walter tambke
198 posts


walter tambke
@WTambke
Tru steel buildings, rhino mini storage, Peach state academy daycares, fire station express car wash, Thors wine and sprints and castle dry cleaners,











$LQDA Judge Andrews has no court hearings for the rest of the week. I'm expecting a ruling from him by Friday. This trial will get posted in the CJRA (list of shame) on the 31st of March if no ruling. He has been on this list once before and ruled 3 weeks later.






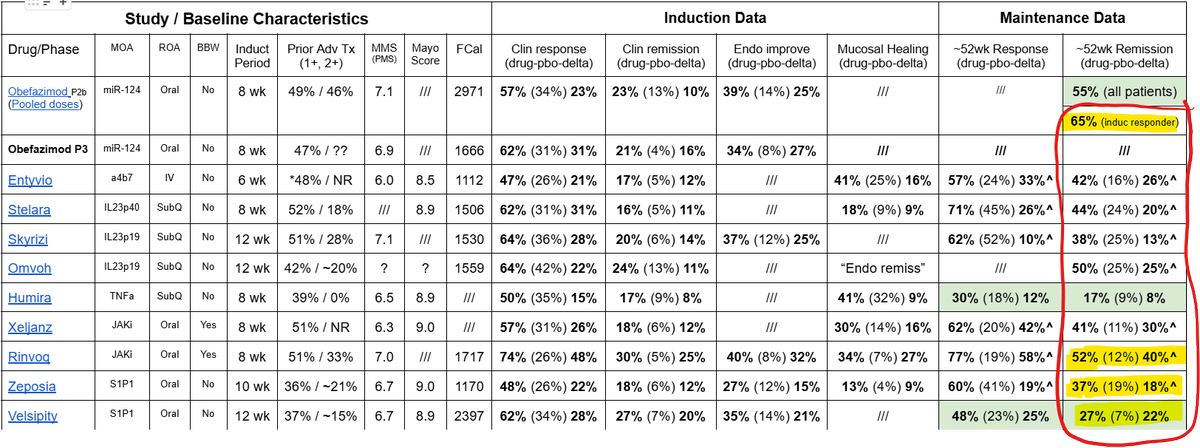
Lot's of requests to discuss the Wedbush report on $ABVX today. It's a doozy. Obviously I'm a biased bull so sure, take my thoughts with a grain of salt, but this is truly, awful work from my perspective. Let's dig in on a few of the key issues.

$ABVX too risky to miss BO. Back in with size.

We are short Paris-listed hotel chain Accor SA $AC.PA $AC “Accor, Epstein, and Ukrainian Orphans: Our Investigation Into Child Trafficking” Our new report is available on our website: grizzlyreports.com/accor/