Ethan Rowin retweetledi
Ethan Rowin
58 posts

Ethan Rowin
@EthanRowin
Co-Director Hypertrophic Cardiomyopathy Center, Lahey Hospital and Medical Center; Own tweets not reflective of employer or industry; LFGM
Katılım Kasım 2022
94 Takip Edilen155 Takipçiler
Ethan Rowin retweetledi

Low Incidence of Atrial Fibrillation in Patients with Obstructive HCM Treated with Aficamten: An Analysis from the REDWOOD-HCM, SEQUOIA-HCM and FOREST-HCM Trials #OpenAccess
@EthanRowin @LaheyHospital
heartrhythmjournal.com/article/S1547-…
English
Ethan Rowin retweetledi

In obstructive #HCM, #pVO2, #NYHA, and #KCCQ don’t always align!
Weak correlations and frequent patient-level discordance raise considerations for optimal metric selection in clinical practice and clinical trial design.
🔗kwnsfk27.r.eu-west-1.awstrack.me/L0/https:%2F%2…

English

Honored to join @JAHA_AHA Early Career Editorial Board starting January 2026.
Grateful for the opportunity to learn the editorial process and contribute to high-quality cardiovascular and arrhythmia research. #CardioTwitter #EPeeps

English
Ethan Rowin retweetledi

Low Incidence of Atrial Fibrillation in Patients with Obstructive HCM Treated with Aficamten: An Analysis from the REDWOOD-HCM, SEQUOIA-HCM and FOREST-HCM Trials #OpenAccess
@EthanRowin @LaheyHospital
heartrhythmjournal.com/article/S1547-…
English
Ethan Rowin retweetledi

We presented long-term data from #FOREST #HCM on the incidence and impact of atrial fibrillation in patients treated with aficamten in the Cardiomyopathy LBCT session
Led by @EthanRowin
#ESC2025
#CardioTwitter

English
Ethan Rowin retweetledi

Extended Ambulatory ECG Monitoring Enhances Identification of Higher-Risk Ventricular Tachyarrhythmias in Patients with Hypertrophic Cardiomyopathy #OpenAccess
@EthanRowin @MartinMaronMD
heartrhythmjournal.com/article/S1547-…

English
Ethan Rowin retweetledi

Amazing session on #HCM and arrhythmias — from the newer high-risk makers to challenges of VT ablation, role of PFA, and updated exercise guidelines. #EPeeps #HRS2025 #2025pacesetter @EthanRowin



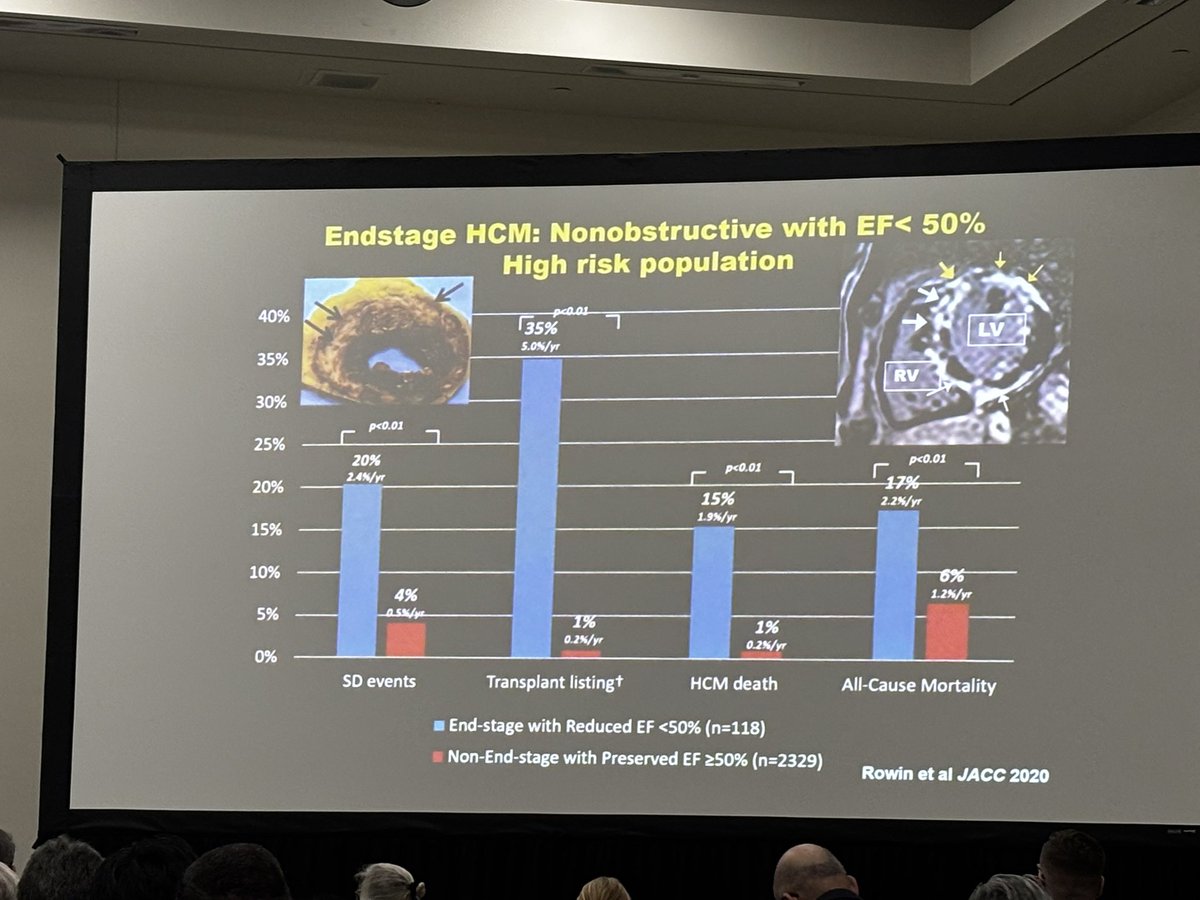
English

@MasriAhmadMD Agree re trade offs and also surprised with a 6month duration btwn echos for pts with EF55-60%, especially since in MAVA-LTE ~6% of patients had either EF<50 or acute HF btwn wks 48 and 156. So potential for more adverse outcomes with this change. Will see with time.
English
Ethan Rowin retweetledi

New mavacamten FDA label update
Most relevant parts:
1) Initiation strategy remains the same
2) Maintenance phase echo requirement dropped to once every 6 months instead of once every 3 months
This will help with patient and staff burden during the maintenance phase. I would have considered LVEF>60% rather than 55% given the variability and the trade-offs. We plan to continue to study the real-world use of mavacamten and safety outcomes with these changes.
Such changes will limit the clinicians' ability to get more frequent echos if concerned about a particular patient since most payers will follow the REMS and decline additional echos.
Initiation phase remains rigid without flexibility in echo scheduling, and the downtitration requirements did not change either.
Overall really positive changes that our patients will like but there will be trade offs.
#CardioTwitter


English
Ethan Rowin retweetledi

How have HCM mortality rates changed before and after COVID ❓
1⃣ Prior to COVID, mortality rates declined by ~ half📉
2⃣ After COVID, mortality rates increased by ~30%, partially undoing some of the prior reductions in HCM mortality.📈
Open access📜: ahajournals.org/doi/full/10.11…

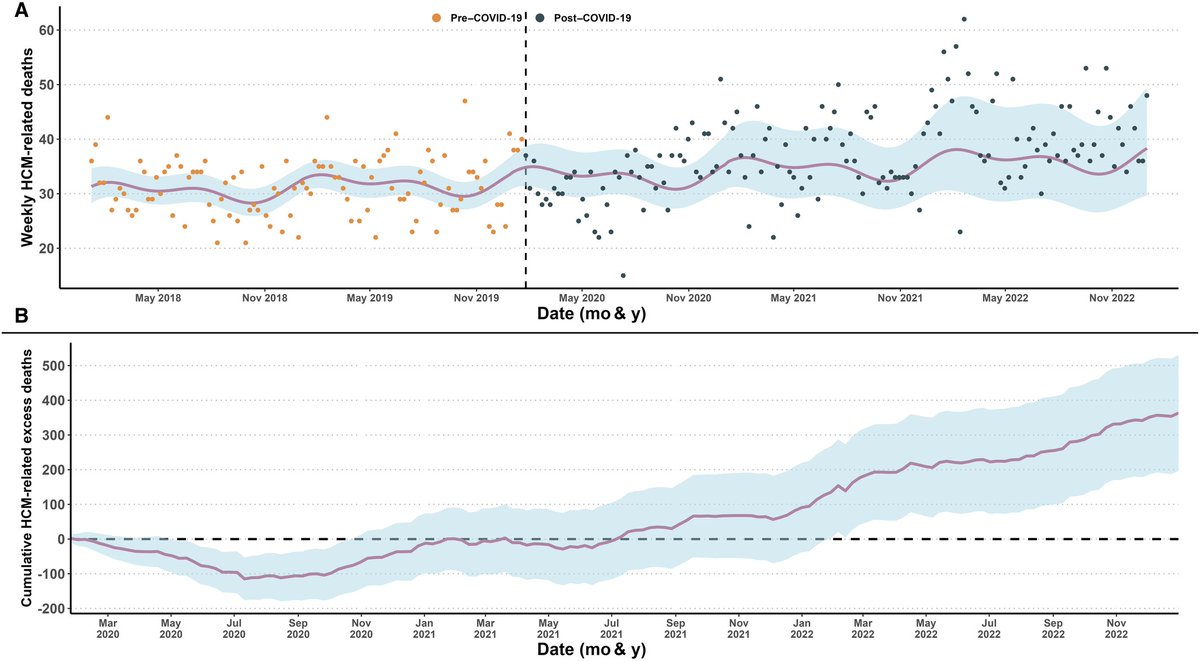
English
Ethan Rowin retweetledi

Really valuable and novel data presented by Dr. @EthanRowin — Does LV apical aneurysm size matter for SCD risk in #HCM? 🏥
Multicenter data says yes!
#ACC25 #CardioTwitter


English
Ethan Rowin retweetledi

“Excited to share our latest editorial published in @CircAHA - Our study highlights T1 mapping as a novel marker for heart failure progression in hypertrophic cardiomyopathy (HCM).
@EthanRowin @JAHA_AHA @AHAScience

English
Ethan Rowin retweetledi

Mavacamten in #nonobstructive #HCM design and baseline characteristics paper is out (ODYSSEY trial) !
Biggest unmet need in HCM is nonobstructive HCM. We look forward to the results.
Impressive baseline characteristics with significant burden of disease
Sample size caught my eye. The study planned to enroll 420 patients, yet the paper shows 580 patients were randomized. The paper does not provide rationale (if I missed it please point it out). This makes ODYSSEY the largest study conducted in #HCM
Other interesting aspects: unclear how titration is being approached, and this is the first time we see a global study of mavacamten with 1 mg being used.
Results expected this year
jacc.org/doi/epdf/10.10…




English
Ethan Rowin retweetledi

Disopyramide appears safe and effective in treating symptomatic outflow obstruction in HCM over extended durations of time (≥5 years). #AHAJournals @danmassera @nyulangone @EthanRowin @LaheyHospital ahajrnls.org/4hC5AnC

English
Ethan Rowin retweetledi

Disopyramide for long-term treatment of obstructive #hcm? Check out our work looking at patients treated for ≥5 years at @nyulangone @LaheyHospital with 67% still on drug after ~7 years
@EthanRowin @MartinMaronMD
ahajournals.org/doi/full/10.11…
English

@jeffreygeske @hcmsociety Had a great time talking with @jeffreygeske about management of apical aneurysms in HCM
English
Ethan Rowin retweetledi

#cardiotwitter: How do you approach apical aneurysms in hypertrophic #cardiomyopathy? Excited to contribute to the latest @hcmsociety "In the Thick of It" podcast with @EthanRowin #cvHCM pod.link/1694758840/epi…
English

@MasriAhmadMD Congrats! Impressive results. The AFib data is remarkably low and very reassuring. Do you have publicly available info on timing of the few EF drops?
English

We presented new data from #FOREST #HCM which is an ongoing 5 year long term extension trial of aficamten. This analysis is restricted to 280 pts with #oHCM
Primary endpoint at week 12
Titration period in the first 6 weeks given its half life
Follow along
#CardioTwitter


English
Ethan Rowin retweetledi

@RickNishimura and Dr. Barry Maron at #HCMSummit8. Great to hear from many experts on HCM. @HCMSummit @SteveOmmen @MartinMaronMD

English

