

PanCAN
38.4K posts


@PanCAN
Our vision is to create a world in which all patients with pancreatic cancer will thrive. 💜


















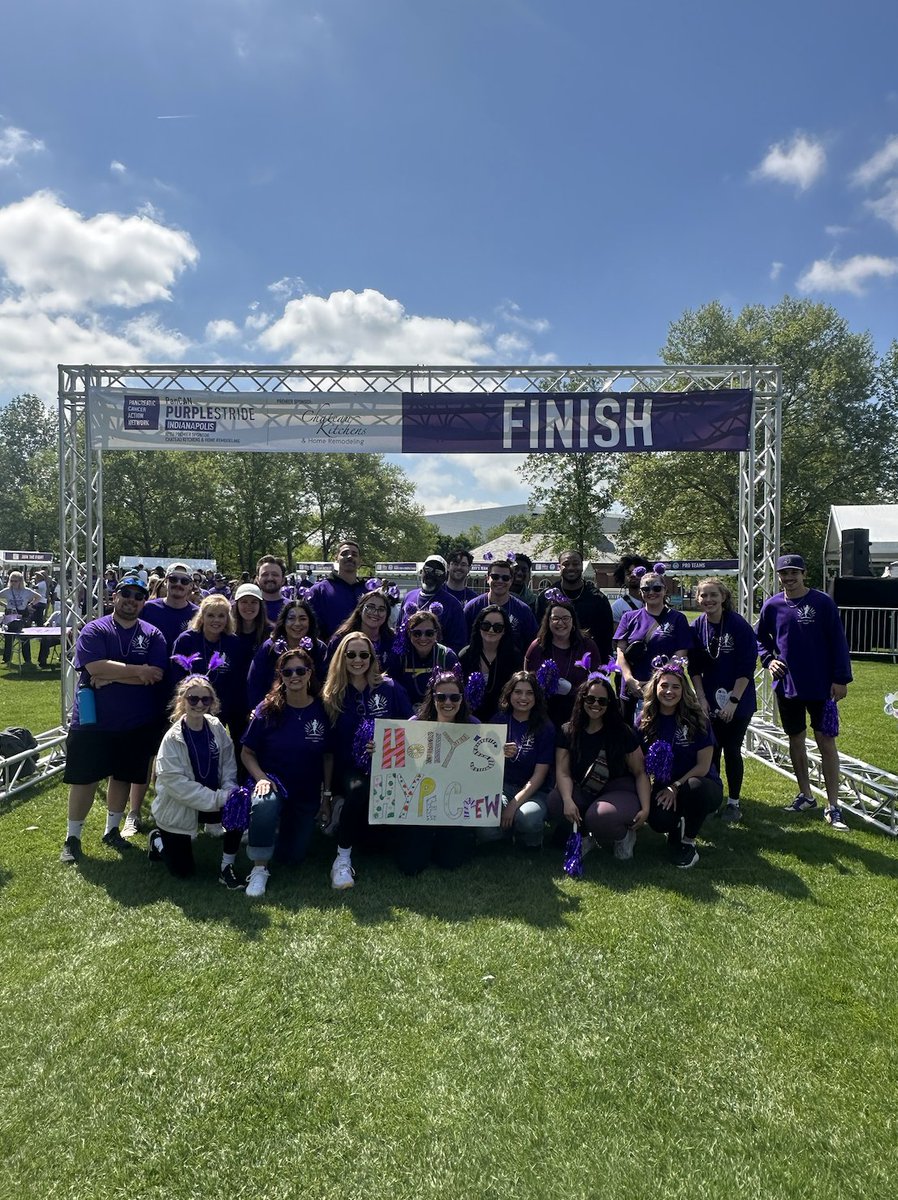



























Today, the U.S. Food and Drug Administration (FDA) announced that it has issued a “safe to proceed” letter to @RevMedicines, allowing the company to initiate an expanded access treatment protocol (EAP) for daraxonrasib for patients with previously treated metastatic pancreatic ductal adenocarcinoma (PDAC). 💊 Expanded access is a potential pathway for patients with serious or life-threatening conditions to gain access to investigational medicines outside of a clinical trial when they are unable to participate in a clinical trial and all other appropriate treatment options have been exhausted. In response to the FDA’s letter, Revolution Medicines said that this “represents a critical step in the process of opening an EAP” and that they are “moving as quickly as possible to ensure safe and equitable access to daraxonrasib for eligible patients in the United States,” according to a statement from the company. Read Revolution Medicines’ Expanded Access Policy, including how to submit a request through a licensed treating physician and how daraxonrasib is extending survival for #pancreaticcancer patients at 🔗 pcan.at/rjul9sza.

