Mike Kalutkiewicz retweetledi
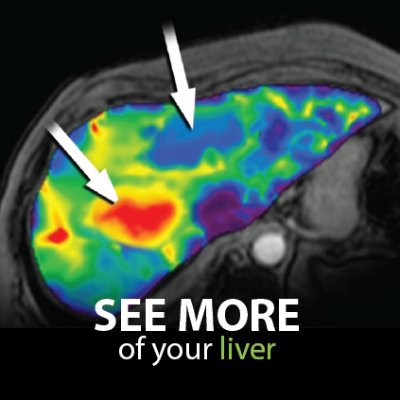
We're excited to report that Resoundant’s MRE Accepted into FDA Biomarker Qualification Program as a Reasonably Likely Surrogate Endpoint (RLSE) in MASH Trials
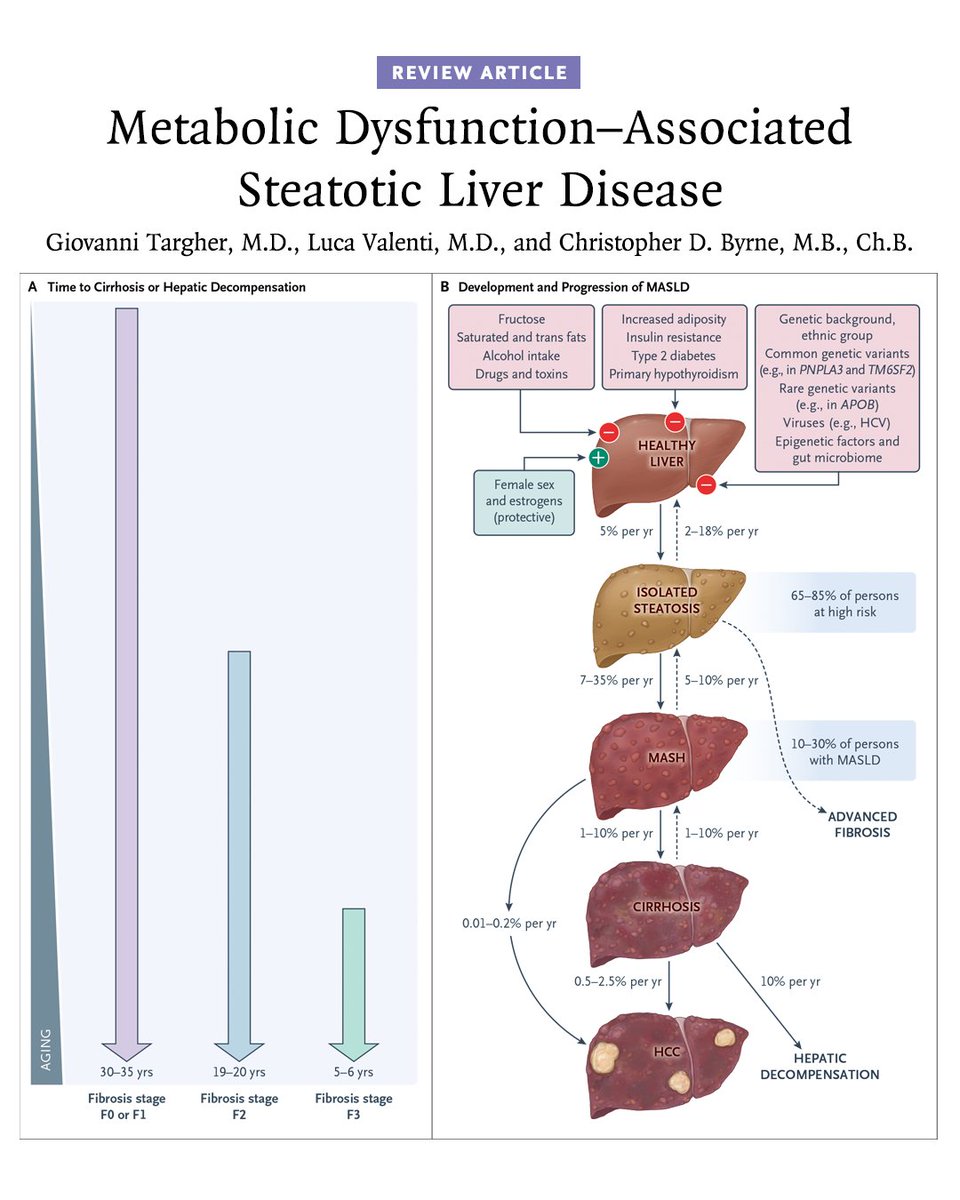
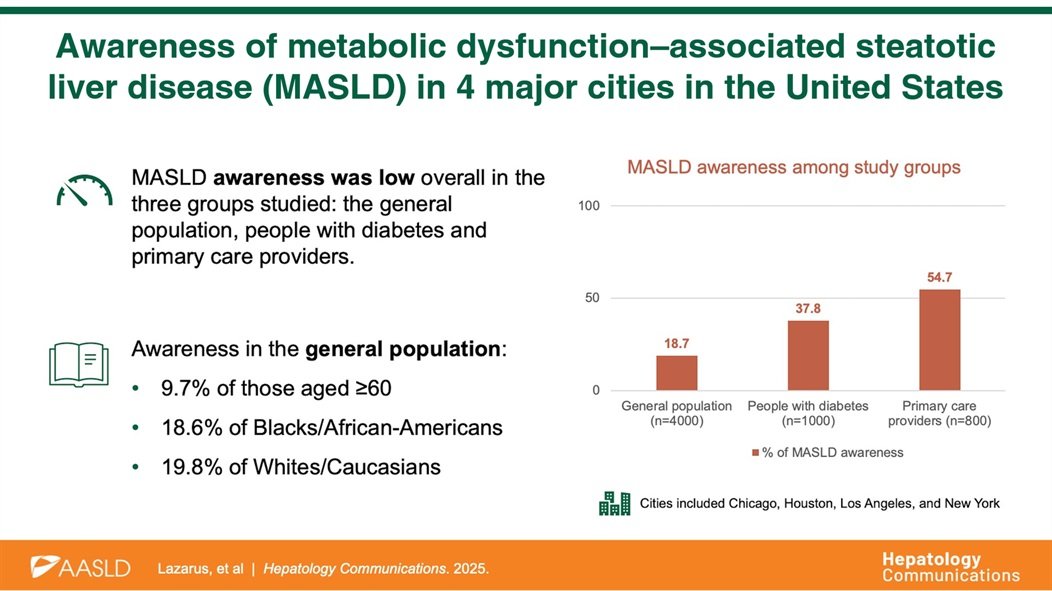
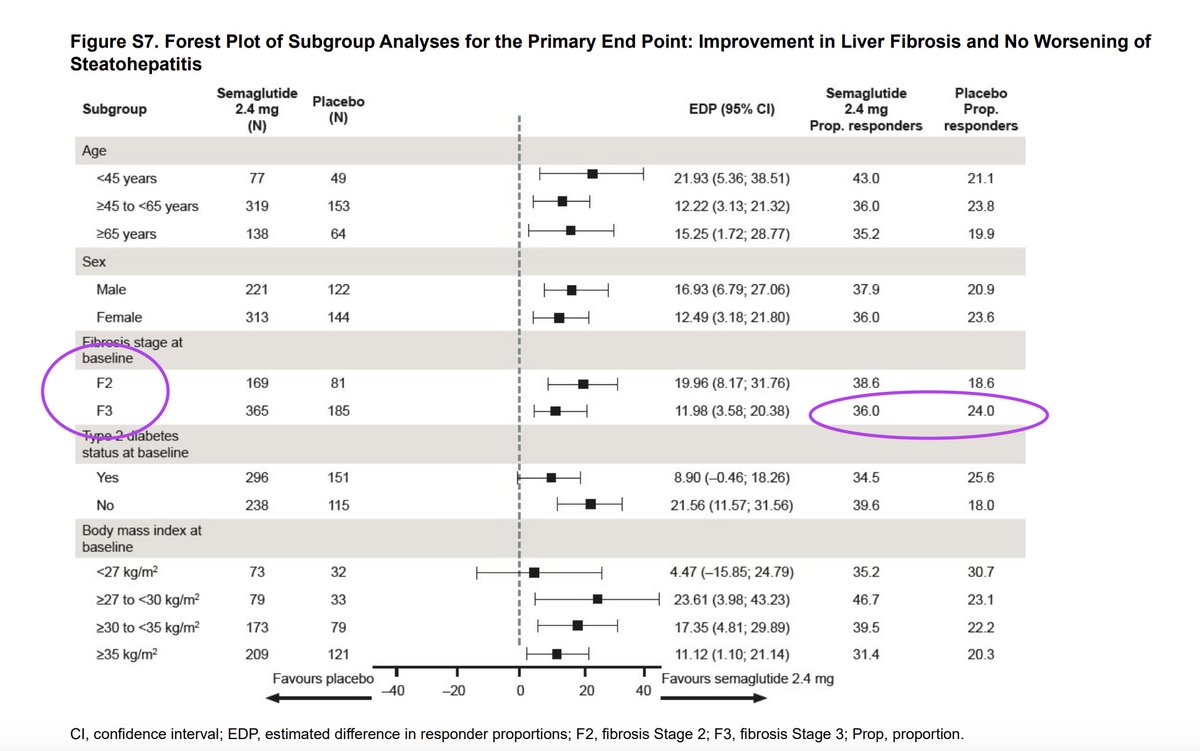
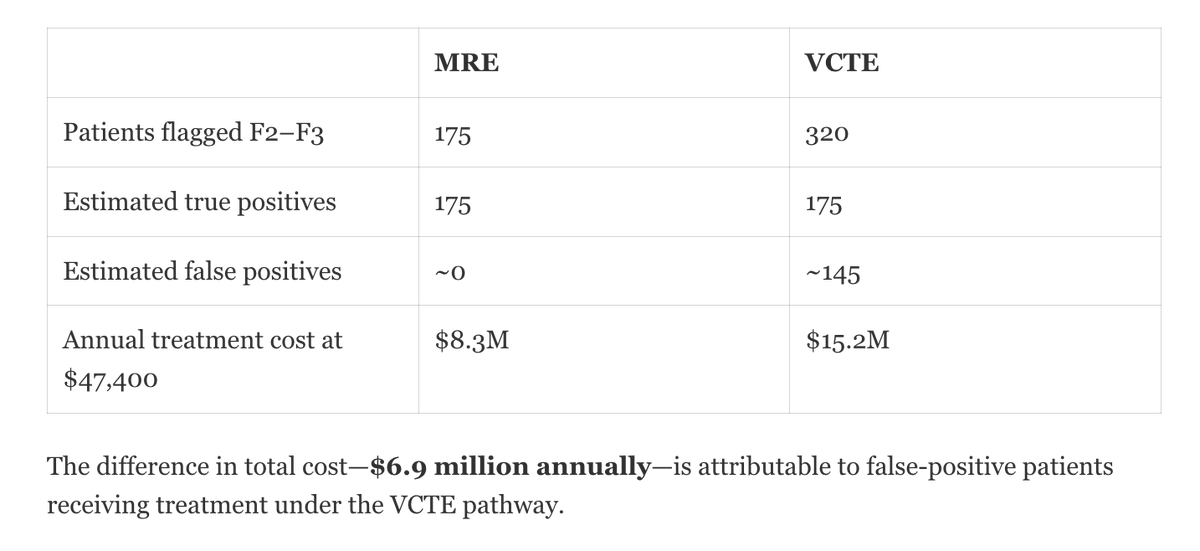
Resoundant, Inc. today announced that the U.S. Food and Drug Administration (FDA) has accepted the company’s Letter of Intent into the FDA’s Biomarker Qualification Program (BQP) for Liver Stiffness Measurement (LSM) by Magnetic Resonance Elastography (MRE) as a Reasonably Likely Surrogate Endpoint (RLSE) in clinical trials for adults with noncirrhotic metabolic dysfunction–associated steatohepatitis (MASH) with moderate to advanced fibrosis (F2–F3).
Acceptance into the BQP represents an important step in the FDA’s formal biomarker qualification process and reflects the agency’s openness to outcome-linked, non-invasive tests (NITs) that may improve the efficiency and feasibility of MASH drug development. The proposed context of use positions MRE as a quantitative biomarker intended to support assessment of fibrosis burden and its relationship to the risk of liver-related events and outcomes.
Fibrosis progression is the primary driver of adverse clinical outcomes in MASH. However, reliance on liver biopsy in clinical trials presents well-recognized challenges, including procedural risk, sampling variability, reader discordance, high cost, and barriers to patient recruitment and retention. These limitations have contributed to longer timelines and increased development complexity across the field.
MRE is an MRI-based technique that directly measures the mechanical properties of liver tissue and provides a continuous and reproducible assessment of fibrosis burden, enabling longitudinal evaluation without repeated invasive procedures. Advancing MRE within the FDA’s biomarker qualification framework has the potential to reduce reliance on biopsy and support more efficient, scalable, and patient-friendly clinical trial designs.
English