Sabitlenmiş Tweet
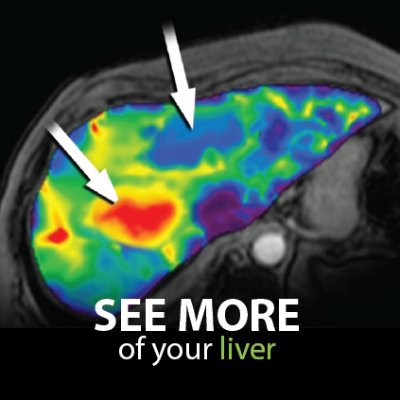
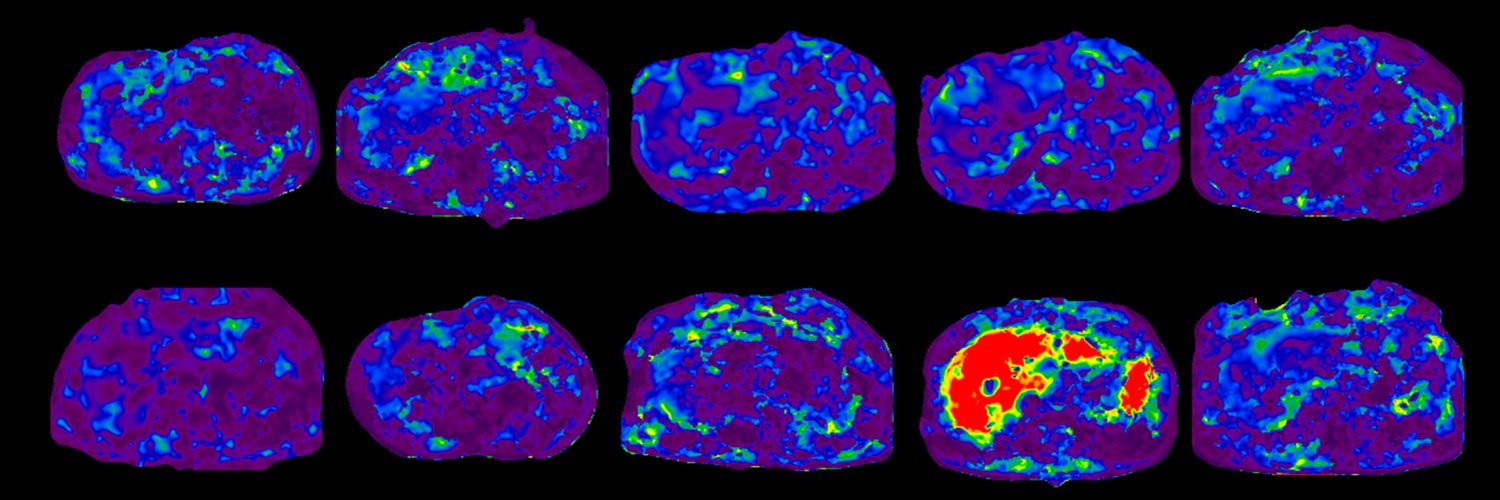
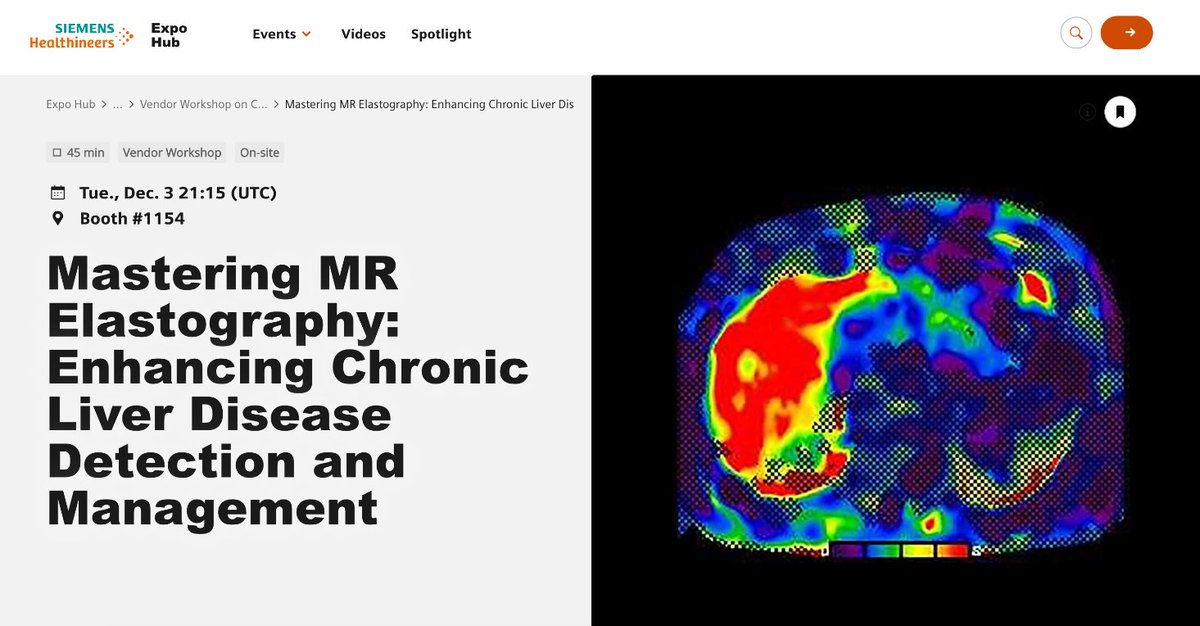
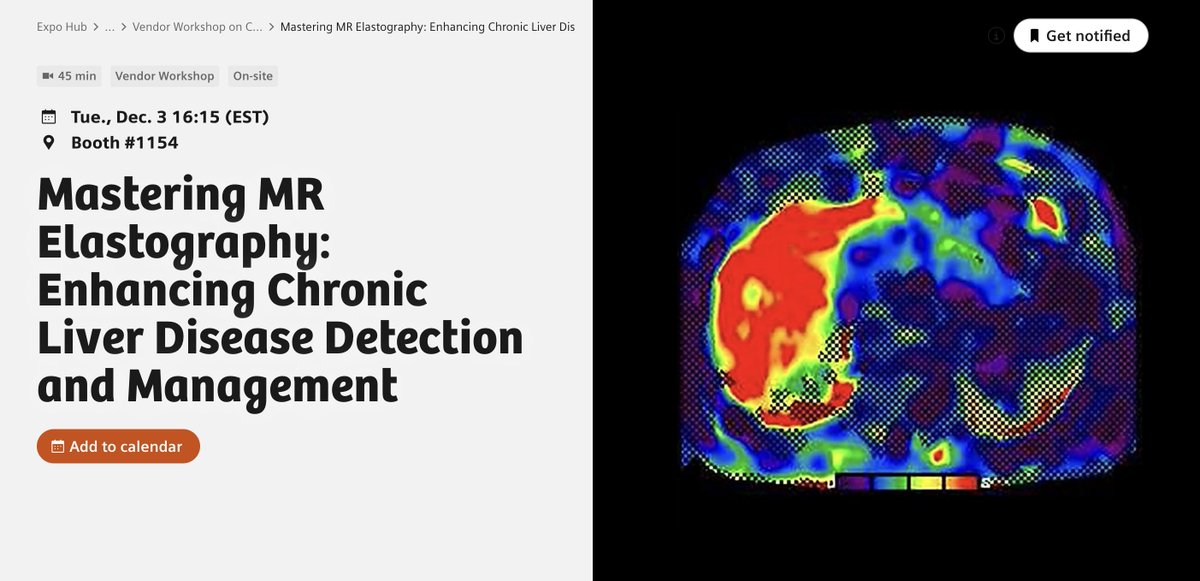
Excited about the new @AASLDtweets practice guidelines on NAFLD/NASH that point to the routine use of the MAST and MEFIB scores for at-risk NASH and fibrosis staging?
Now you can use coulditbenash.org to calculate your patients' risk 🎯
English