Francesco Claps retweetledi
Francesco Claps
4.4K posts


Francesco Claps
@FraClaps
MD, PhD candidate @UniLeiden | Consultant Urological Surgeon @iov_irccs | Fellow @NKI_nl | Former fellow Fundaciòn IVO | @SIU_Italia Communications Office
Katılım Aralık 2011
199 Takip Edilen795 Takipçiler
Francesco Claps retweetledi

Design and Rationale of the ROGUE-1 Registry: A Multicentric Prospective Registry for Primary T1 Nonmuscle-invasive Bladder Cancer by David D'Andrea et al on behalf of the ROGUE-1 Investigators
Read the full article: buff.ly/a4L34IS
We thank the authors for choosing EUO to publish your work!
@uroweb
@mroupret
@GPloussard
@jteoh_hk
@Ric_Campi
@CaPsurvivorship
@LauraMarandino
@RenuEapen
@Ecastromarcos
@OncoAlert
@Sciencedirect

English
Francesco Claps retweetledi

Priority Article in EUO today: An Editorial
Immunotherapy Escalation in BCG-Naïve NMIBC: Time for Selectivity, Not Enthusiasm by Marco Moschini, Maria Carmen Mir, Jonathan Aning, and Alessandro Volpe on behalf of EAU Oncology Section
Read the full article: buff.ly/U6qXTYb
We thank the authors for their work!
@uroweb
@mroupret
@GPloussard
@jteoh_hk
@Ric_Campi
@CaPsurvivorship
@LauraMarandino
@RenuEapen
@Ecastromarcos
@OncoAlert
@Sciencedirect

English
Francesco Claps retweetledi

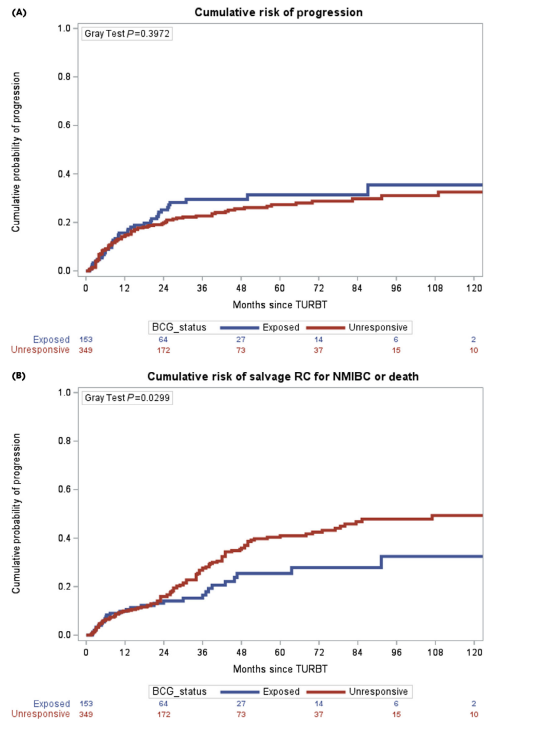
We validated BCG failure classifications
~5-year progression ≈30% in both BCG-exposed and BCG-unresponsive groups—but salvage RC is used more often in BCG-unresponsive pts
doi.org/10.1111/bju.70… @Pietro9609 @UroMoschini @BenjaminPradere @RobertoContieri @SBoorjian @BJUIjournal
English
Francesco Claps retweetledi

Diagnostic Value of Fluorine-18-fluorodeoxyglucose Positron Emission Tomography/Computed Tomography in Staging Histological Subtypes of Invasive Bladder Cancer by Renee A.G. Lijnen et al
Read the article: buff.ly/wjGnWUU
We thank the authors for trusting #EUO with your work.
@LAUrology_NL
@TBoellaard
@FraClaps
@uroweb
@mroupret
@GPloussard
@jteoh_hk
@Ric_Campi
@CaPsurvivorship
@UroDocAsh
@LauraMarandino
@RenuEapen
@Ecastromarcos
@OncoAlert
@Sciencedirect

English
Francesco Claps retweetledi

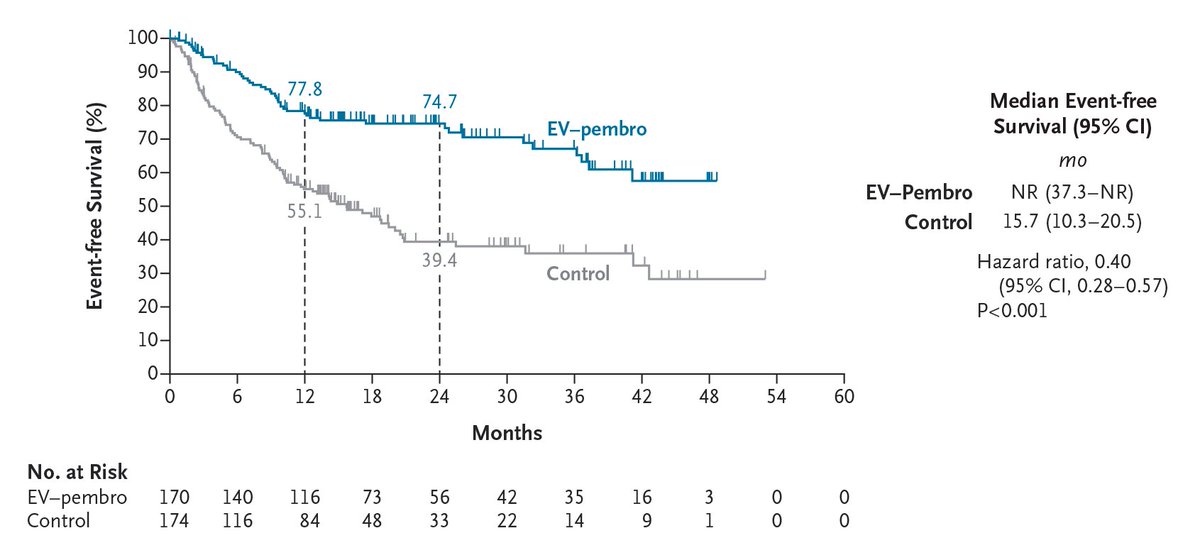
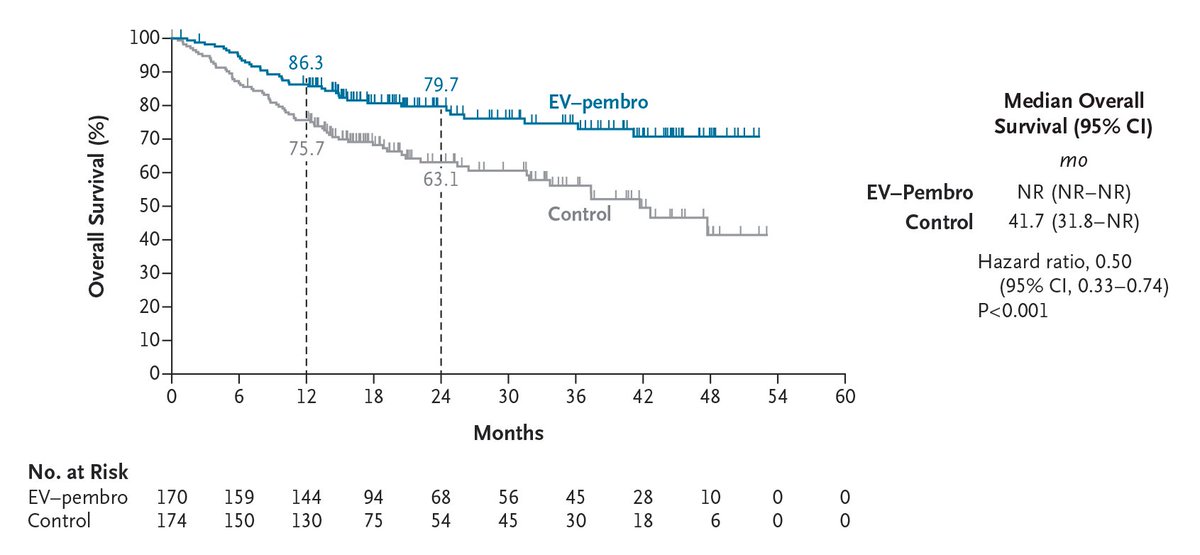
Original Article: Perioperative Enfortumab Vedotin and Pembrolizumab in Bladder Cancer (phase 3 KEYNOTE-905/EV-303 trial) nej.md/4czl8sG
Editorial: Enfortumab Vedotin plus Pembrolizumab as Perioperative Therapy nej.md/4twnv5T
#Oncology
English
Francesco Claps retweetledi

Honored to announce the second edition of SYNERGY.
Save the date!
SYNERGY@Synergy_Uro
SYNERGY 2026 – Second Edition Save the Date: 🗓️ May 28–29, 2026 📍 Naples, Italy 👉 This year’s scientific program will focus on locally advanced and oligometastatic GU tumors, in an integrated MDT setting
English
Francesco Claps retweetledi

in J Urol this month a decision analysis of PLND during RP
Take home message: benefits of PLND outweigh risk for most patients.
auajournals.org/doi/10.1097/JU…
@VickersBiostats @UrologyMSK #prostatecancer
@Uroweb @AmerUrological
English
Francesco Claps retweetledi

📣 Corso di Perfezionamento UNIPA: “Diagnostica e Procedure Urologiche Guidate da Imaging”
🗓️ Marzo–Novembre 2026 | 400 ore | 16 CFU
Focus su imaging in urologia (eco, TC, RM mp, PET/CT) + procedure guidate.
⏳ Scadenza: 03/02/2026
Info: dipartimento.meprecc@unipa.it
Italiano
Francesco Claps retweetledi

🎆📢First of 2026!
Should we really give MMC maintenance in intermediate-risk #NMIBC, and for how long?
#EAU guidelines recommend adj chemo but the optimal schedule remains unclear
We tried to answer this clinically relevant gap.👇🧵
@Uroweb @urotoday @oncodaily @SpringerNature


English

📬We are pleased to share our comment on @JAMA_current
Prostatitis continues to represent diagnostic and therapeutic challenges. We discuss key aspects of prostatitis, with particular emphasis on the role of imaging in prostatic abscess' evaluation 👇
jamanetwork.com/journals/jama/…
English
Francesco Claps retweetledi

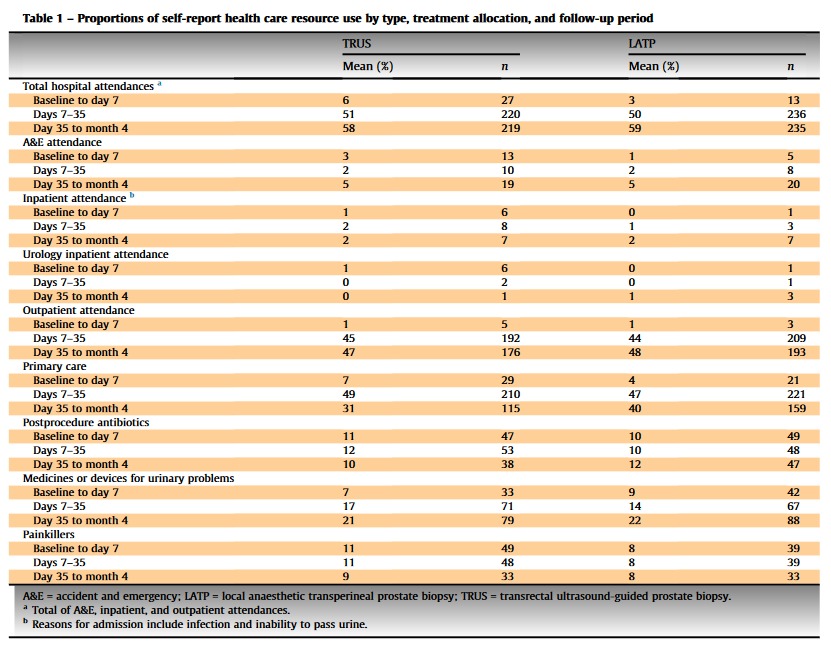
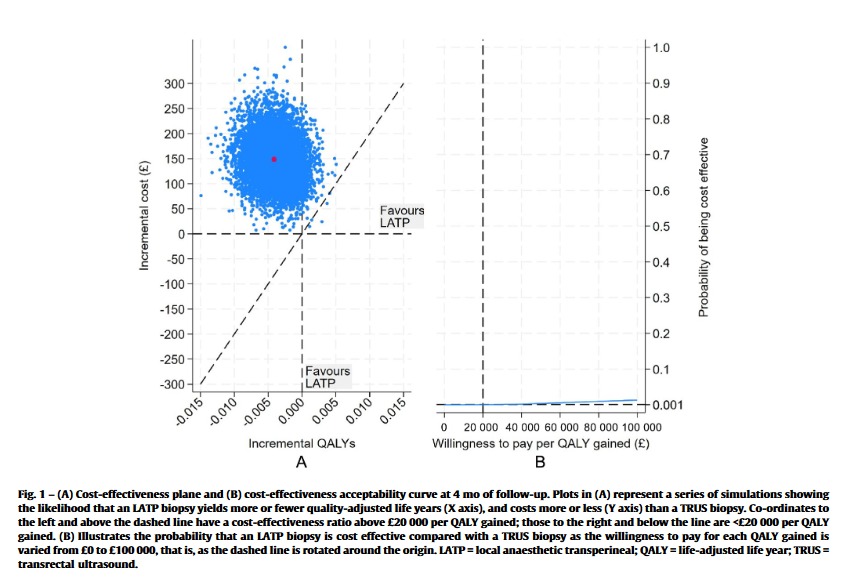
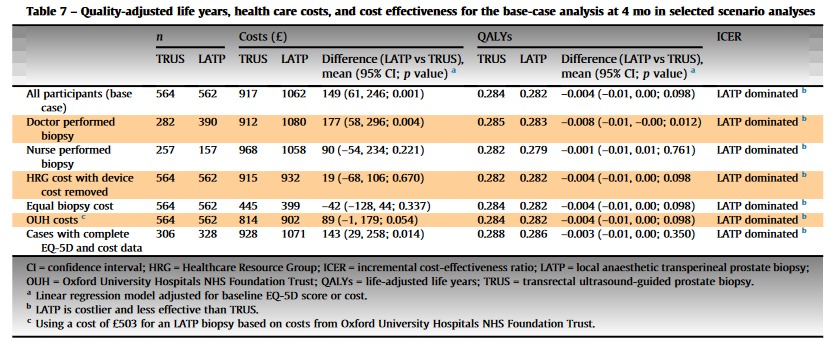
Pleased to publish #CostEffectiveness data #LATPvsTRUS in @EurUrolFocus
THM:
•average cost TRUS=£917; LATP=£1062 (∵ device cost/takes longer)
•#LATP not cost-effective (QALY)
But...
•only 4m f/u so doesnt account for ⬆️ PCa diag
#TRANSLATE_Trial
eu-focus.europeanurology.com/article/S2405-…

English
Francesco Claps retweetledi

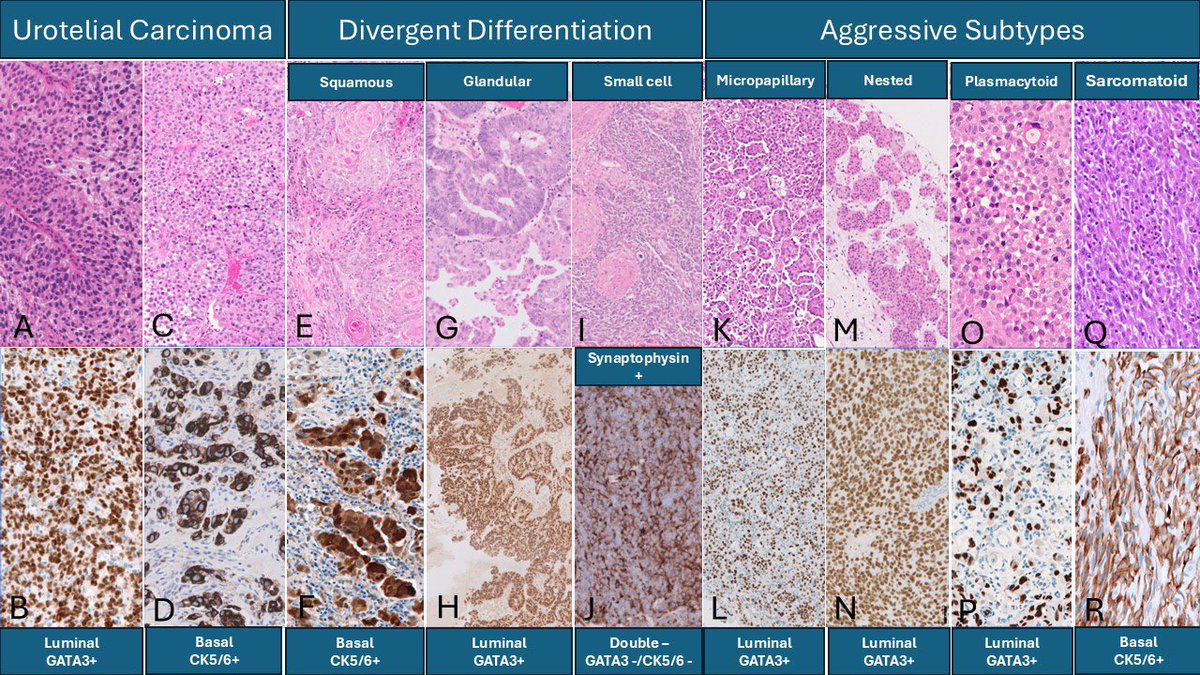
Need an update on the molecular pathology of bladder cancer? I am delighted to share a comprehensive review article by Dr. Antonio Lopez-Beltran and colleagues, published in the 2026 Annual Review Issue of Histopathology @Histo_Journal @daniel_berney
🔗 Article: onlinelibrary.wiley.com/doi/abs/10.111…
Advances in molecular classification, biomarker development, and personalized therapies are transforming the management of bladder cancer. The rapid expansion of therapeutic targets - together with ongoing clinical validation—underscores the essential role of pathologists, as pre-analytical and analytical considerations directly impact the approval, adoption, and optimal use of new cancer drugs. Integrating molecular and morphologic data into routine pathology reporting will be critical to support clinical decision-making and enable precision-based treatment.
Molecular classification remains a rapidly evolving research area with major potential to reshape clinical practice. The marked molecular and morphological heterogeneity of urothelial carcinoma presents challenges, but also offers opportunities to refine patient stratification and tailor therapeutic interventions. Emerging liquid-based biomarkers—including circulating tumor DNA (ctDNA) and urinary tumor DNA (utDNA)—show strong promise for detecting minimal residual disease, monitoring treatment response, and assessing disease dynamics in a minimally invasive manner.
For more GU-focused reviews, the full 2026 Histopathology Annual Review Issue (Special GU Edition) is available here:
🔗onlinelibrary.wiley.com/toc/13652559/2…


English
Francesco Claps retweetledi

#mdpijcm
🩺#HighlyCitedPaper
Citations 29🙌
The Prognostic Significance of Histological Subtypes in Patients with 𝐌𝐮𝐬𝐜𝐥𝐞-𝐈𝐧𝐯𝐚𝐬𝐢𝐯𝐞 𝐁𝐥𝐚𝐝𝐝𝐞𝐫 𝐂𝐚𝐧𝐜𝐞𝐫
👉mdpi.com/2885444
👥@GGiannarini @AlessiaCim @Nicp85Pavan @ongarluc_1991 @FraClaps
@MediPharma_MDPI

English
Francesco Claps retweetledi

BCG-unresponsive NMIBC trials feel confusing—and inconsistent EFS reporting is a big reason why
We analyze the CIS+ cohorts of 4-FDA registration trials to provide interpretable EFS estimates
🧵👇
@Annals_Oncology
@tompowles1
@JoshMeeks
@wandering_gu
doi.org/10.1016/j.anno…

English
Francesco Claps retweetledi

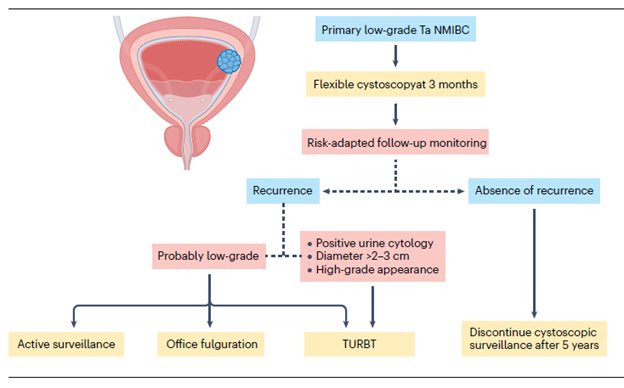
“Low-grade Non–Muscle-Invasive Bladder Cancer: Molecular Landscape, Treatment Strategies, and Emerging Therapies” - just published in Nature Reviews Urology @NatRevUrol. This State-of-the-Art review by Drs. Roger Li @UrogerliMD, Lexi Wen, and colleagues @philippespiess @spsutkaMD @LDyrskjot @DrShariat @UroDocAsh offer an outstanding overview of advances in molecular biology, diagnosis, targeted therapies, and clinical management for #bladdercancer. A great honor to be part of this effort.
Summary:
The management of low-grade non–muscle-invasive bladder cancer (NMIBC) is undergoing rapid evolution, driven by a growing recognition of the need for nuanced, risk-adapted strategies that minimize overtreatment. Yet widespread adoption of de-escalated approaches remains limited by the lack of well-defined, evidence-based guidelines tailored to this favorable-prognosis disease subset. Clear recommendations - particularly regarding surveillance frequency, duration, and criteria for de-intensified care - will likely require international consensus efforts supported by robust prospective data.
A major priority for future research is refining risk stratification. Distinguishing patients at truly increased risk of progression from those with indolent disease will enable more personalized management, including appropriate use of active surveillance and reduced surveillance intensity. Advances in molecular profiling, urine-based biomarkers, and AI-assisted pathology show promise for identifying meaningful biomarkers and histologic patterns to support such stratification, but rigorous prospective validation remains essential before broad clinical adoption.
AI integration into clinical workflows offers additional opportunities to enhance diagnostic accuracy, predict recurrence or progression, and support individualized decision-making. Multimodal models capable of real-time risk assessment may eventually guide therapy selection, but challenges - including the need for diverse training datasets, transparent algorithms, and clear ethical and regulatory frameworks - must be addressed before routine clinical use.
Despite favorable overall outcomes, managing low-grade NMIBC remains challenging owing to high recurrence rates and continued reliance on invasive cystoscopy and imperfect biomarkers. Until emerging diagnostic tools are validated, established clinicopathological systems such as the IBCG risk stratification model will continue to guide treatment decisions. Patients with multiple risk factors generally warrant intravesical therapy, while those without risk factors may be reasonable candidates for active surveillance. The ongoing BCG shortage further underscores the need to balance clinical benefit with treatment burden and to evaluate new intravesical therapies against their potential financial and clinical toxicity.
Meaningful progress in low-grade NMIBC will depend on prospective validation of emerging technologies and therapies, paired with thoughtful integration into evidence-based guidelines. By aligning molecular insights, technological innovation, and risk-adapted clinical practice, the field can improve outcomes while reducing unnecessary interventions and surveillance for this common, yet often overtreated, disease.
The article is available at the following link:
nature.com/articles/s4158…

English

Great brainstorming session today at #EMUC25 focused on advancing the most current insights in urothelial carcinoma research 🚀

English
Francesco Claps retweetledi

One slide says it all! Terrific distillation by @MichvdHeijden of what to do in emerging pt populations in #bladdercancer, namely pts who received perioperative IO. Mirrors discussions we have been having in #kidneycancer. #ESMO25

English
Francesco Claps retweetledi

⚡️ Cost-effectiveness of treatments for BCG-unresponsive CIS - analysis from our multicenter IBCG team. Five strategies modeled:
🔹Radical cystectomy (RC)
🔹Pembrolizumab
🔹Nadofaragene firadenovec
🔹Nogapendekin (NAI-BCG)
🔹Gemcitabine + Docetaxel
➡️Comprehensive head-to-head economic model for this disease. 1️⃣/5 🔗
@AmandaMyersMD @RuchikaTalwarMD @pjhensley11 @SWilliams_MD @AmyLuckenbaugh @UrogerliMD @megynkelly @WesKassouf @AndreaNecchi @VigneshPackiam @garysteinbergmd @BogdanaSchmidt @SimaPorten @IBCG_BladderCA

English

