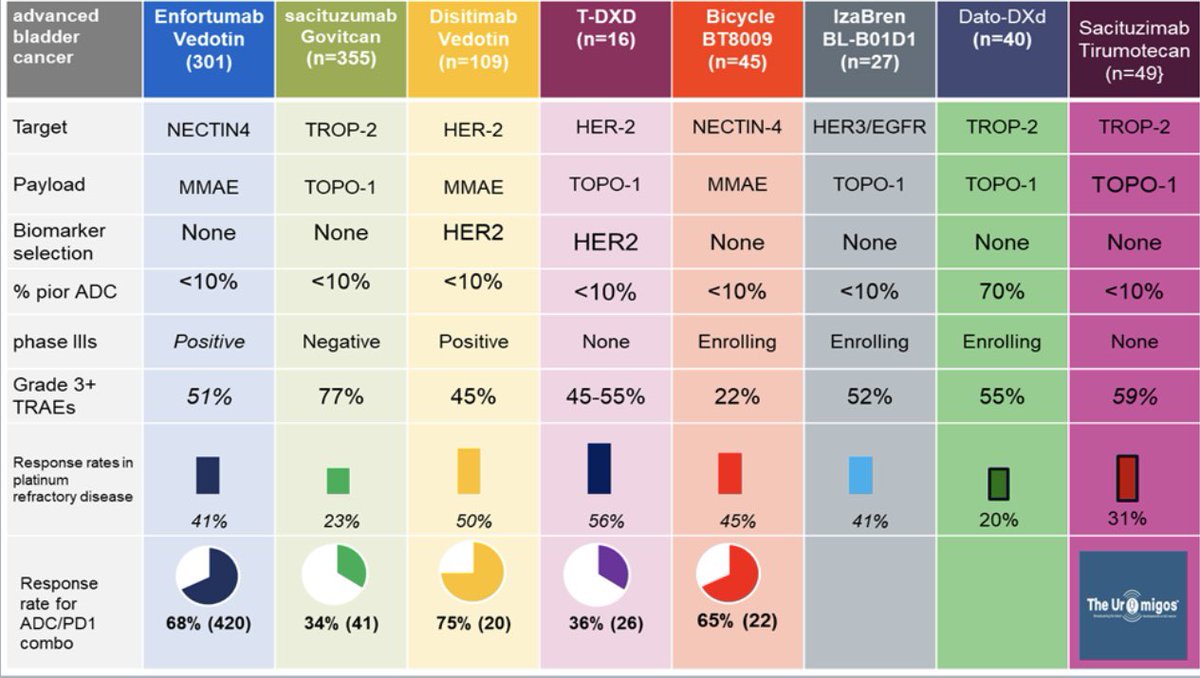
🚨 Hot off the press in @ASCO journal @JCOOA_ASCO 👉Pleased to share our study 👉 "Impact of FDA Project Optimus Guidance on Design of Early-Phase Clinical Trials" 📊 Analysis of 367 phase I #oncology trials (2021-2024) reveals Project Optimus is transforming cancer drug development: 📈 Bayesian designs surged 48% → 75% 🎯 30% now include dose-optimization plans ⚖️ Moving beyond max tolerated dose approach 👉This shift toward smarter, more patient-centered trial design marks a pivotal moment in early-phase cancer research @OncoAlert @JCO_ASCO @PamelaKunzMD #ClinicalTrials #CancerResearch #ProjectOptimus #PrecisionMedicine #FDAGuidance Open access Link: ascopubs.org/doi/10.1200/OA…

