Sabitlenmiş Tweet
Helms Research Group
5.2K posts


Helms Research Group
@GroupHelms
We solve problems in energy and sustainability with better materials.
Lawrence Berkeley National Lab Katılım Şubat 2014
3.9K Takip Edilen5.2K Takipçiler

Another collab milestone with @LeibfarthGroup @ElizaNeidhart Stephanie Ribet, Karen Bustillo, Eric Dailing @molecularfndry, @brisingr2012 @ReimerLab Taehyun Lee, Logan Kearney, Colin Ophus, Sophia Fricke, Ah-Young Song, Jeff Reimer, Erik Alexanian Joanna Atkin @BerkeleyLab
English

Just out in @ScienceAdvances! We create co-continuous blend architectures from diverse polyolefins using PDK chemistry, revealing in stride how microstructures within each phase communicate and align. Fantastic properties relevant to plastics recycling. science.org/doi/10.1126/sc…

English

@JoshuaSchrier @ACS_AMI Ah. They made a choice to go for “whisky”, and not “whiskey”. Brave
English

My work is often "whiskey inspired", but this new @ACS_AMI paper takes it to a new level pubs.acs.org/doi/10.1021/ac…

English

This is a FRAUDULENT paper, AI-generated. My name was used as an author and I had nothing to do with it, never saw it until today e-pubmed.co.uk/journals/digit…
The "Editors" Angelo Rossi Mori, David Mensah, and Zarnie Khadjesari should be reported.

English

Congrats to Michael Baird (PhD 7 from our group) on this recognition. See you in Chicago!
ACS PMSE Division@acspmse
Congratulations to the 2026 Class of PMSE Future Faculty! Join us at the PMSE Future Faculty Symposium during #ACSFall2026 in Chicago to celebrate these emerging leaders in polymer science and engineering. 📅 Aug 24–27, 2026 📍 Chicago, IL #PMSE #FutureFaculty #PolymerScience
English

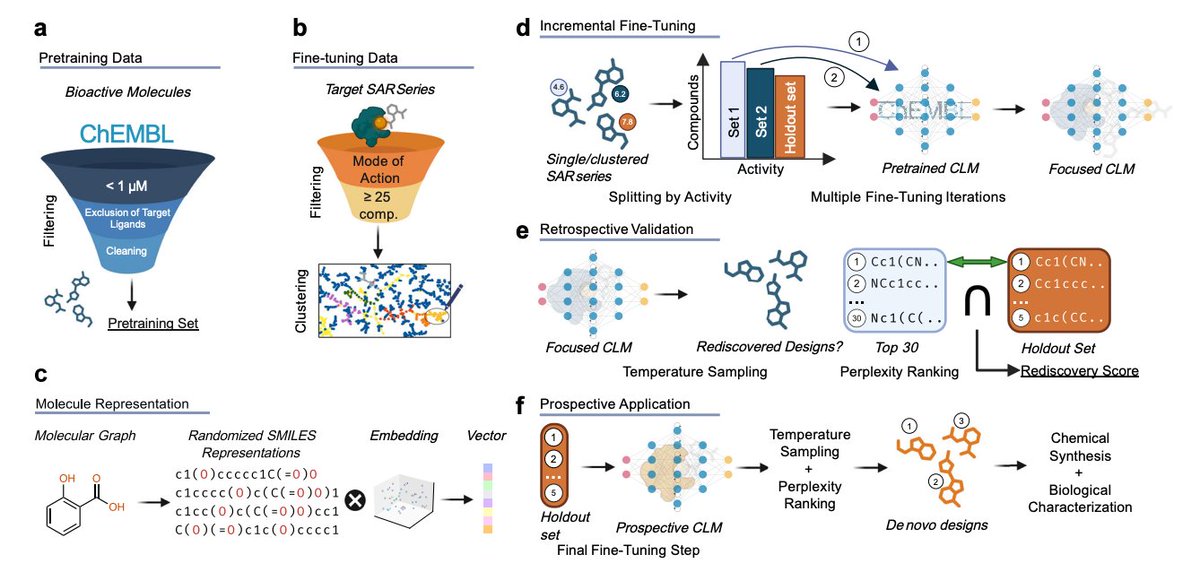
Incrementally trained language models design drug molecules more potent than their training data
Chemical language models (CLMs) are now standard for de novo molecular design: pretrain on millions of SMILES, fine-tune on ligands for a target of interest, sample candidates. But one task has stubbornly resisted them—structural optimization, meaning taking a known active scaffold and squeezing more potency from it without leaning on external oracles like docking or QSAR predictors.
Tim Hörmann and coauthors close this gap with a deceptively simple idea: mimic how a medicinal chemist actually learns. Instead of dumping all known analogs into one fine-tuning pass, they split the structure-activity relationship (SAR) series into subsets of increasing potency and fine-tune an LSTM-based CLM incrementally—one potency tier at a time. The model walks up the activity ladder, refining what it learned at each step.
Retrospectively, across 27 PPARγ agonist SAR series, the incremental strategy consistently beats one-shot fine-tuning at rediscovering held-out high-potency molecules. The perplexity-potency correlation also becomes positive and sharper, meaning the model internally "knows" which samples should be most active—without any external scorer.
Then comes the prospective test. Applied to a benzimidazole PPARγ agonist scaffold, 9 synthesized top-ranked designs all outperform the best known training molecule. Five benzoic acid derivatives hit EC50 values of 0.6–3.1 nM, 12 to 62× more potent than the reference. A second campaign on RORγ inverse agonists produces a 30 nM sulfonamide 20× more potent than its closest training neighbor—and the model correctly picked a rare but potency-driving N-tert-butyl motif present in just one training molecule, capturing long-range SAR dependencies.
Two points stand out from an ML angle: the added potency dimension in the training schedule creates a smoother gradient landscape that LSTMs exploit well, and perplexity alone—a model-intrinsic quantity—becomes a reliable ranking signal, removing the external-oracle bottleneck that has constrained generative drug design.
For industrial drug discovery pipelines, this is a practical shift: hit-to-lead optimization can be driven directly from in-house SAR tables without building a bespoke scoring model for every target, which matters especially where docking or QSAR predictors are unreliable. It brings generative chemistry meaningfully closer to the daily workflow of medicinal chemistry teams.
Paper: Hörmann et al., Nature Communications (2026) — CC BY 4.0 | doi.org/10.1038/s41467…
English

Join us in A401 - Georgia World Congress Center 8AM-12PM for a fantastic line up in energy materials: Delia Milliron, Joaquín Rodríguez-Lopez, Yan Yao, Wei Wang, Yet-Ming Chiang, Kristin Persson and myself. #acs
Română

Have you experienced that your are an author of an article that you never wrote, read or even its existence. Today, Prof. Yury Gogotsi and I surprisely found that we have "co-authored" a paper on a journal World Journal of Advanced Engineering Technology and Sciences. We immediately wrote the following letter to the journal editor. Please watch out!
Dear Editors,
It came to our attention that an AI-fabricated article titled “Influence of Reaction Parameters on the Crystallinity and Aspect Ratio of Lanthanum Nanorods" appeared in your journal with the authorship assigned to us. We have not authored this article and request that you immediately remove it from your website and any connected sites.
researchgate.net/profile/Lara-V…
Sincerely,
Yury Gogotsi and Zhong Lin Wang
English

@NatureSynthesis Figure 3 Panel b has an incorrect chemical structure for the guanidinium cation
English

Now online:
News & Views on the Article by Xiwang Zhang & co-workers
nature.com/articles/s4416… ($)
Nanosheet-confined spaces for high-density polymer membrane synthesis by Lie Liu & Xiaoqiang An
nature.com/articles/s4416… ($)
English

Just out in @PNASNews ! We use nanoparticle-surfactants to create droplet interface bilayers with ion-selective nanopores, enabling memristive ionic conduction, size-/charge-selective transport, and neuromorphic plasticity. @doescience @molecularfndry pnas.org/doi/10.1073/pn…
English

@NUChemistry @NorthwesternEng @tedsargentNU_TO @TrienensInst @IINanoNU @WeinbergCollege plastics are already low carbon....it's the scale at which they are made that leads to heaps of carbon
English

Renewables are cheap—but intermittent. ⚡ @NUChemistry researchers from the Sargent Group built a low-energy electrolyzer that turns waste syngas into ethylene, moving toward low-carbon plastics.
🔗 bit.ly/4kCBG54

English

Collab led by Shira Haber, Nicodemo Ciccia, and Zhengxing Peng with Feipeng Yang, Julia Im, Mutian Hua, Sophia Fricke, Raynald Giovine, Cheng Wang, John Hartwig, and Jeffrey Reimer @doescience funded @molecularfndry @UCBerkeley @advlightsource
English

Just out in @ChemicalScience ! We use NMR relaxometry to explore the microstructure of upcycled HDPE, wherein we find amidyl groups confined to the rigid amorphous phase. pubs.rsc.org/en/content/art…
English

Outstanding collab: Dong-Min Kim, Gan Chen, Michael A. Baird, Livia Pugens Matte, Youngmin Ko, Carson O. Zellmann-Parrotta, Jiwoong Bae, Zhaoyang Chen, Yan Yao, Mary C. Scott, Ying Chen @molecularfndry @BerkeleyLab @PNNLab @UHouston @UCBerkeley
Filipino

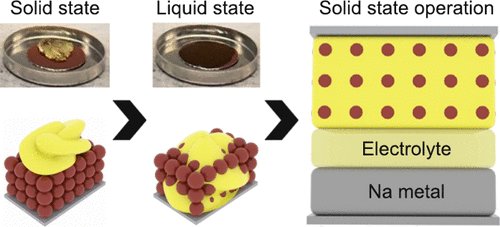
Just out in @ACSEnergyLett ! We showcase thermoformable organo ionic conductors with high Na+ conductivity in solid-state sodium metal batteries with organic cathodes. @energystoragera @doescience pubs.acs.org/doi/10.1021/ac…
English

@JoshuaSchrier @theNASciences For a second, I thought they were reporting on a public release of alcohol at sea, akin to the boston tea party
English

@theNASciences They appropriately send the announcement... so early in the morning 🎶🤣
English

I’m pleased that @theNASciences is finally addressing the age old question: What do you do with a drunken sailor? 🎶 🍻⛵️ 🤣 nationalacademies.org/projects/DBASS…
English

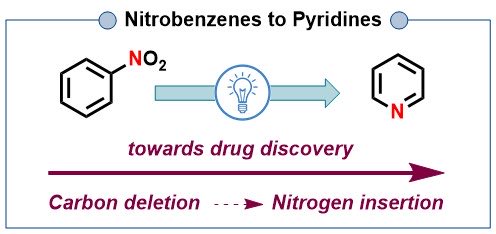
Nitrobenzenes to pyridines via N-atom insertion and selective carbon deletion (C5 or ipso-C).
onlinelibrary.wiley.com/share/AS8BW3AU…
English

