UTEP Biomedical Engineering retweetledi

Researchers at the Institute for Basic Science (IBS) in South Korea have identified a non-invasive technique to significantly enhance the brain's waste clearance mechanism.
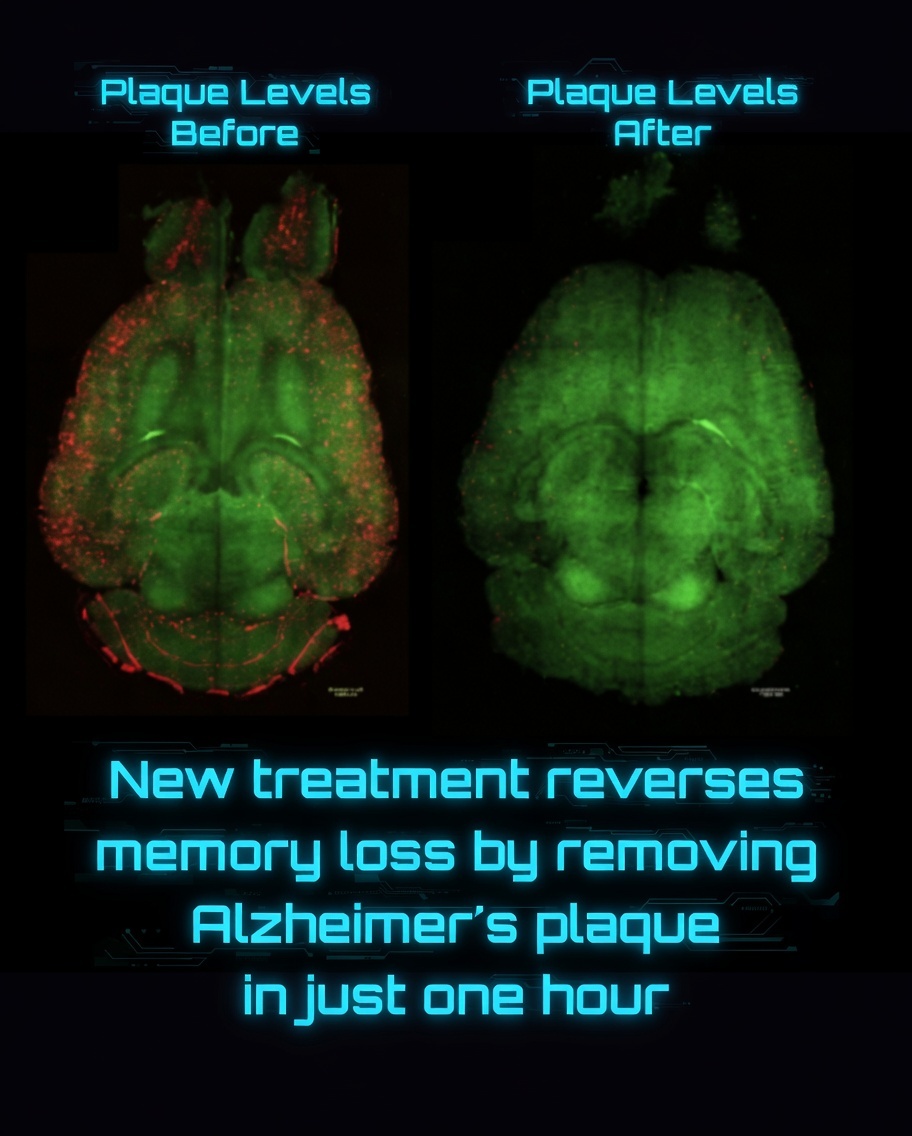
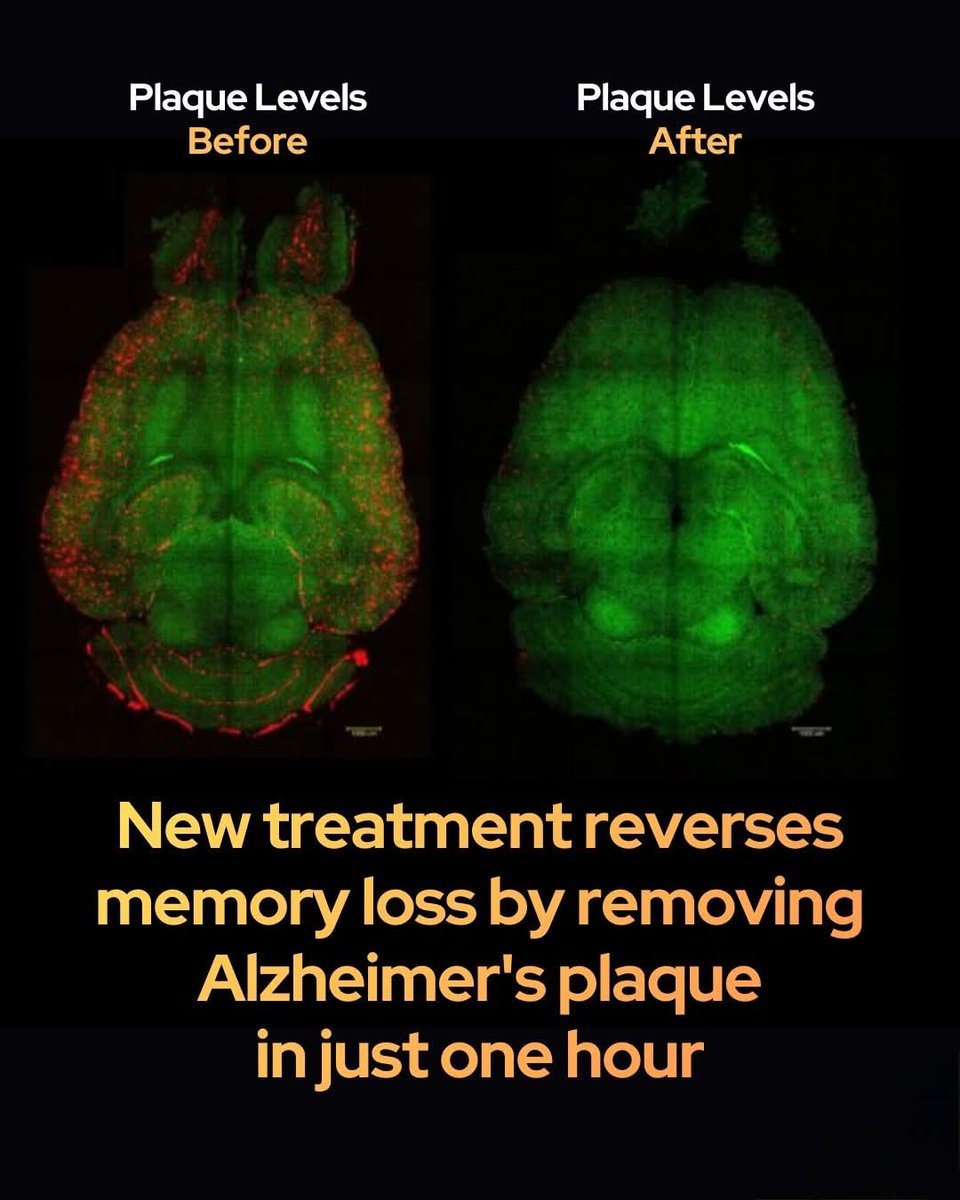
By applying gentle mechanical stimulation to the skin overlying lymphatic vessels in the face and neck, they dramatically increased cerebrospinal fluid (CSF) outflow in mice—effectively doubling drainage in many cases and fully restoring impaired clearance in aged animals to youthful levels.
This approach relies on a custom force-regulated mechanical device that lightly compresses and strokes the skin, targeting superficial cervical lymphatics without interfering with their natural contractions or requiring drugs or invasive procedures. The stimulation boosts CSF flow through previously underappreciated drainage pathways that connect the brain to superficial lymph nodes via facial, nasal, and palatal lymphatic networks—routes confirmed in both mice and primates, with strong implications for potential human relevance.
The discovery addresses age-related declines in CSF drainage, which contribute to the buildup of toxic proteins like amyloid-β and tau associated with Alzheimer's disease and other neurodegenerative conditions. By reversing drainage deficits in older mice, the method offers a promising proof-of-concept for improving brain health in aging populations.
The researchers emphasize that this remains preclinical work conducted in animal models, and human translation will require further studies to assess safety, optimal protocols, long-term benefits, and efficacy against diseases like Alzheimer's. Nonetheless, the findings suggest exciting possibilities for developing simple, wearable, or clinical devices—perhaps akin to targeted facial massage tools—to support natural brain detoxification and potentially slow neurodegeneration.
[Jin, H., Yoon, J.-H., Hong, S. P. et al. (2025). Increased CSF drainage by non-invasive manipulation of cervical lymphatics. Nature. DOI: 10.1038/s41586-025-09052-5]

English