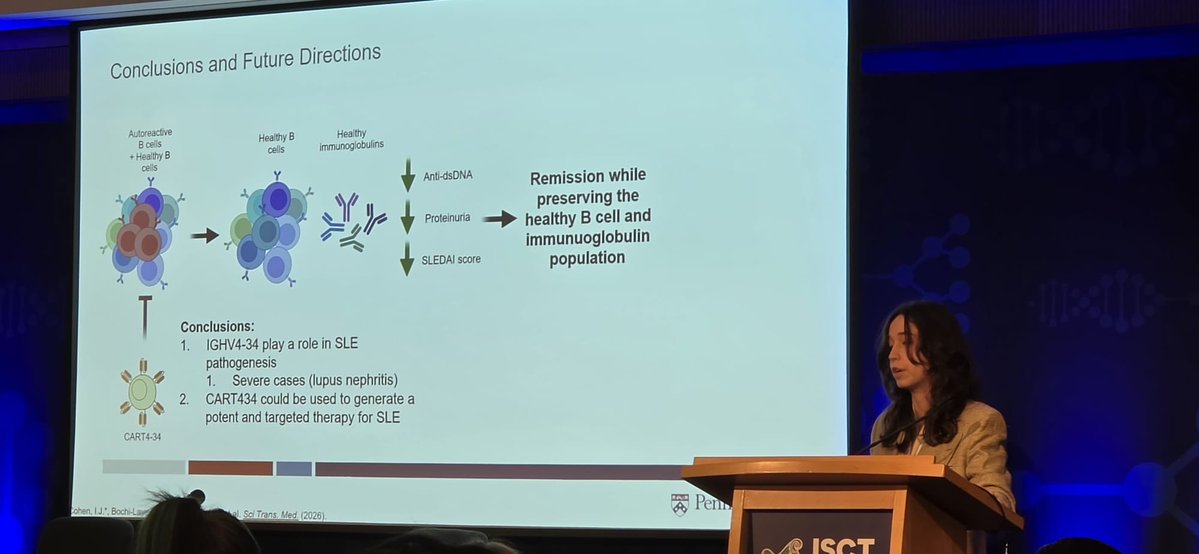
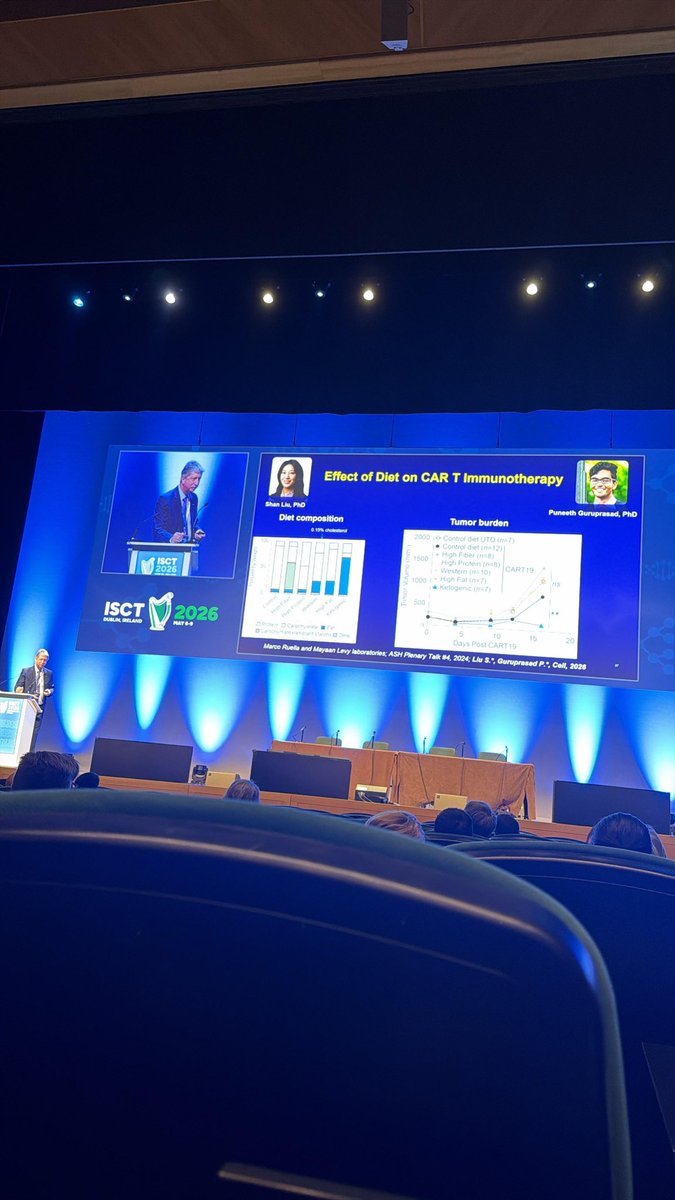
Sabitlenmiş Tweet

🎙️Excited to announce the website for Ring That Bell 🔔 which I co-wrote w internationally acclaimed Irish country music artist @mags__mccarthy. It tells the emotional story of a cancer patient facing relapse and the fierce advocacy, support, and groundbreaking science that offer new hope.
#cancer #cancerresearch #immunotherapy #hope
ringthatbellsong.com

English