Wassim Daoud Khatoun
104 posts

Wassim Daoud Khatoun
@WDKhatoun
GU Oncology Research Fellow @DanaFarber | MD @lebamericanuni | Aspiring Oncologist 🧬 | Healthcare advocate | he/him
Boston, MA Katılım Mart 2024
133 Takip Edilen91 Takipçiler

@ziremozay @YaleIMed @neerajaiims @umangtalking @montypal @maughanonc @NazliDizman @ReneeSaliby @huntsmancancer @nsayeghmd @ggebraelmd @LuisMezaco @ZeynepZengin @EbrahimiHedyeh @kor_shah @DrYukselUrun @osutcuoglu @BraunMDPhD Congrats Zeynep! Very well deserved
English
Wassim Daoud Khatoun retweetledi

I’m going to Yale!! #Match2026
Still can’t believe I matched at my #1 choice @YaleIMed
I'm so grateful to my mentors, friends, and family for supporting me every step of the way and believing in me!
@neerajaiims @umangtalking @montypal @maughanonc @NazliDizman @ReneeSaliby

English
Wassim Daoud Khatoun retweetledi

Pinch me!!! I’m going to be an OBGYN at my top choice @MethodistHosp 🌟
It truly takes a village and I’m endlessly grateful for the incredible support of my mentors, family, and friends 🩵
So excited for what’s ahead!!🤰🏻✨
#match2026 #obgyn

English

@MarcMachaalani @YaleIMed @DrChoueiri @BraunMDPhD @VincentWenxinXu @Clara__Steiner @eddy_saad @yekeduz_emre @ReneeSaliby @AminNassarMD @thenasheffect @TalalZarif1 Many congrats!! Very well deserved 👑
English
Wassim Daoud Khatoun retweetledi

Thrilled to have matched at @YaleIMed!
Beyond grateful to my amazing mentors, friends, and family for supporting me every step of the way! #Match2026


English
Wassim Daoud Khatoun retweetledi
Wassim Daoud Khatoun retweetledi

Honored to continue to bring Colleagues more Great Science out of #GU26 & Sending Our Congratulations and gratitude to @ASCO for another Amazing meeting
It is our Great Privilege to introduce One of our founding Faculty🚨 and dear friend Dr. Toni Choueiri of Dana farber🇺🇸discussing two Trials he was involved in:
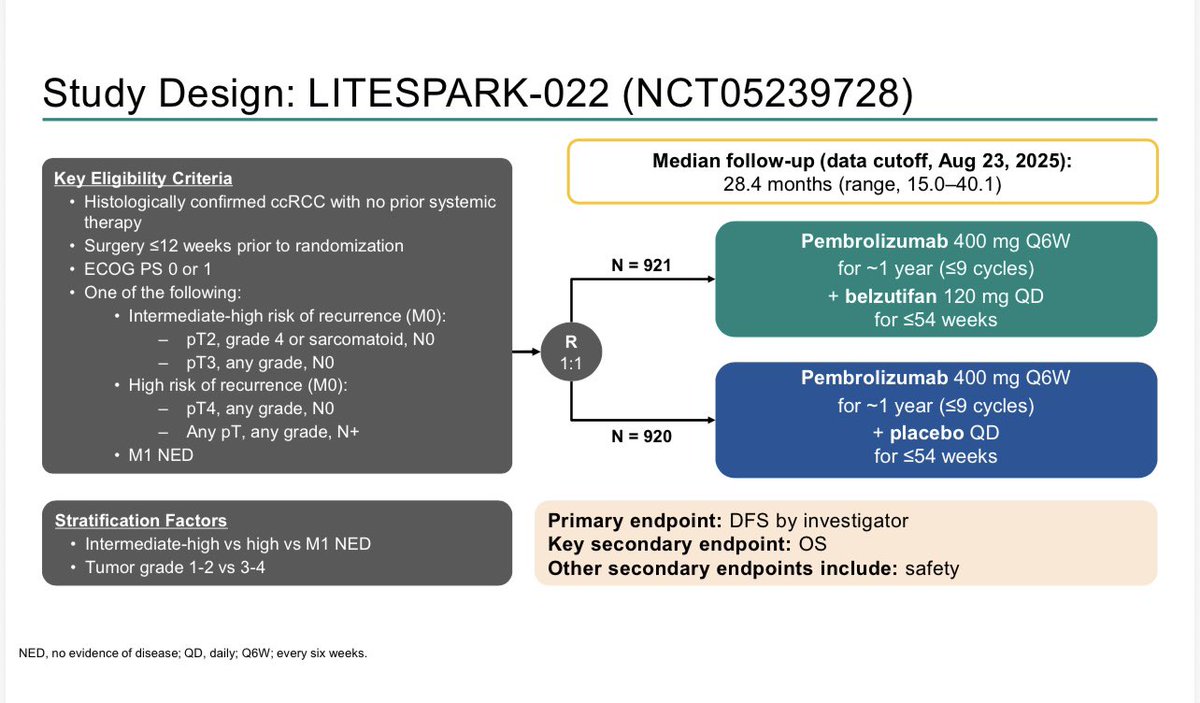
LITESPARK-022
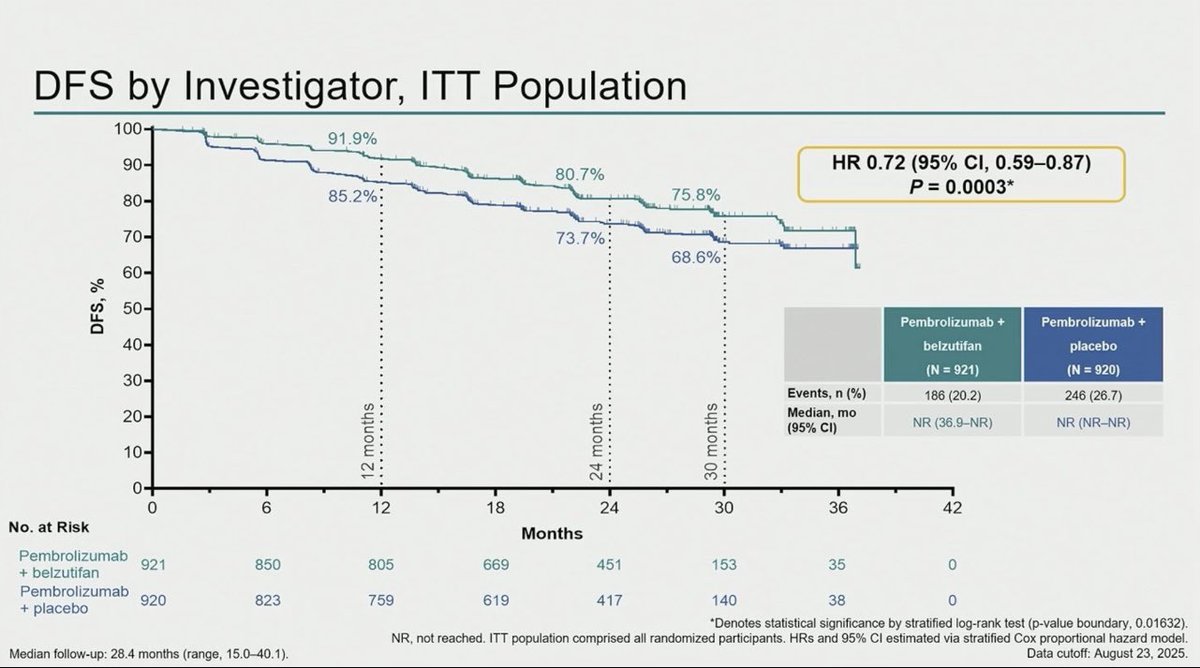
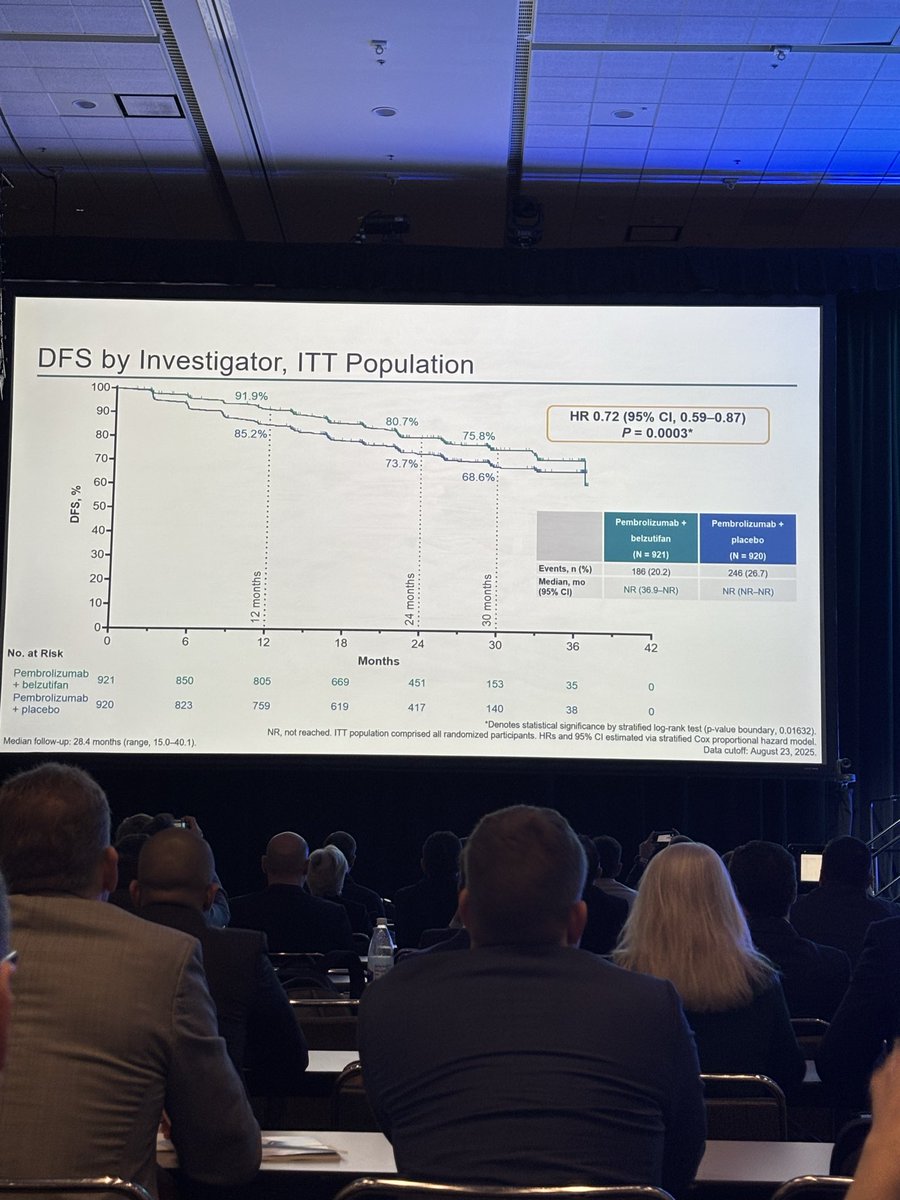
Adding belzutifan to pembrolizumab for one year in patients with high-risk clear cell RCC after nephrectomy significantly improved disease-free survival, reducing the risk of recurrence or death by 28% (HR 0.72) compared with pembrolizumab alone. At 24 months, DFS rates were 80.7% versus 73.7%. The combination increased grade ≥3 adverse events, notably anemia, hypoxia, and liver enzyme elevations, but these were generally manageable with dose modification and supportive care.
ARC-20
Evaluated casdatifan, a HIF-2α inhibitor, in heavily pretreated metastatic clear-cell RCC. Among 127 patients, objective response rate reached 31–35% with durable responses and median PFS up to 12.2 months. Deep, sustained serum erythropoietin reductions correlated with clinical benefit. Treatment was generally manageable, supporting phase 3 evaluation of casdatifan 100 mg daily.
@DrChoueiri
@motzermd
@DrIacovelli
@DrYukselUrun
@elena_verzoni
@tompowles1
@montypal
@crisbergerot
@DrDanielHeng
@apolo_andrea
@PGrivasMDPhD
@TiansterZhang
@neerajaiims
@amerseburger
@drenriquegrande
@BraunMDPhD
@cdanicas
@brian_rini
@AUC3_Official
@nataliagandur
@MarcMachaalani
@Mustafajsalehmd
@NazliDizman
@DrYukselUrun
@yekeduz_emre
@WeiweiBian
@ReneeSaliby
@eddy_saad
@Clara__Steiner
@marc_eid
@pablombarrios
@WDKhatoun
@Jad_El_Masri
@lilli_ash
@RashadNawfal
@RazaneHChehade
@elioibrahim8
@NafehGaelle
@shaye_carver
@zeyun_lu
@SylvanBacaLab
English
Wassim Daoud Khatoun retweetledi
Wassim Daoud Khatoun retweetledi
Wassim Daoud Khatoun retweetledi

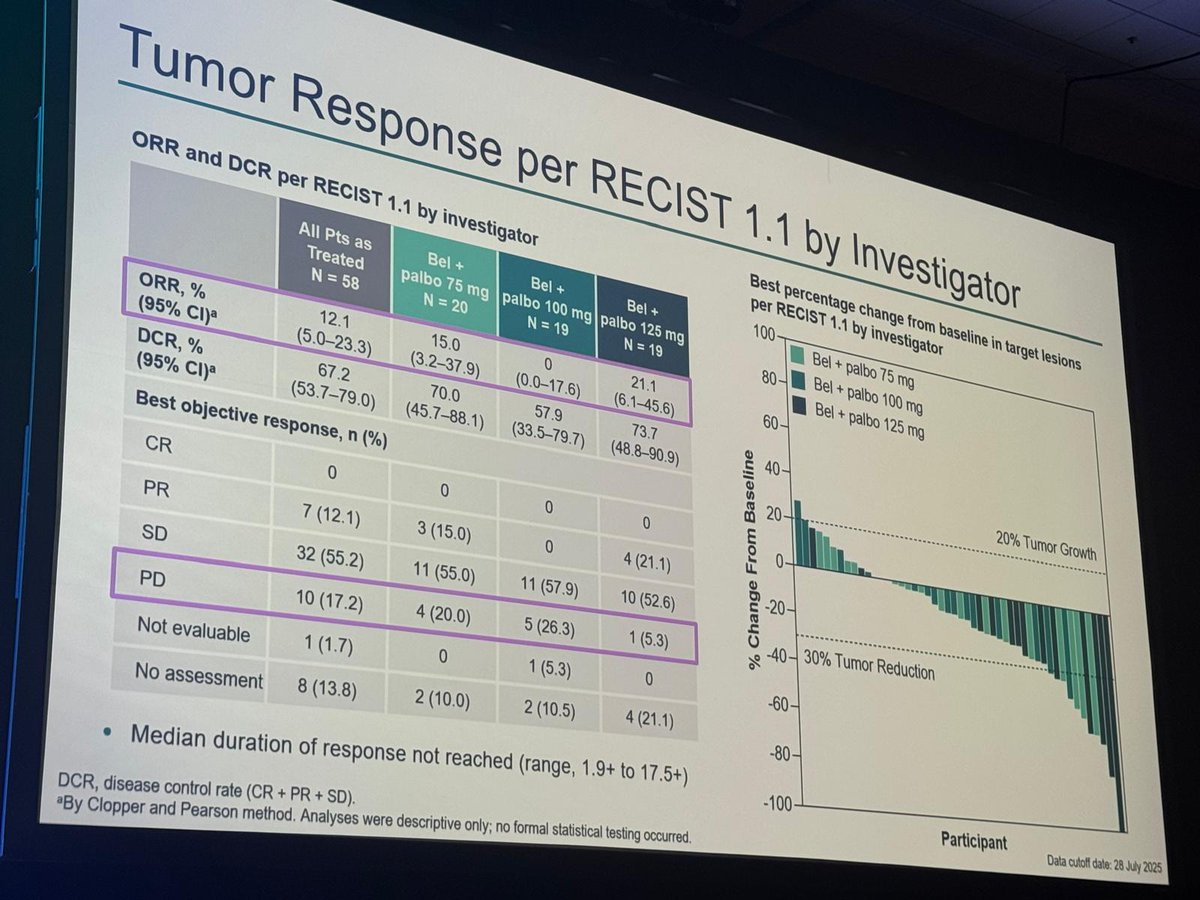
Great presentation by Dr. David McDermott on the LITESPARK-024 part 1 (phase 1/2) dose-escalation belzutifan + palbociclib in heavily pretreated advanced ccRCC:
- ORR: 21.1% (palbo 125 mg), DCR: 57.9-73.7% across dose levels
- mPFS: 9.1 months at the full dose of palbo
- DLTs: G3 anemia + G3 hypoxia (both at the highest dose)
Safety profile was manageable, but a larger cohort is needed to better investigate the added benefit.
#GU26
#RCC
@OncoAlert



English
Wassim Daoud Khatoun retweetledi

⭐️New era in adjuvant therapy for RCC
@DrChoueiri presenting LS-022:
Pembro+Belzutifan showing improved DFS vs Pembro (HR:072)
Exciting for 2026 and beyond!
#GU26 #ASCO @DrChoueiri @DanaFarber @OncoAlert



English
Wassim Daoud Khatoun retweetledi

Very proud of @Mustafajsalehmd @DanaFarber_GU presenting our work with @SashaGusevPosts ! Super star in the making.

English
Wassim Daoud Khatoun retweetledi

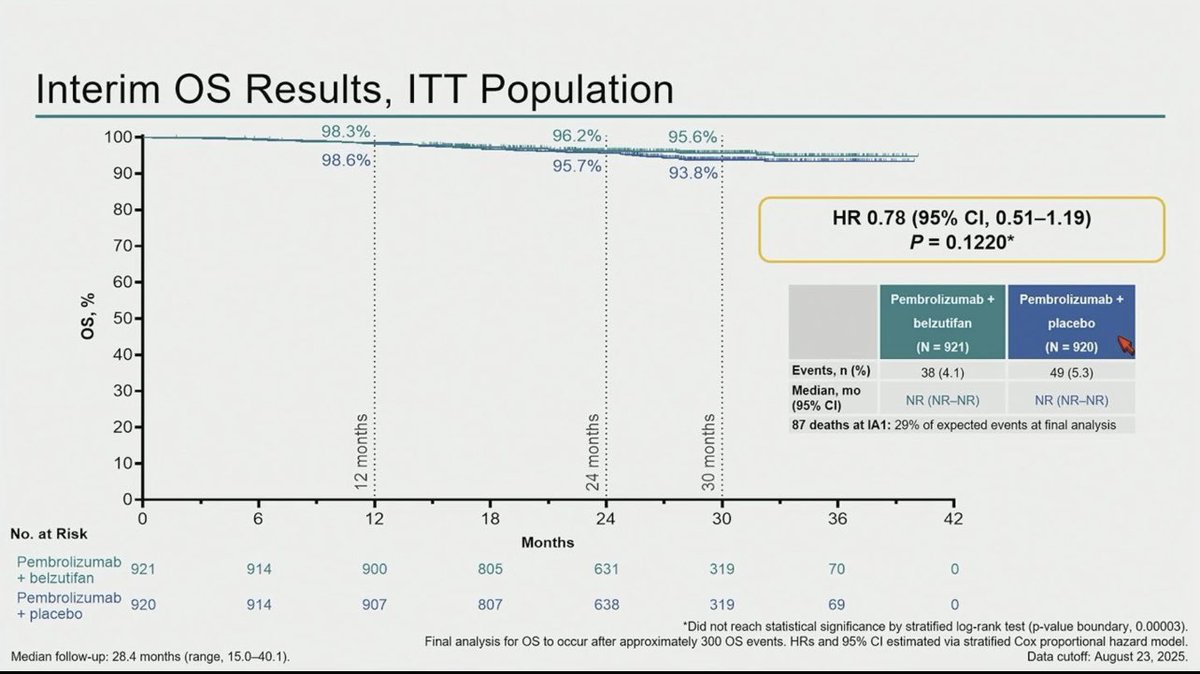
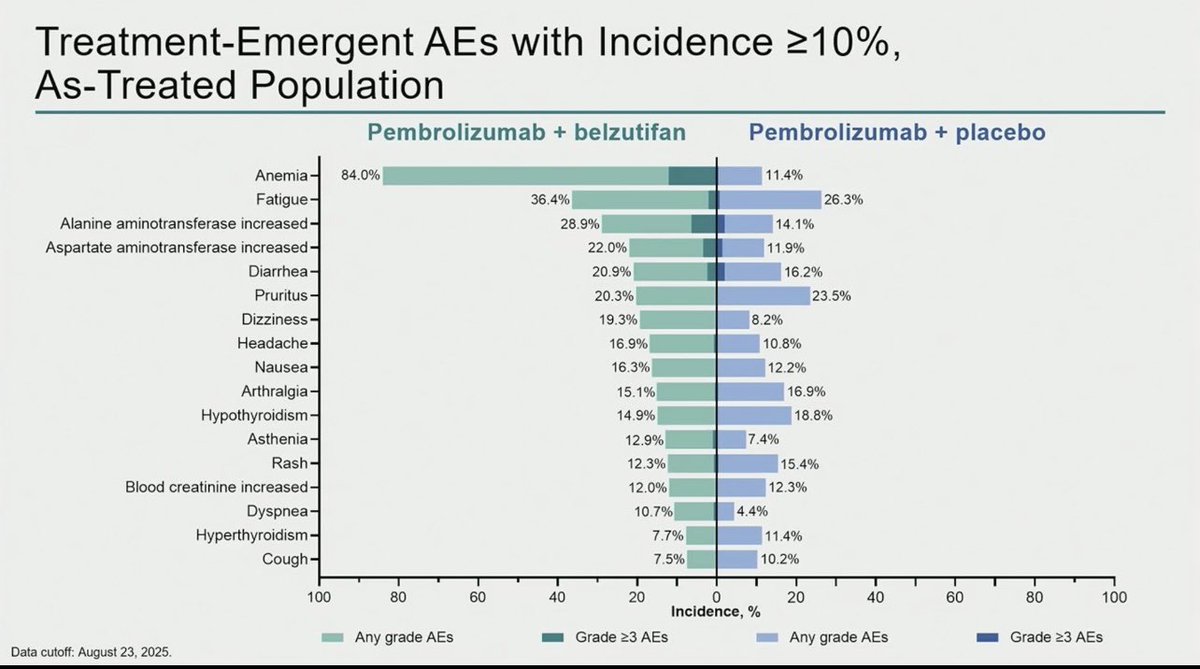
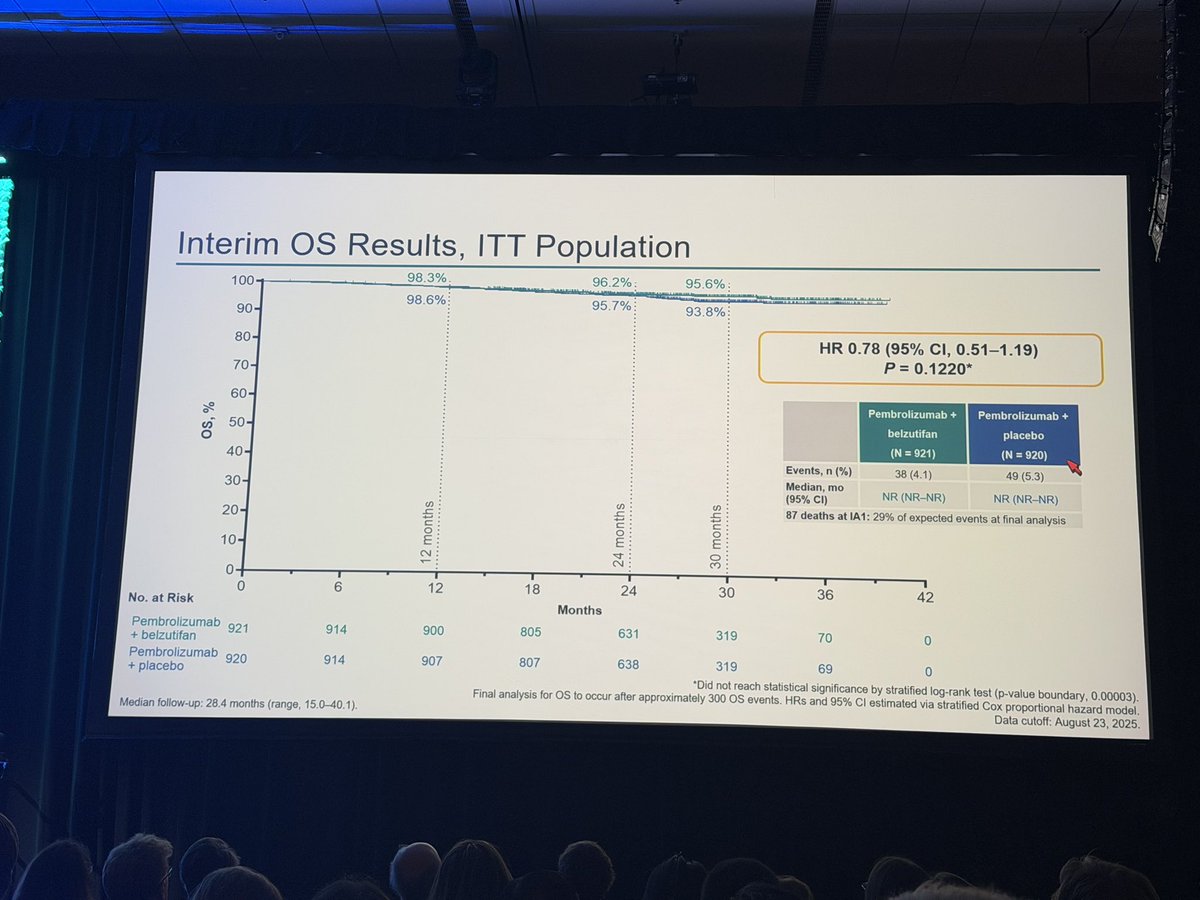
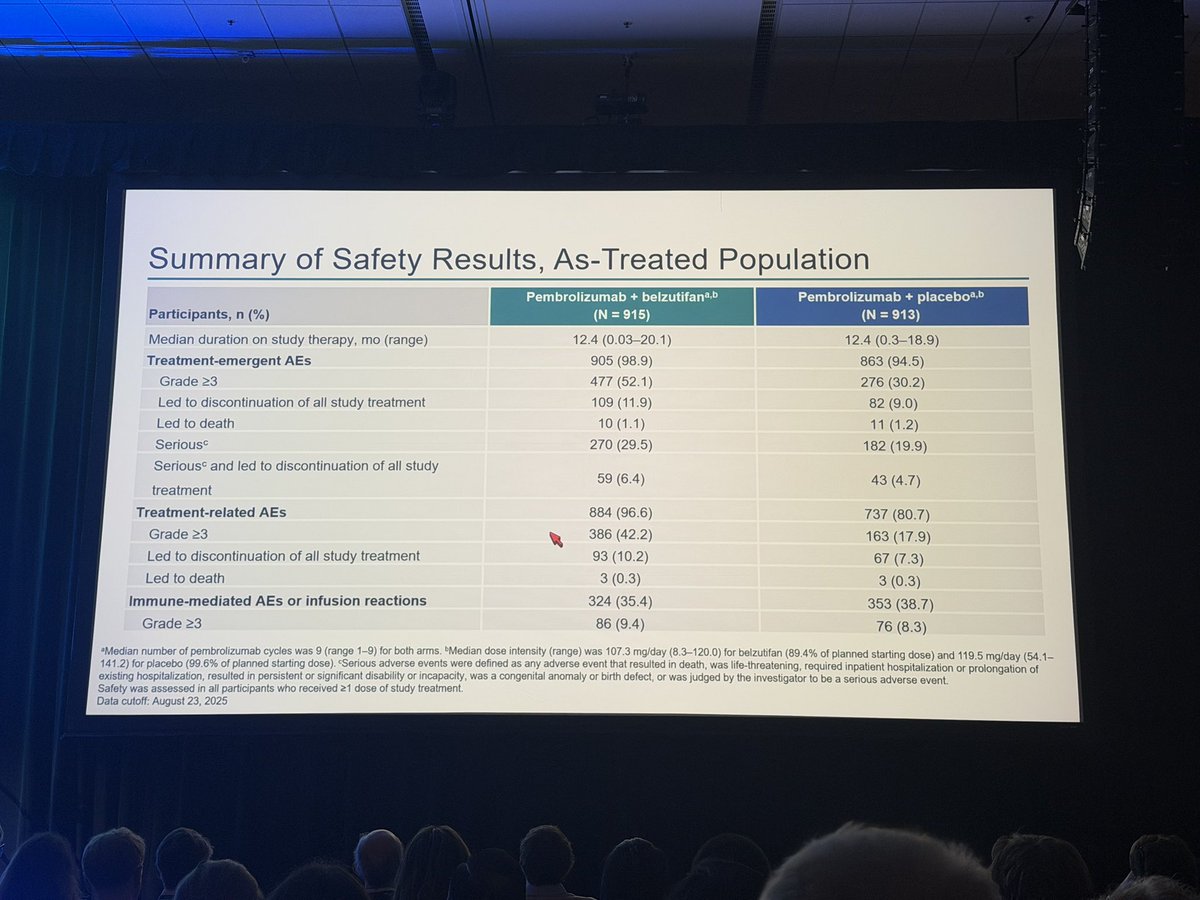
R3 LITESPARK-022: Adjuvant Pembrolizumab + Belzutifan vs Pembrolizumab for Clear Cell Renal Cell Carcinoma #GU26 showed significantly delayed DFS HR 0.72 (95% CI, 0.59–0.87) active across all subgroups. Immature OS (30 mnth OS 93% CDs 96%). ⬆️ Anaemia fatigue and transaminitis but only 4% discontinuation rate for belzutifan. The shape of the DFS curve and early OS trending the right way looks like initial analysis of KN564 and we know where that ended, making pem/bel attractive. @OncoAlert @DrChoueiri
English
Wassim Daoud Khatoun retweetledi

Adjuvant pembrolizumab + belzutifan improved DFS vs pembrolizumab alone in high risk resected ccRCC.
A meaningful step. Selection and follow up will be key.
Proud to be an investigator in this milestone. @DrChoueiri @tompowles1
#gu26 #cancer #oncology #ASCO @oncoalert @ASCO
@yekeduz_emre @CParkMD @OncBrothers @Larvol @nataliagandur @crisbergerot
@ONCOassist @IMG_Oncologists


English
Wassim Daoud Khatoun retweetledi

A little history recap:
2021: KN564 is positive
2024: KN564 shows the first OS improvement in adjuvant therapy in ANY type of cancer
2026: let's do better than KN564, pembro/bel > pembro
@DrChoueiri
English

Once again @DrChoueiri delivers practice-changing data in ccRCC.
In LITESPARK-022, adjuvant pembro+bel improved DFS vs pembro+placebo post-nephrectomy (HR 0.72, p=0.0003). Exceptional presentation by a great mentor!
@ASCO #GU26 @DanaFarber @DanaFarber_GU @OncoAlert @urotoday


English
Wassim Daoud Khatoun retweetledi

Phase 3 LITESPARK-011: Belzutifan + Lenvatinib vs Cabozantinib in pretreated metastatic clear cell renal cancer #GU25 shows +ve PFS HR 0.75 , OS HR 0.85 (non-significant), ⬆️ RR 53% vs 40%, G3+ TRAEs 72% vs 66%. Cabozantinib is hard to beat making this a standard of care. Using the most active drugs first rather than sequencing is debatable, but not all patients can sequence. We still don’t have significant OS post in IO refractory RCC @OncoAlert


English
Wassim Daoud Khatoun retweetledi

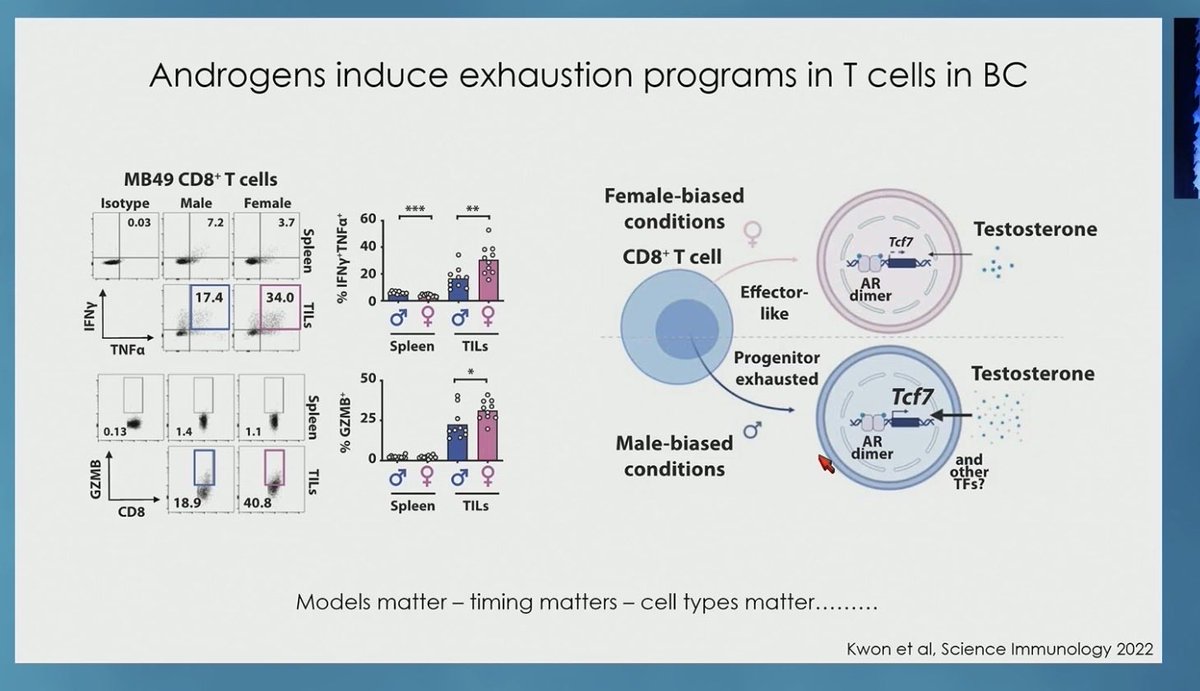
@ImmunoBladder Dr. Molly Ingersoll highlights many hormonal and immunologic differences in women with #BladderCancer. Should we be developing gender specific biomarkers and therapies? @ASCO #GU26



English