

BioSniper
215 posts











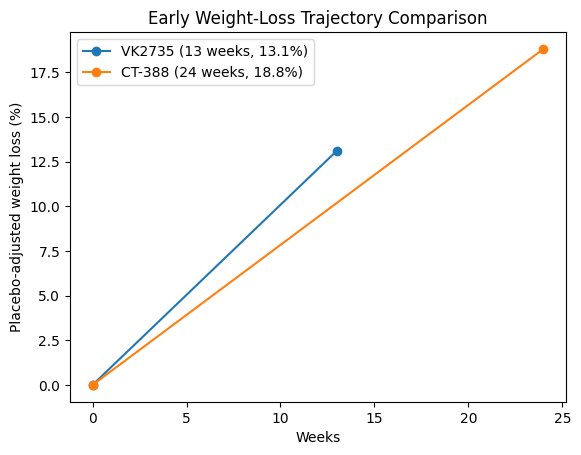

$VKTX "closed-loop" VK2735 ecosystem could be a key differentiator in the commercial setting. Was interesting


This is the same $NVO guy who was just a couple of weeks ago talking that Metsera deal would have cut two years of drug development for novo. Now either in denial or trying to keep it down so target biotechs don’t shrug off to even > $20 B $LLY $VKTX youtu.be/YpqMNlUvyyU?si…






The mental state confusion by $NVO management on obesity MOA superiority can’t be more clear than M Lange answer a little ago to analyst question about $LLY tirzepatide vs CagriSema Redefine 4 trial that novo , in self inflicting manner , decided to run and will be reported q1. ? Despite clear disadvantage of carisema over even semaglutide in terms of tolerability and even CV risk factors , M Lange continues to state the opposite on tirzepatide. No wonder NVO is moving on $MTSR and $PFE not presenting a higher bid $VKTX






𝐒𝐅 𝐇𝐞𝐚𝐥𝐭𝐡𝐜𝐚𝐫𝐞 𝐖𝐞𝐞𝐤: @WaveLifeSci's CEO Paul Bolno discusses the company's INHBE obesity program, AATD, PNPLA3, and more. $WVE Full video: biotechtv.com/post/wave-life…



$NVO is moving amycretin from phase 1 to 3 straight with much less data set ( with FDA approval ) Also did a small phase 3 for oral semaglutide ( didn’t have to do 4000 pt trials ) There is no way FDA would block $VKTX from going to oral phase 3. Not a single legitimate reason. More than 5000 pts already got exposed. Viking already interacted with trial sites. They should get it going quickly with only 500-800 pts. CEO referred to oral amylin more than once last few weeks and today. They must be working hard on combining oral vk2735 and new amylin. He even mentioned one or two tablet today. If you want to see how much preclinical work they did on oral formulation , look at the oral vk2735 patent dug out by @GilaMonstrum patentimages.storage.googleapis.com/90/43/52/ea76e…





